Fluvoxamine
- Molecular FormulaC15H21F3N2O2
- Average mass318.335 Da
- 54739-18-3
Fluvoxamine, sold under the brand name Luvox among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class[5] which is used primarily for the treatment of obsessive–compulsive disorder (OCD).[6] It is also used to treat depression and anxiety disorders, such as panic disorder, social anxiety disorder, and post-traumatic stress disorder.[7][8]
FLUVOXAMINE MALEATE
C19H25F3N2O6, 434.4 g/mol
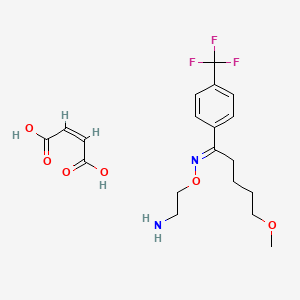
1-Pentanone, 5-methoxy-1-(4-(trifluoromethyl)phenyl)-, O-(2-aminoethyl)oxime, (E)-, (Z)-2-butenedioate (1:1)
(Z)-but-2-enedioic acid;2-[(E)-[5-methoxy-1-[4-(trifluoromethyl)phenyl]pentylidene]amino]oxyethanamine
Luvox, 61718-82-9
CAS 54739-20-7, Fevarin, Luvox CR
Synonyms
-
5-Methoxy-4′-(trifluoromethyl)valerophenone (E)-O-(2-aminoethyl)oxime, maleate (1:1)
-
5-Methoxy-4′-trifluoromethylvalerophenone (E)-O-2-aminoethyloxime monomaleate
-
DU23000
-
-
Fevarin
-
Fluvoxamine maleate
-
Luvox
-
Luvox CR
-
SME 3110
-
UNII-5LGN83G74V
-
| Originator Company | Solvay SA |
| Active Companies | AbbVie Inc; Abbott Laboratories; Meiji Seika Pharma Co Ltd; Solvay SA |
| Launched (Obsessive compulsive disorder – EU – Dec-1983) |
In the EU, the product is indicated for the treatment of obsessive compulsive disorder (OCD) and for the treatment of major depressive disorder (MDD)
In Japan, Luvox is indicated for the treatment of adult or pediatric OCD, social anxiety disorder (SAD) and MDD
USFDA The drug was approved for the treatment of OCD and SAD in April 2008
CHINA
In 2000, the drug was launched in China for the treatment of OCD and MDD
Patents and Generics
FDA exclusivity expired in the US in June 2000. Generic versions have been on the market since that time. Generic fluvoxamine was still available in the US by May 2007, despite the fact the Solvay/Jazz product had not been relaunched . By October 2004, the drug was also off patent in most European countries .
Medical uses
Fluvoxamine is approved in the United States for OCD,[9][6] and social anxiety disorder.[10] In other countries (e.g., Australia,[11][12] the UK,[13] and Russia[14]) it also has indications for major depressive disorder. In Japan it is currently approved to treat OCD, SAD and MDD.[15][16] Fluvoxamine is indicated for children and adolescents with OCD.[17] The drug works long-term, and retains its therapeutic efficacy for at least one year.[18] It has also been found to possess some analgesic properties in line with other SSRIs and tricyclic antidepressants.[19][20][21]
There is tentative evidence that fluvoxamine is effective for social phobia in adults.[22] Fluvoxamine is also effective for GAD, SAD, panic disorder and separation anxiety disorder in children and adolescents.[23] There is tentative evidence that fluvoxamine may help some people with negative symptoms of chronic schizophrenia.[24][25]
A double-blind controlled study found that fluvoxamine may prevent clinical deterioration in outpatients with symptomatic COVID-19. The study had important limitations: it was run fully remotely; it had a small sample size (150) and short follow-up duration (15 days).[26] The accompanying editorial noted that, although this study is important enough to choose out of more than 10,000 other COVID-19 related submissions, it “presents only preliminary information” and “the findings should be interpreted as only hypothesis generating; they should not be used as the basis for current treatment decisions.”[27] Similarly, the study authors themselves cautioned that “the trial’s results should not be treated as a measure of fluvoxamine’s effectiveness against COVID-19 but as an encouraging indicator that the drug warrants further testing.”[28] A prospective open-labelled cohort study showed similar results.[29]
Adverse effects
Gastrointestinal side effects are more common in those receiving fluvoxamine than with other SSRIs.[30] Otherwise, fluvoxamine’s side-effect profile is very similar to other SSRIs.[2][9][11][13][31][32]
- Common (1–10% incidence) adverse effects
- Nausea
- Vomiting
- Weight loss
- Yawning
- Loss of appetite
- Agitation
- Nervousness
- Anxiety
- Insomnia
- Somnolence (drowsiness)
- Tremor
- Restlessness
- Headache
- Dizziness
- Palpitations
- Tachycardia (high heart rate)
- Abdominal pain
- Dyspepsia (indigestion)
- Diarrhea
- Constipation
- Hyperhidrosis (excess sweating)
- Asthenia (weakness)
- Malaise
- Sexual dysfunction (including delayed ejaculation, erectile dysfunction, decreased libido, etc.)
