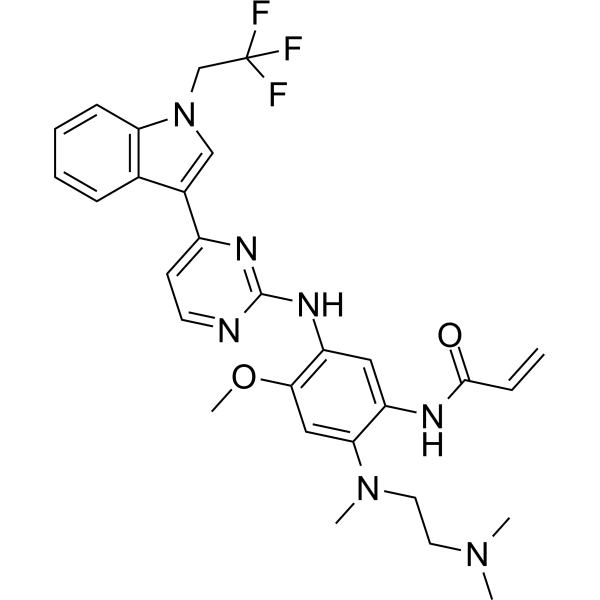
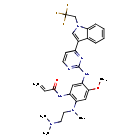
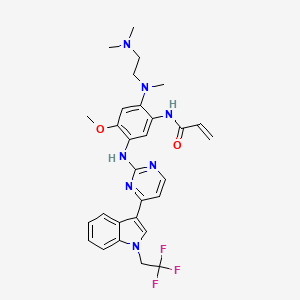
Befotertinib
D-0316, 0XT2CPR891
CAS No. : 1835667-63-4, MESYLATE CAS No. 2226167-02-6
- 2-propenamide, n-(2-((2-(dimethylamino)ethyl)methylamino)-4-methoxy-5-((4-(1-(2,2,2-trifluoroethyl)-1h-indol-3-yl)-2-pyrimidinyl)amino)phenyl)-
- N-(2-((2-(dimethylamino)ethyl)(methyl)amino)-4-methoxy-5-((4-(1-(2,2,2-trifluoroethyl)-1h-indol-3-yl)pyrimidin-2-yl)amino)phenyl)prop-2-enamide
- N-[2-[2-(dimethylamino)ethyl-methylamino]-4-methoxy-5-[[4-[1-(2,2,2-trifluoroethyl)indol-3-yl]pyrimidin-2-yl]amino]phenyl]prop-2-enamide
| Molecular Weight | 567.61 |
|---|---|
| Formula | C29H32F3N7O2 |
Befotertinib (D-0316) is an orally active EGFR tyrosine kinase inhibitor. Befotertinib can inhibit the proliferation of tumor cells. Befotertinib can be used in the research of EGFR T790M-positive non-small cell lung cancer (NSCLC).
Befotertinib is an orally available inhibitor of the epidermal growth factor receptor (EGFR) mutant form T790M, with potential antineoplastic activity. Upon administration, befotertinib specifically binds to and inhibits EGFR T790M, a secondarily acquired resistance mutation, which prevents EGFR-mediated signaling and leads to cell death in EGFR T790M-expressing tumor cells. Compared to some other EGFR inhibitors, befotertinib may have therapeutic benefits in tumors with T790M-mediated drug resistance. EGFR, a receptor tyrosine kinase that is mutated in many tumor cell types, plays a key role in tumor cell proliferation and tumor vascularization.
