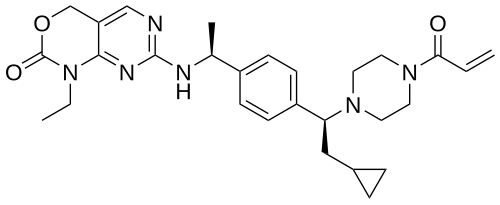
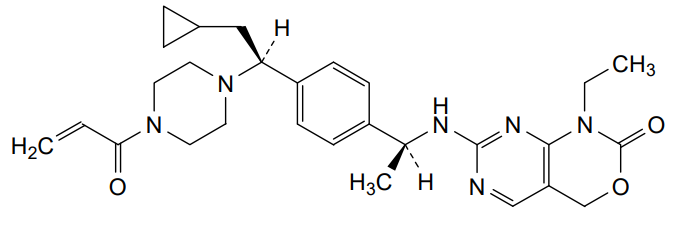
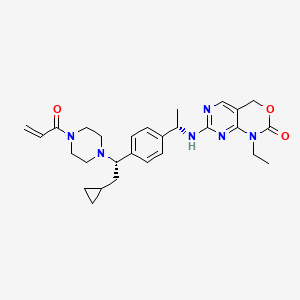
Crelosidenib
CAS 2230263-60-0
7-{[(1S)-1-(4-{(1S)-1-[4-(prop-2-enoyl)piperazin-1-yl]-2-cyclopropylethyl}phenyl)ethyl]amino}-1-ethyl-1,4-dihydro-2Hpyrimido[4,5-d][1,3]oxazin-2-one
isocitrate dehydrogenase 1 (IDH1) inhibitor, antineoplastic
MF C28H36N6O3 MW 504.6 g/mol
- LY3410738
- 7-[[(1S)-1-[4-[(1S)-2-cyclopropyl-1-(4-prop-2-enoylpiperazin-1-yl)ethyl]phenyl]ethyl]amino]-1-ethyl-4H-pyrimido[4,5-d][1,3]oxazin-2-one
- 7-(((1S)-1-(4-((1S)-2-cyclopropyl-1-(4-prop-2-enoylpiperazin-1-yl)ethyl)phenyl)ethyl)amino)-1-ethyl-4H-pyrimido(4,5-d)(1,3)oxazin-2-one
Crelosidenib is an investigational new drug that is being evaluated for the treatment of cancer. It acts as a selective inhibitor of isocitrate dehydrogenase 1 (IDH1), an enzyme that plays a crucial role in cellular metabolism and is frequently mutated in various cancers, including cholangiocarcinoma.[1][2]
Crelosidenib is an orally available inhibitor of mutant form of the isocitrate dehydrogenase type 1 (IDH1; IDH-1; IDH1 [NADP+] soluble), including the substitution mutation at arginine (R) in position 132, IDH1(R132), with potential antineoplastic activity. Upon oral administration, crelosidenib specifically and covalently binds to and modifies a single cysteine (Cys269) in the allosteric binding pocket of mutant forms of IDH1, thereby inactivating IDH1. This inhibits the formation of the oncometabolite 2-hydroxyglutarate (2HG) from alpha-ketoglutarate (a-KG). This depletes 2-HG levels, prevents 2HG-mediated signaling and leads to both an induction of cellular differentiation and an inhibition of cellular proliferation in tumor cells expressing mutant forms of IDH1. In addition, crelosidenib has the ability to cross the blood-brain barrier (BBB). IDH1 mutations, including IDH1(R132) mutations, are highly expressed in certain malignancies, including gliomas; they initiate and drive cancer growth by both blocking cell differentiation and catalyzing the formation of 2HG.