- Xerostomia (dry mouth)
- Uncommon (0.1–1% incidence) adverse effects
- Arthralgia
- Hallucination
- Confusional state
- Extrapyramidal side effects (e.g. dystonia, parkinsonism, tremor, etc.)
- Orthostatic hypotension
- Cutaneous hypersensitivity reactions (e.g. oedema [buildup of fluid in the tissues], rash, pruritus)
- Rare (0.01–0.1% incidence) adverse effects
- Mania
- Seizures
- Abnormal hepatic (liver) function
- Photosensitivity (being abnormally sensitive to light)
- Galactorrhoea (expulsion of breast milk unrelated to pregnancy or breastfeeding)
- Unknown frequency adverse effects
- Hyperprolactinaemia (elevated plasma prolactin levels leading to galactorrhoea, amenorrhoea [cessation of menstrual cycles], etc.)
- Bone fractures
- Glaucoma
- Mydriasis
- Urinary incontinence
- Urinary retention
- Bed-wetting
- Serotonin syndrome — a potentially fatal condition characterised by abrupt onset muscle rigidity, hyperthermia (elevated body temperature), rhabdomyolysis, mental status changes (e.g. coma, hallucinations, agitation), etc.
- Neuroleptic malignant syndrome — practically identical presentation to serotonin syndrome except with a more prolonged onset
- Akathisia — a sense of inner restlessness that presents itself with the inability to stay still
- Paraesthesia
- Dysgeusia
- Haemorrhage
- Withdrawal symptoms
- Weight changes
- Suicidal ideation and behaviour
- Violence towards others[33]
- Hyponatraemia
- Syndrome of inappropriate antidiuretic hormone secretion
- Ecchymoses
Interactions[edit]
Fluvoxamine inhibits the following cytochrome P450 enzymes:[34][35][36][37][38][39][40][41][42]
- CYP1A2 (strongly) which metabolizes agomelatine, amitriptyline, caffeine, clomipramine, clozapine, duloxetine, haloperidol, imipramine, phenacetin, tacrine, tamoxifen, theophylline, olanzapine, etc.
- CYP3A4 (moderately) which metabolizes alprazolam, aripiprazole, clozapine, haloperidol, quetiapine, pimozide, ziprasidone, etc.[43]
- CYP2D6 (weakly) which metabolizes aripiprazole, chlorpromazine, clozapine, codeine, fluoxetine, haloperidol, olanzapine, oxycodone, paroxetine, perphenazine, pethidine, risperidone, sertraline, thioridazine, zuclopenthixol, etc.[44]
- CYP2C9 (moderately) which metabolizes nonsteroidal anti-inflammatory drugs, phenytoin, sulfonylureas, etc.
- CYP2C19 (strongly) which metabolizes clonazepam, diazepam, phenytoin, etc.