PAPER
J. Med. Chem. 2017, 60, 6480−6515.
PATENT
WO 2019218987
https://patentscope.wipo.int/search/en/WO2019218987
[0054]
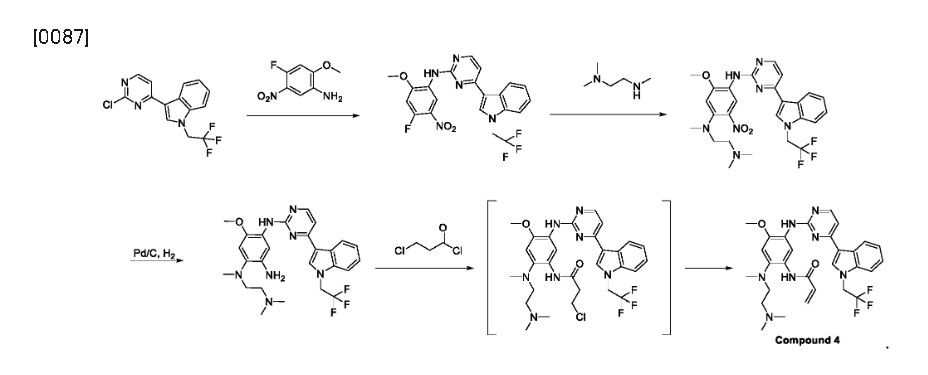
U.S. Publication No. 2017/0355696 A1 describes a method of preparing Compound 4 and various pharmaceutically acceptable salts thereof. The exemplified synthetic process in U. S. Publication No. 2017/0355696 A1 includes a two-step conversion from the aniline compound, corresponding to Compound 1 of this disclosure, into the bismesylate of Compound 4, which has a low yield.
[0055]
As shown herein, representative methods of preparation of Compound 4, or a pharmaceutically acceptable salt, (or alternatively referred to as synthetic methods) , can provide the desired Compound 4, or a pharmaceutically acceptable salt, in improved yield and high purity and can be adapted for large-scale manufacture.
[0056]
In various embodiments, the present invention provides a novel method of preparing Compound 4, or a pharmaceutically acceptable salt thereof. The method typically includes converting a compound of Formula III, or a salt thereof, into compound 4, typically under an elimination reaction condition:
Syn
https://doi.org/10.1021/acs.jmedchem.4c02079
J. Med. Chem. 2025, 68, 2147−2182
Befotertinib (Surmana). Befotertinib (17), an oral, highly selective, third generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) developed by Betta Pharmaceuticals and InventisBio, was approved in China in May 2023 for the second-line treatment of patients
with locally advanced or metastatic nonsmall cell lung cancer (NSCLC) with positive EGFR T790 M mutation who have disease progression on previous EGFR TKI therapy. 140 139 NSCLC
has a high incidence and disease burden in China, which has spurred the development of multiple EGFR TKIs by Chinese companies.
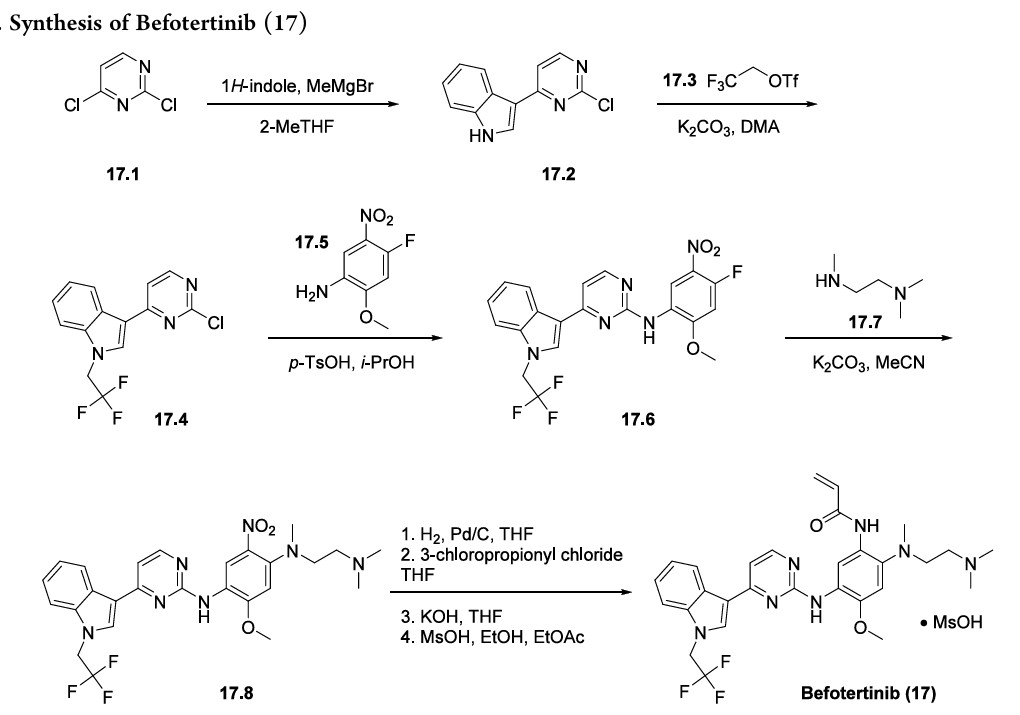
Achromatography-free process route to befotertinib (17) has been reported in the patent literature by researchers at InventisBio (Scheme 29), although details about scale and yields were not provided.
