


PALTUSOTINE
CAS 2172870-89-0
- CRN00808
- F2IBD1GMD3
WeightAverage: 456.497
Monoisotopic: 456.17616767
Chemical FormulaC27H22F2N4O
3-[4-(4-Amino-1-piperidinyl)-3-(3,5-difluorophenyl)-6-quinolinyl]-2-hydroxybenzonitrile
fda 2025, approvals 2025
To treat acromegaly in adults who had an inadequate response to surgery and/or for whom surgery is not an option
- OriginatorCrinetics Pharmaceuticals
- ClassAmines; Antineoplastics; Antisecretories; Fluorobenzenes; Nitriles; Piperidines; Quinolines; Small molecules
- Mechanism of ActionSomatostatin receptor 2 agonists
- Orphan Drug Status – Acromegaly
- PreregistrationAcromegaly
- Phase IIMalignant carcinoid syndrome
- 08 May 2025Crinetics Pharmaceuticals expects potential EMA decision for paltusotine in Acromegaly, in the first half of 2026
- 08 May 2025FDA assigns PDUFA action date of 25/09/2025 for paltusotine for acromegaly
- 08 May 2025Crinetics Pharamceuticals plans the phase III CAREFNDR trial for Malignant carcinoid syndrome (PO), in the second quarter of 2025
Paltusotine is a selective somatostatin receptor type 2 (SST2) agonist in development by Crinetics Pharmaceuticals for the treatment of acromegaly and certain neuroendocrine tumors. It is a small molecule delivered orally.[1][2][3][4]
SCHEME
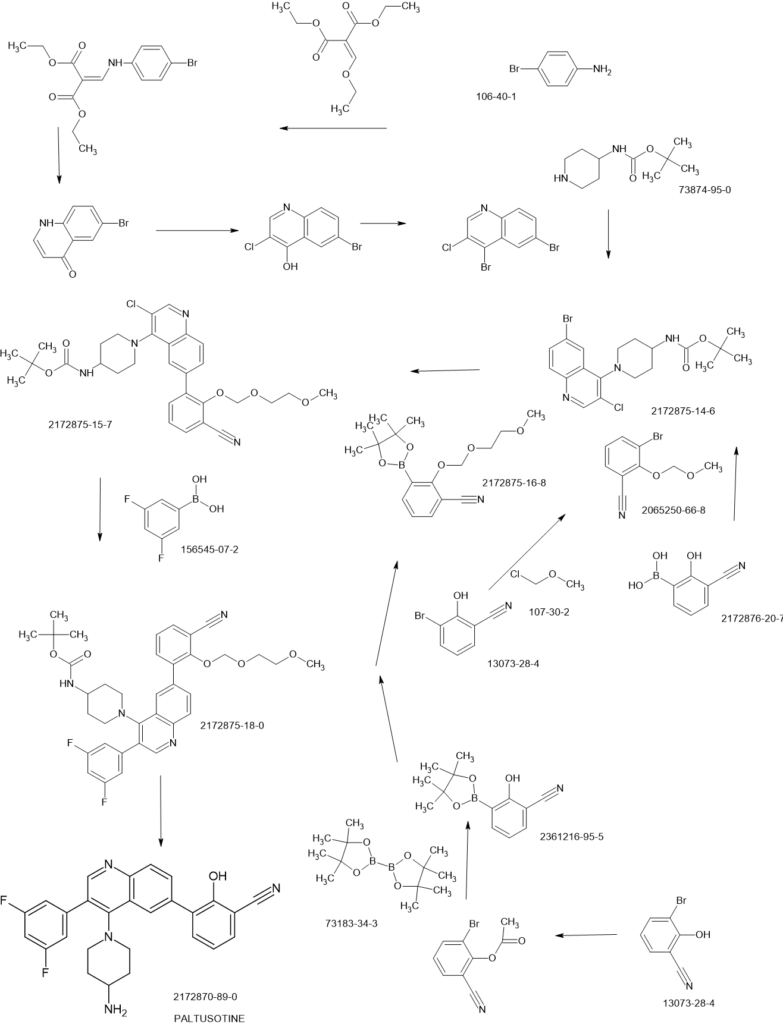

PAPER
https://pubs.acs.org/doi/10.1021/acsmedchemlett.2c00431
Discovery of Paltusotine (CRN00808), a Potent, Selective, and Orally Bioavailable Non-peptide SST2 Agonist


Step 2-1, preparation of [1-(6-bromo-3-chloro-quinolin-4-yl)-piperidin-4-yl]-carbamic acid tertbutyl ester: To a DMSO solution of 6-bromo-3,4-dichloroquinoline (950 mg, 1 Eq, 3.43 mmol)
was added tert-butyl piperidin-4-ylcarbamate (841 mg, 98% Wt, 1.2 Eq, 4.12 mmol) and DIPEA
(1.19 g, 1.60 mL, 3 Eq, 10.3 mmol). The resulting mixture was heated at 60 °C for overnight.