- CYP2B6 (weakly) which metabolizes bupropion, cyclophosphamide, sertraline, tamoxifen, valproate, etc.
By so doing, fluvoxamine can increase serum concentration of the substrates of these enzymes.[34]
The plasma levels of oxidatively metabolized benzodiazepines (e.g., triazolam, midazolam, alprazolam and diazepam) are likely to be increased when co-administered with fluvoxamine. However the clearance of benzodiazepines metabolized by glucuronidation (e.g., lorazepam, oxazepam, temazepam)[45][46] is unlikely to be affected by fluvoxamine.[47] It appears that benzodiazepines metabolized by nitro-reduction (clonazepam, nitrazepam) are unlikely to be affected by fluvoxamine.[48] Using fluvoxamine and alprazolam together can increase alprazolam plasma concentrations.[49] If alprazolam is coadministered with fluvoxamine, the initial alprazolam dose should be reduced to the lowest effective dose.[50][51]
Fluvoxamine and ramelteon coadministration is not indicated.[52][53]
Fluvoxamine has been observed to increase serum concentrations of mirtazapine, which is mainly metabolized by CYP1A2, CYP2D6, and CYP3A4, by 3- to 4-fold in humans.[54] Caution and adjustment of dosage as necessary are warranted when combining fluvoxamine and mirtazapine.[54]
Fluvoxamine seriously affects the pharmacokinetics of tizanidine and increases the intensity and duration of its effects. Because of the potentially hazardous consequences, the concomitant use of tizanidine with fluvoxamine, or other potent inhibitors of CYP1A2, should be avoided.[55]
Fluvoxamine’s interaction with St John’s wort can lead to increased serotonin levels and potentially lead to serotonin syndrome.[citation needed]
Pharmacology
| Site | Ki (nM) |
|---|---|
| SERT | 2.5 |
| NET | 1,427 |
| 5-HT2C | 5,786 |
| α1-adrenergic | 1,288 |
| σ1 | 36 |
Fluvoxamine is a potent selective serotonin reuptake inhibitor with around 100-fold affinity for the serotonin transporter over the norepinephrine transporter.[35] It has negligible affinity for the dopamine transporter or any other site, with the sole exception of the σ1 receptor.[59][60] It behaves as a potent agonist at this receptor and has the highest affinity (36 nM) of any SSRI for doing so.[59] This may contribute to its antidepressant and anxiolytic effects and may also afford it some efficacy in treating the cognitive symptoms of depression.[61] Unlike fluoxetine, fluvoxamine’s metabolites are inactive, without a significant effect on serotonin or norepinephrine uptake.[62]
History
Fluvoxamine was developed by Kali-Duphar,[63] part of Solvay Pharmaceuticals, Belgium, now Abbott Laboratories, and introduced as Floxyfral in Switzerland in 1983.[63] It was approved by the U.S. Food and Drug Administration (FDA) in 1994, and introduced as Luvox in the US.[64] In India, it is available, among several other brands, as Uvox by Abbott.[65] It was one of the first SSRI antidepressants to be launched, and is prescribed in many countries to patients with major depression.[66] It was the first SSRI, a non-TCA drug, approved by the U.S. FDA specifically for the treatment of OCD.[67] At the end of 1995, more than ten million patients worldwide had been treated with fluvoxamine.[68][failed verification] Fluvoxamine was the first SSRI to be registered for the treatment of obsessive compulsive disorder in children by the FDA in 1997.[69] In Japan, fluvoxamine was the first SSRI to be approved for the treatment of depression in 1999[70][71] and was later in 2005 the first drug to be approved for the treatment of social anxiety disorder.[72] Fluvoxamine was the first SSRI approved for clinical use in the United Kingdom.[73]
Society and culture
Manufacturers include BayPharma, Synthon, and Teva, among others.[74]
SYN


SYN
J. Zhejiang Univ. (Medical Sci.) (2003), 32 (5), 441-442
PATENT
WO 2014178064
The present invention relates to an improved and industrially applicable process for the preparation of fluvoxamine maleate of formula I,
Fluvoxamine or (E)-5-methoxy-1 -[4-(trifluoromethyl)phenyl]pentan- 1 -one-O-2-aminoethyl oxime is an antidepressant which functions as a selective serotonin reuptake inhibitor (SSRI). Fluvoxamine is used for the treatment of major depressive disorder (MDD), obsessive compulsive disorder (OCD), and anxiety disorders such as panic disorder and post-traumatic stress disorder (PTSD). Fluvoxamine CR (controlled release) is approved to treat social anxiety disorder.