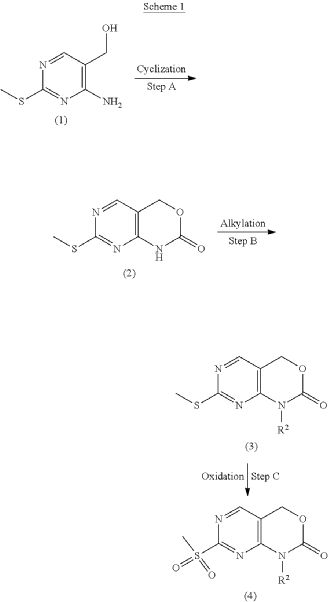
Syn
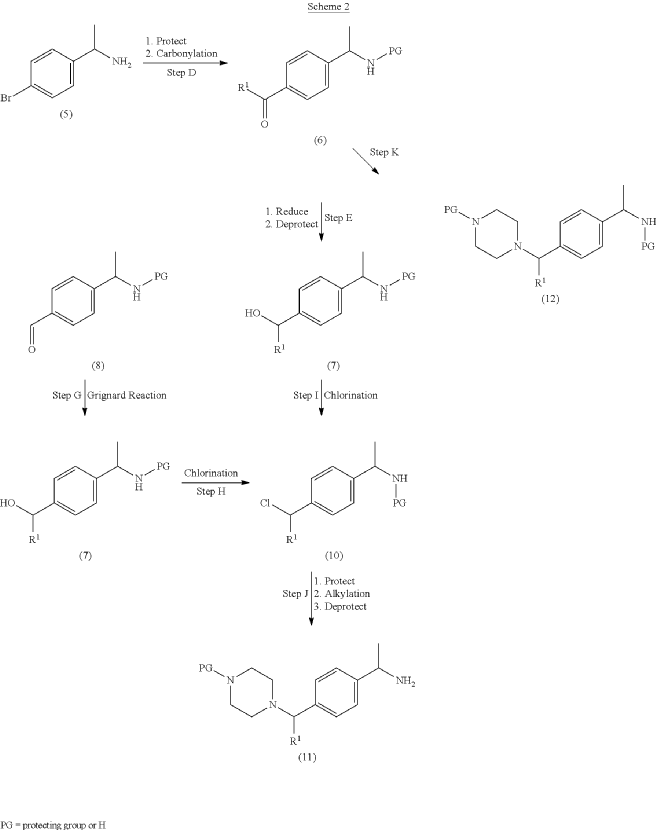
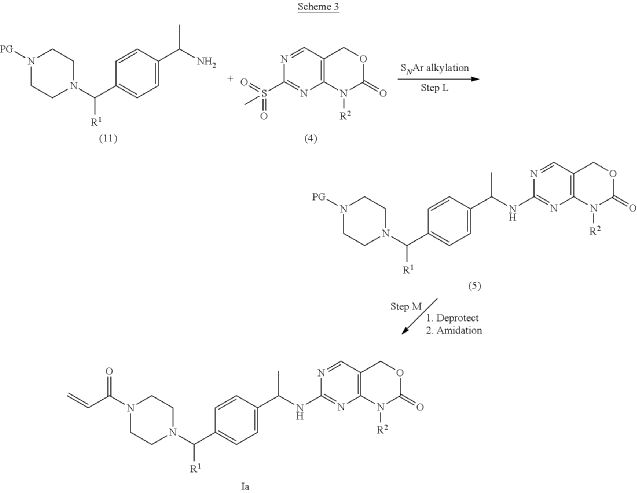
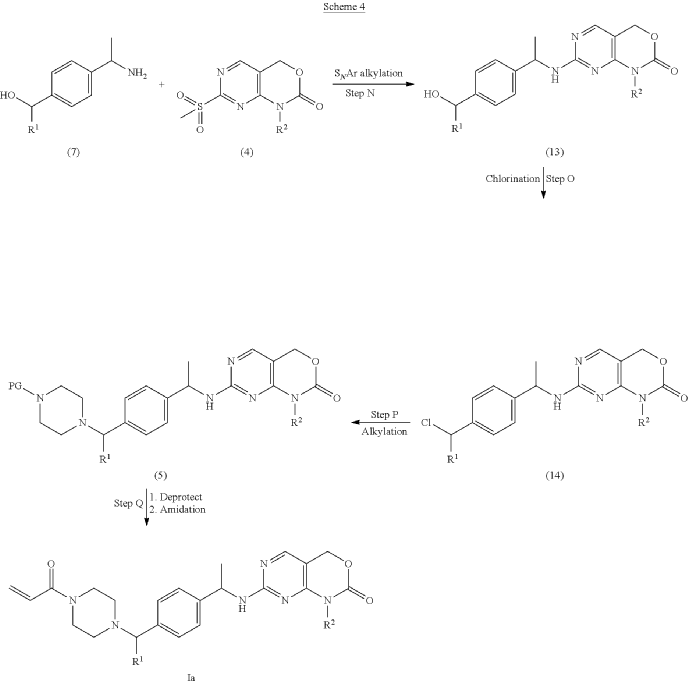
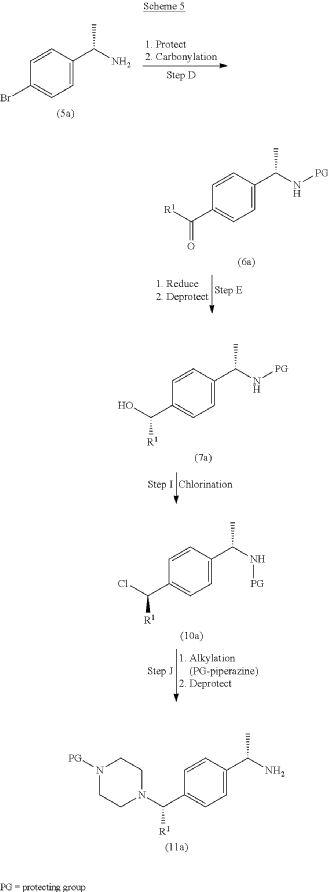
example 2 [US11001596B2]
https://patentscope.wipo.int/search/en/detail.jsf?docId=US289829390&_cid=P12-MG4UBU-88518-1

PAT
- 7-phenylethylamino-4h-pyrimido[4,5-d][1,3]oxazin-2-one compounds as mutant idh1 and idh2 inhibitorsPublication Number: US-2021206780-A1Priority Date: 2016-12-16
- 7-phenylethylamino-4h-pyrimido[4,5-d][1,3]oxazin-2-one compounds as mutant idh1 and idh2 inhibitorsPublication Number: US-2021230185-A1Priority Date: 2016-12-16
- 7-phenylethylamino-4h-pyrimido[4,5-d][1,3]oxazin-2-one compounds as mutant idh1 and idh2 inhibitorsPublication Number: CA-3045303-CPriority Date: 2016-12-16Grant Date: 2022-05-17
- 7-phenylethylamino-4H-pyrimido [4,5-D ] [1,3] oxazin-2-one compounds as inhibitors of mutant IDH1 and IDH2Publication Number: CN-110072867-BPriority Date: 2016-12-16Grant Date: 2022-07-08
- Mutant IDH1 and IDH2 inhibitorsPublication Number: CN-115109075-APriority Date: 2016-12-16
- Pyrido[4,3-d][1,3]oxazin-2-one compounds as mutant idh1 and idh2 inhibitorsPublication Number: EP-3763717-B1Priority Date: 2016-12-16Grant Date: 2023-03-08