141 142 The reaction sequence closely follows that of osimertinib, a third generation EGFR inhibitor
that was first approved in 2015 and was covered in our previous review.
Osimertinib and befotertinib share a common backbone, differing only in N-substitution on the indole ring.
Friedel−Crafts arylation of 1H-indole with 2,4-dichloropyrimidine (17.1) gave the 3-pyrimidinyl indole 17.2. The trifluoroethyl moiety in indole 17.4 was introduced via Nalkylation of 17.2 with triflate 17.3. This was followed by an SAr reaction with nitroaniline 17.5 to provide amino pyrimidine 17.6. Next, N,N,N′-trimethylethylenediamine (17.7) displaced the electrophilic aryl fluoride in an SNArreaction to generate intermediate 17.8. The acrylamide moiety was installed using a three-step sequence: hydrogenolytic
reduction of the nitro group to the corresponding aniline, acylation with 3-chloropropanoyl chloride, and immediate elimination to the acrylamide. Mesylate salt formation and crystallization furnished befotertinib mesylate (17) in eight steps from 17.1.
(139) Blair, H. A. Befotertinib: first approval. Drugs 2023, 83, 1433−
1437.
(140) Lau, S. C. M.; Ou, S.-H. I. And still they come over troubled
waters: can Asia’s third-generation EGFR tyrosine kinase inhibitors
(Furmonertinib, Aumolertinib, Rezivertinib, Limertinib, Befotertinib,
SH-1028, and Lazertinib) affect global treatment of EGFR+ NSCLC. J.
Thorac. Oncol. 2022, 17, 1144−1154.
(141) Dai, X.; Jiang, Y. Preparation of pyrimidine derivative and its
pharmaceutical salt as EGFR inhibitors for the treatment of cancer and
other diseases. WO 2019218987, 2019.
(142) Flick, A. C.; Ding, H. X.; Leverett, C. A.; Kyne, R. E.; Liu, K. K.
C.; Fink, S. J.; O’Donnell, C. J. Synthetic approaches to the new drugs
approved during 2015. J. Med. Chem. 2017, 60, 6480−6515.



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
- [1]. Nagasaka M, et, al. Beyond Osimertinib: The Development of Third-Generation EGFR Tyrosine Kinase Inhibitors For Advanced EGFR+ NSCLC. J Thorac Oncol. 2021 May;16(5):740-763. [Content Brief][2]. Blair HA. Befotertinib: First Approval. Drugs. 2023 Oct;83(15):1433-1437. [Content Brief]
/////////Befotertinib, APPROVALS 2023, CHINA 2023, Betta Pharmaceuticals, InventisBio, CANCER, D-0316, D 0316, 0XT2CPR891