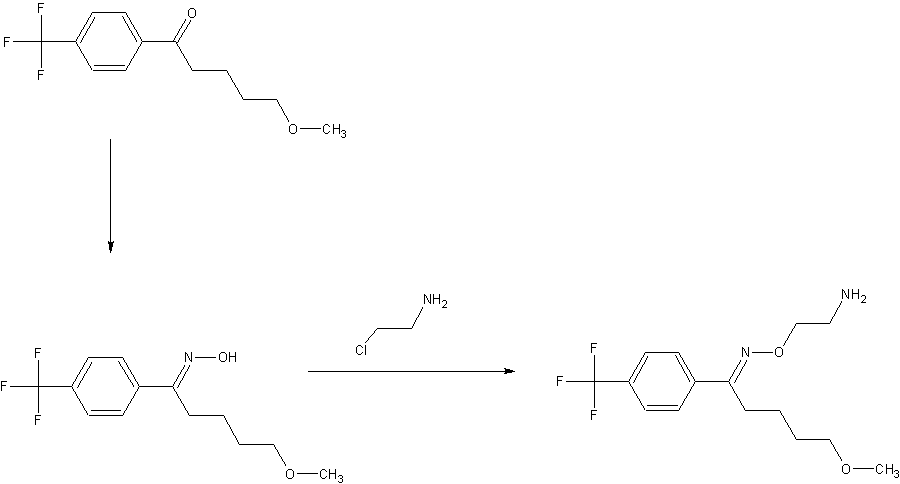
Fluvoxamine maleate and compounds were first disclosed in US patent 4,085,225. According to said patent, Fluvoxamine maleate prepared by alkylation reaction of 5-methoxy-4′-trifluoromethylvalerophenone oxime, compound of formula III with 2-chloroethylamine hydrochloride in dimethylformamide in the presence of a base such as potassium hydroxide powder for two days at 25°C.
Subsequently the solvent is removed under vacuum then the residue is acidified and extracted with ether to remove the unreacted oxime followed by basification. The obtained fluvoxamine base in ether extract is washed with sodium bicarbonate solution. The fluvoxamine base is then treated with maleic acid in absolute ethanbl and the residue obtained by concentration under vacuum is recrystallized from acetonitrile to obtain fluvoxamine maleate. The process is very much tedious, time consuming as it requires two days for the reaction completion. Operations like removal of dimethylformamide, ether, ethanol makes process cumbersome at plant level. Requirement of
various solvents lead the process to be non-eco-friendly. Moreover the patent is silent about yield and purity of the product.
In an alternate route described in US patent 4,085,225, the oxime of formula III is converted to formula I in a five step process i.e. alkylation of formula III with ethylene oxide. The reaction solvent is ethanol in which lithium is already dissolved. The reaction further involves addition of acetic acid to give the hydroxyethyl compound of formula A as oil. The compound of formula A is purified chromatographically over the silica gel, which is converted to a mesylate compound of formula B by treating with methanesulfonyl chloride and triethylamine at -5 to 0°C, then aminated with ammonia in methanol at 100°C using autoclave for 16 hours followed by removal of methanol and extraction in ether to give fluvoxamine base.
The base is then converted to the maleate salt formula I, which is finally purified by recrystallization from acetonitrile.