- 7-phenylethylamino-4H-pyrimido[4,5-d][1,3]oxazin-2-one compounds as mutant IDH1 and IDH2 inhibitorsPublication Number: US-11629156-B2Priority Date: 2016-12-16Grant Date: 2023-04-18
- 7-phenylethylamino-4H-pyrimido[4,5-d][1,3]oxazin-2-one compounds as mutant IDH1 and IDH2 inhibitorsPublication Number: US-11649247-B2Priority Date: 2016-12-16Grant Date: 2023-05-16



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | LY3410738 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2230263-60-0 |
| PubChem CID | 135125140 |
| IUPHAR/BPS | 12340 |
| ChemSpider | 115009279 |
| UNII | A4DU555RMD |
| KEGG | D12708 |
| Chemical and physical data | |
| Formula | C28H36N6O3 |
| Molar mass | 504.635 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Zarei M, Hue JJ, Hajihassani O, Graor HJ, Katayama ES, Loftus AW, et al. (February 2022). “Clinical development of IDH1 inhibitors for cancer therapy”. Cancer Treatment Reviews. 103 102334. doi:10.1016/j.ctrv.2021.102334. PMID 34974243.
- Demir T, Moloney C, Mahalingam D (July 2024). “Emerging targeted therapies and strategies to overcome resistance in biliary tract cancers”. Critical Reviews in Oncology/Hematology. 199 104388. doi:10.1016/j.critrevonc.2024.104388. PMID 38754771.
.///////////Crelosidenib, Antineoplastic, cholangiocarcinoma, LY3410738, LY 3410738














