BMT-145027
CAS ?
C23H14ClF3N4
Exact Mass: 438.0859
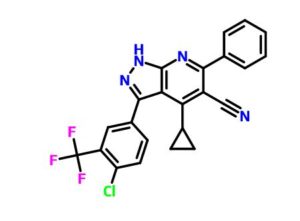
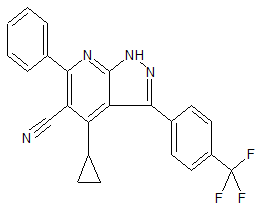
3-(4-chloro-3-(trifluoromethyl)phenyl)-4-cyclopropyl-6-phenyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile
3-(4-chloro-3- (trifluoromethyl)phenyl)-4-cyclopropyl-6-phenyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile
1H NMR (600 MHz, DMSO-d6) δ = 14.46 (br. s., 1H), 8.24 (s, 1H), 8.14 (d, J=8.1 Hz, 1H), 7.88 (d, J=8.3 Hz, 1H), 7.84 (dd, J=6.1, 2.7 Hz, 2H), 7.61 – 7.55 (m, 3H), 2.50 – 2.45 (m, 1H), 0.74 – 0.68 (m, 2H), 0.65 – 0.59 (m, 2H).
13C NMR (126 MHz, DMSO-d6) δ 160.5, 155.0, 153.0, 144.1, 138.3, 135.4, 133.9, 132.0, 131.2, 130.3, 129.7, 128.9, 128.9, 128.8, 127.0 (q, J=30.5 Hz), 118.1, 112.4, 103.9, 14.6, 9.4.
LCMS (method A) tR, 2.01 min, MS Anal. Calcd. for [M+H]+ C23H15ClF3N4: 439.09; found: 439.15.
LC/MS HPLC methods: method A: Column: Phenomenex Luna 30 x 2.0 mm 3um; Mobile Phase A: 10:90 acetonitrile:water with 0.1% TFA; Mobile Phase B: 90:10 acetonitrile:water with 0.1% TFA; Temperature: 40 °C; Gradient: 0-100% B over 2 min; Flow: 1 mL/min.


DETAILS WILL BE UPDATED…………
Senior Research Investigator II at Bristol-Myers Squibb
Highly effective leader seeking to apply innovative thinking and critical analysis to strategy and scientific challenges. Diverse educational background, including recent MBA studies, provides foundation for excellent communication, collaboration, and team building across organizational functions. Experience includes 13 years of cutting-edge scientific research in a global work environment, specializing in the fields of organic chemistry and drug discovery.
Experience
Senior Research Investigator II
Bristol-Myers Squibb
– Present (2 years 4 months)Wallingford, CT
Oncology Discovery Chemistry, Program: Bromodomain and Extra-Terminal Inhibitor Program, undisclosed target
BMT-145027 is a potent mGluR5 PAM with no inherent mGluR5 agonist activity. BMT-145027 is a non-MPEP site PAM to demonstrate in vivo efficacy. BMT-145027 has mGluR5 PAM EC50 = 47 nM, with fold shit = 3.5, and is effective in mouse NOR. The metabotropic glutamate receptor 5 (mGluR5) is an attractive target for the treatment of schizophrenia due to its role in regulating glutamatergic signaling in association with the N-methyl-D-aspartate receptor (NMDAR).
PAPER

The metabotropic glutamate receptor 5 (mGluR5) is an attractive target for the treatment of schizophrenia due to its role in regulating glutamatergic signaling in association with the N-methyl-d-aspartate receptor (NMDAR). We describe the synthesis of 1H-pyrazolo[3,4-b]pyridines and their utility as mGluR5 positive allosteric modulators (PAMs) without inherent agonist activity. A facile and convergent synthetic route provided access to a structurally diverse set of analogues that contain neither the aryl-acetylene-aryl nor aryl-methyleneoxy-aryl elements, the predominant structural motifs described in the literature. Binding studies suggest that members of our new chemotype do not engage the receptor at the MPEP and CPPHA mGluR5 allosteric sites. SAR studies culminated in the first non-MPEP site PAM, 1H-pyrazolo[3,4-b]pyridine 31 (BMT-145027), to improve cognition in a preclinical rodent model of learning and memory.
Development of 1H-Pyrazolo[3,4-b]pyridines as Metabotropic Glutamate Receptor 5 Positive Allosteric Modulators
Matthew D. Hill*, Haiquan Fang, Jeffrey M. Brown, Thaddeus Molski, Amy Easton, Xiaojun Han, Regina Miller, Melissa Hill-Drzewi, Lizbeth Gallagher, Michele Matchett, Michael Gulianello, Anand Balakrishnan, Robert L. Bertekap, Kenneth S. Santone, Valerie J. Whiterock, Xiaoliang Zhuo, Joanne J. Bronson, John E. Macor, and Andrew P. Degnan
Research and Development, Bristol-Myers Squibb, 5 Research Parkway, Wallingford, Connecticut 06492-7660, United States
ACS Med. Chem. Lett., Article ASAP
DOI: 10.1021/acsmedchemlett.6b00292, http://pubs.acs.org/doi/abs/10.1021/acsmedchemlett.6b00292
*Tel: 1-203-677-7102. Fax: 1-203-677-7884. E-mail: matthew.hill@bms.com.

SIMILAR STR
Synthesis (2016), 48, (14), 2201-2204.
A Multicomponent Approach to Highly Substituted 1H-Pyrazolo[3,4-b]pyridines
Compound 12 (500 mg, 65% yield: 1H NMR (500 MHz, DMSO-d6 δ 14.41 (br. s., 1H, 7.86 – 7.80 (m, 3H, 7.76 (dt, J=7.1, 1.6 Hz, 1H, 7.61 – 7.51 (m, 5H, 2.48 – 2.45 (m, 1H, 0.73 – 0.66 (m, 2H, 0.62 – 0.57 (m, 2H. 13C NMR (400 MHz, DMSO-d6 δ 159.98, 154.67, 152.15, 144.68, 137.75, 135.74, 132.69, 129.87, 129.71, 129.14, 128.99, 128.34, 128.24, 128.21, 117.61, 111.88, 103.05, 14.01, 8.75. IR (film: 3228 (s, 3052 (w, 2228 (m, 1581 (s, 1555 (s, 1503 (w, 1447 (m, 1284 (m cm–1. HRMS (ESI: m/z [M+H]+ calcd for C22H16N4Cl: 371.1058; found: 371.1053.
Compound 13 (103 mg, 28% yield: 1H NMR (500 MHz, DMSO-d6 δ 14.50 (br. s., 1H, 8.03 (d, J=7.9 Hz, 2H, 7.92 – 7.80 (m, 4H, 7.63 – 7.55 (m, 3H, 2.51 (br. s., 1H, 0.65 (d, J=7.6 Hz, 2H, 0.56 (d, J=4.3 Hz, 2H. MS (ESI: m/z = 405.15 [M+H]+.
///////////BMT-145027, glutamat, mGluR5, novel object recognition, positive allosteric modulator, schizophrenia
c1(c(c(c2c(n1)nnc2c3ccc(c(c3)C(F)(F)F)Cl)C4CC4)C#N)c5ccccc5
ClC(C=C1)=C(C(F)(F)F)C=C1C2=NNC3=C2C(C4CC4)=C(C#N)C(C5=CC=CC=C5)=N3