There are lots of disadvantages involve like more unit operations, use of various solvents and handling of ethylene oxide which is also known for its carcinogen effect. More unit operations lead to long occupancy of reactors in the plant as well as man power, high energy consumption and require bigger plant. These all parameters make the process commercially unviable as wel l as environmentally non-feasible. Further, purification of the compound of formula A requires cumbersome technique i.e chromatography over silica gel as well as lengthy work-up procedure in U.S. Pat. No. 4,085,225 requires complete removal of organic solvents at various stages.
US patent 6,433,225 discloses the process for preparing fluvoxamine maleate, prepared by alkylating 5-methoxy-4′-trtfluoromethylvalerophenone oxime, compound of formula III with 2-chloroethylamine hydrochloride in toluene and PEG-400 (polyethyleneglycol-400) as facilitator in the presence of a base potassium hydroxide powder at 30-35°C to obtain fluvoxamine base in
toluene layer is then treated with maleic acid in water. The precipitated fluvoxamine maleate is filtered and washed with toluene and dried. The obtained dried cake recrystallized with water to get fluvoxamine maleate. The process disclosed in the patent is silent about actual purity of the product. As per our scientist’s observation alkylation reaction at the temperature of 30-35°C may lead to non completion of reaction and results lower yield. Additional step of purification may further lead to loss of yield.
Thus, present invention fulfills the need of the art and provides an improved and industrially applicable process for preparation of fluvoxamine maleate, which provides fluvoxamine maleate in high purity and overall good yield.
EXAMPLES:
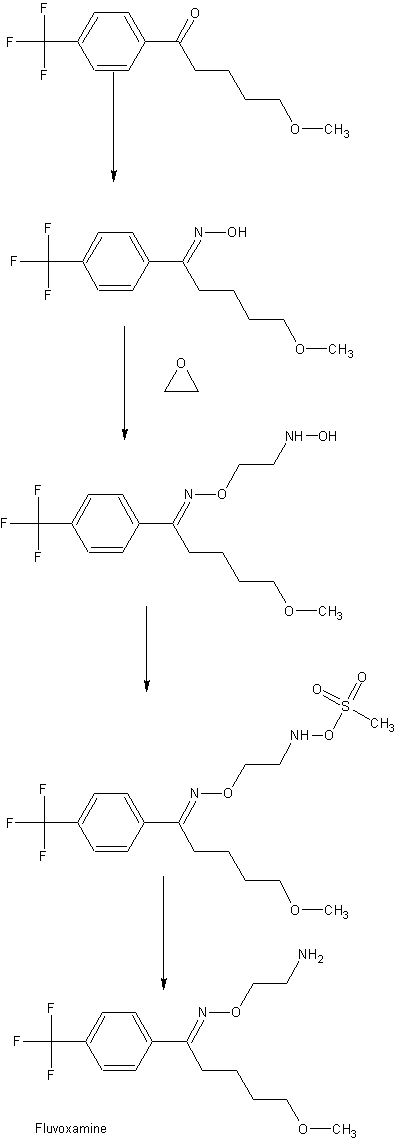
Stage – 1 : Preparation of (1E)-N-hydroxy-5-methoxy-1-(4-trifluoromethyI pheny 1) pentan-1-imine formula III
To a stirred solution of 5-methoxy- 1 -(4-trifluoromethylphenyl) pentan-1 -one ( 150 gm) in methanol (750 ml), sodium carbonate (granule) (72 gm) and hydroxylamine hydrochloride (59.64 gm) were added at temperature 25-30°C. The reaction mass was heated 45-50°C for 10- 15 minutes followed by maintaining the reaction mass at temperature 45-50°C for 8-9 hours under stirring. The reaction mass was cooled to 25-30°C and filtered under vacuum to remove unreacted inorganic matter, then distilled out the methanol completely from the collected filtrate under vacuum at temperature below 50°C. The obtained slurry was cooled to 25-30°C and water (300 ml) was added into the residue followed by the addition of hexane (300×2 ml) and stirred for 30 minutes. The layers were separated. The collected organic layer was stirred for 5- 10 minutes at temperature 25-30°C followed by cooling the mass at temperature -5°C to – 10°C, stirred for 30-40 minutes and filtered at the same temperature. The product was suck dried at -5 to -10°C and further in vacuum at 25-30°C for 2-3 hours to give 138 – 142 gm of title compound. HPLC purity: >98.5%
Stage – 2: Preparation of crude fluvoxamine maleate formula I