The reaction crude was quenched with water, extracted with EtOAc, washed with brine,
concentrated and purified by silica gel chromatography to afford tert-butyl (1-(6-bromo-3-
chloroquinolin-4-yl)piperidin-4-yl)carbamate (0.95 g, 2.2 mmol, 63 %) as an off-white solid. 1H
NMR (500 MHz, CDCl3) δ 8.66 (s, 1H), 8.25 (d, J=5 Hz, 1H), 7.94 (d, J=10 Hz, 1H), 7.74 (d,
J=10 Hz, 1H), 4.61 (s, 1H), 3.76 (s, 1H), 3.51 (m, 2H), 3.37 (m, 2H), 2.13-2.15 (m, 2H), 1.73-
1.65 (m, 2H), 1.48 (s, 9H). MS [M+H]
+= 442.0.
Step 4-2, preparation of 1-{3-chloro-6-[3-cyano-2-(2-methoxy-ethoxymethoxy)-phenyl]-
quinolin-4-yl}-piperidin-4-yl)-carbamic acid tert-butyl ester: To a THF (5.0 mL) solution of [1-
(6-bromo-3-chloro-quinolin-4-yl)-piperidin-4-yl]-carbamic acid tert-butyl ester (1.0 mmol, 440
mg) and 2-(2-methoxy-ethoxymethoxy)-3-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-
benzonitrile (1.4 eq., 1.4 mmol, 460 mg) was added PdCl2dppf (0.1 eq., 0.1 mmol, 75 mg) and
KOAc (3.0 eq., 3.0 mmol, 300 mg). N2 was bubbled through the reaction solution for 5 min and
0.5 mL water was added. The resulting mixture was heated at 80 °C for 1 h. LCMS analysis
showed about 50% of the starting material has been converted to the desired product. Additional
2-(2-methoxy-ethoxymethoxy)-3-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-benzonitrile
(1.4 eq., 1.4 mmol, 460 mg), PdCl2dppf (0.1 eq., 0.1 mmol, 75 mg) and KOAc (3.0 eq., 3.0
mmol, 300 mg) were added and the resulting solution was heated at 80 °C for another 2 h. The
reaction solution was combined with silica gel and concentrated. The residue obtained was
purified by silica gel chromatography eluting with ethyl acetate/hexane (0~50%) to give 0.512 g
of the desired product as white solid. MS [M+H]
+= 567.6.
Step 4-3, preparation of {1-[6-[3-cyano-2-(2-methoxy-ethoxymethoxy)-phenyl]-3-(3,5-difluorophenyl)-quinolin-4-yl]-piperidin-4-yl}-carbamic acid tert-butyl ester: To a dioxane (5 mL)
solution of (1-{3-chloro-6-[3-cyano-2-(2-methoxy-ethoxymethoxy)-phenyl]-quinolin-4-yl}-
piperidin-4-yl)-carbamic acid tert-butyl ester (0.5 mmol, 283 mg) was added Pd(amphos)Cl2 (0.1
eq., 0.05 mmol, 37 mg), 3, 5-difluorophenyl boronic acid (3.0 eq., 1.5 mmol, 250 mg) and
K2CO3 (4.0 eq., 2.0 mmol, 276 mg). N2 was bubbled through the reaction solution for 5 min and
0.5 mL water was added. The resulting mixture was heated at 95 °C for 0.5 h and LCMS analysis
showed that starting material was completely consumed. The reaction solution was concentrated
with silica gel and purified by silica gel chromatography eluting with ethyl acetate/hexane
(0~50%) to give 0.170 g of the desired product as white solid. MS (M+H)+= 645.6.
Step 4-4, preparation of 3-[4-(4-amino-piperidin-1-yl)-3-(3,5-difluoro-phenyl)-quinolin-6-yl]-2-hydroxybenzonitrile: to the dichloromethane (5.0 mL) solution of {1-[6-[3-cyano-2-(2-methoxyethoxymethoxy)-phenyl]-3-(3,5-difluoro-phenyl)-quinolin-4-yl]-piperidin-4-yl}-carbamic acid
tert-butyl ester (0.264 mmol, 170 mg) was added trifluroroacetic acid (2.0 mL) and the resulting
mixture was stirred at ambient temperature for 2 h. The reaction solution was concentrated and
purified by C18 reversed phase chromatography eluting with MeCN/water (0~40%). Pure
fractions were combined, neutralized with saturated NaHCO3, extracted with ethyl acetate and
dried with MgSO4. The organic solution was concentrated with HCl in ether (2.0 M) to give the
final compound as HCl salt (68 mg, 0.138 mmol, 52%).
1H NMR (500 MHz, DMSO-d6) δ 10.77
(br s, 1H), 8.78 (s, 1H), 8.29-8.15 (m, 5H), 7.79 (dd, J=20 Hz, 5 Hz, 2H), 7.41 (m, 1H), 7.26-
7.19 (m, 3H), 3.59 (t, J=12 Hz, 2H), 3.31 (m, 1H), 3.00 (t, J=12 Hz, 2H), 2.05-1.99 (m, 2H),
1.76-1.74 (m, 2H). MS [M+H]
+= 457.5. 13C NMR (DMSO-d6) δ 30.2, 47.4, 50.8, 102.4, 103.2,
113.4, 117.2, 121.4, 124.6, 130.7, 133.1, 134.6, 136.0, 141.7, 156.6, 161.2, 163.2. LCMS purity
98% (254&220 nM). HRMS m/z [M+H]+ Calcd for C27H23F2N4O 457.1834; found 457.1833.
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US235548187&_cid=P20-MCSHXW-73235-1
PATENTS
WO2021011641
WO2018013676
References
- ^ Madan, Ajay; Markison, Stacy; Betz, Stephen F.; Krasner, Alan; Luo, Rosa; Jochelson, Theresa; Lickliter, Jason; Struthers, R. Scott (April 2022). “Paltusotine, a novel oral once-daily nonpeptide SST2 receptor agonist, suppresses GH and IGF-1 in healthy volunteers”. Pituitary. 25 (2): 328–339. doi:10.1007/s11102-021-01201-z. PMC 8894159. PMID 35000098.
- ^ Zhao, Jian; Wang, Shimiao; Markison, Stacy; Kim, Sun Hee; Han, Sangdon; Chen, Mi; Kusnetzow, Ana Karin; Rico-Bautista, Elizabeth; Johns, Michael; Luo, Rosa; Struthers, R. Scott; Madan, Ajay; Zhu, Yunfei; Betz, Stephen F. (12 January 2023). “Discovery of Paltusotine (CRN00808), a Potent, Selective, and Orally Bioavailable Non-peptide SST2 Agonist”. ACS Medicinal Chemistry Letters. 14 (1): 66–74. doi:10.1021/acsmedchemlett.2c00431. PMC 9841592. PMID 36655128.
- ^ Gadelha, Monica R; Gordon, Murray B; Doknic, Mirjana; Mezősi, Emese; Tóth, Miklós; Randeva, Harpal; Marmon, Tonya; Jochelson, Theresa; Luo, Rosa; Monahan, Michael; Madan, Ajay; Ferrara-Cook, Christine; Struthers, R Scott; Krasner, Alan (13 April 2023). “ACROBAT Edge: Safety and Efficacy of Switching Injected SRLs to Oral Paltusotine in Patients With Acromegaly”. The Journal of Clinical Endocrinology & Metabolism. 108 (5): e148 – e159. doi:10.1210/clinem/dgac643. PMC 10099171. PMID 36353760. S2CID 253445337.
- ^ Zhao, Jie; Fu, Hong; Yu, Jingjing; Hong, Weiqi; Tian, Xiaowen; Qi, Jieyu; Sun, Suyue; Zhao, Chang; Wu, Chao; Xu, Zheng; Cheng, Lin; Chai, Renjie; Yan, Wei; Wei, Xiawei; Shao, Zhenhua (21 February 2023). “Prospect of acromegaly therapy: molecular mechanism of clinical drugs octreotide and paltusotine”. Nature Communications. 14 (1): 962. Bibcode:2023NatCo..14..962Z. doi:10.1038/s41467-023-36673-z. ISSN 2041-1723. PMC 9944328. PMID 36810324.
| Legal status | |
|---|---|
| Legal status | Investigational |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 2172870-89-0 |
| PubChem CID | 134168328 |
| ChemSpider | 81367268 |
| UNII | F2IBD1GMD3 |
| Chemical and physical data | |
| Formula | C27H22F2N4O |
| Molar mass | 456.497 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
////////PALTUSOTINE, ORPHAN DRUG, Acromegaly, CRN 00808, F2IBD1GMD3