To a prepared solution of dimethyl sulphoxide (575 ml), potassium hydroxide flakes ( 1 14.64 gm) and water (69 ml), stage-1 (1 15 gm) was added at temperature 40-45°C. The reaction mixture was stirred to get clear solution followed by adding 2-chloroethylamine hydrochloride (86.36 gm) drop wise into the reaction mixture at temperature 40-45°C and maintained for 1 -2 hour. Water (1 150 ml) was added in to the reaction mixture at temperature 25-30°C and stirred for 20-25 minutes. Then toluene (575 ml x 2) was added and stirred for 30 minutes and preceded for separation of layers followed by washing the toluene layer with water ( 1 1 50 x 5 ml). The solution of maleic acid (48.47 gm) dissolved in water (98 ml) was added into above obtained toluene layer and stirred at temperature 25-30°C for 2-3 hours. The reaction mixture was cooled to 0-5°C and maintained for 30-40 minutes at the same temperature. The obtained material was washed with toluene, filtered and suck dried. The wet cake was then added hexane (600 ml) and stirred for 30 minutes at temperature 25-30°C, filtered, washed with hexane and dried to get 161 gm of title compound. HPLC purity: >98.5%
Stage – 3: Preparation of pure fluvoxamine maleate formula I
In to the reaction assembly, water (600 ml) was added and heated to 40-45°C. Stage -2 ( 1 50 gm) was added into the hot water under stirring. The reaction mixture was stirred for 5- 10 minutes, filtered and cooled to 25°C. Toluene (68 ml) was added into the reaction mixture at temperature 25°C and stirred for 30 minutes. Filtered the solid, washed with 10-15°C chilled water and dried to get the pure 127.5 gm fluvoxamine maleate. HPLC purity: >99.8%
Process for isolation of 5-methoxy-1-[4-(trifluoromethyl)phenyl]pentan-1-one formula II
To a solution of cone. HCl (600 ml) and water ( 160 ml), organic residue (250 gm) of ( 1 £)+( 1 Z) of 1 -N-hydroxy-5-methoxy- 1 -[4-(trifluoromethyl) phenyl]pentan-1 -imine and traces of 5-methoxy- 1 -[4-(trifluoromethyl)phenyl]pentan- 1-one (obtained after hexane recovery from stage-1 filtrate) was added at temperature 25-30°C under stirring. The reaction mixture was heated to 67-75°C and maintained for 13-14 hours followed by cool ing the reaction mixture at temperature 25-30°C. Then after hexane (500 x 2 ml) was added into the reaction mixture and stirred for 15 minutes at 25-30°C. The organic layers were separated and sodium bicarbonate solution (25 gm sodium bicarbonate dissolved in 250 ml water) was added into the hexane layer and stirred for 15 minutes. The layers were separated and water (250ml) was added into hexane layer and stirred for 15 minutes at temperature 25-30°C. Further the layers were separated and hexane layer was added activated charcoal ( 12.5 gm) and stirred for 20-30 minutes at temperature 30-35°C. The reaction mixture was filtered and stirred for 5-10 minutes at 25-30°C followed by cooling at 0 to -5°C and stirred for 30-40 minutes at 0 to -5°C. The reaction mixture was filtered and dried to get 150 to l 75 gm of title compound. HPLC purity: >99%.
PATENT
US 20140243544
IN 2013MU01290/WO 2014178064
WO 2014035107
PATENT
https://patents.google.com/patent/US9783492B2/en
Fluvoxamine or (E)-5-methoxy-1-[4-(trifluoromethyl)phenyl]pentan-1-one-O-2-aminoethyl oxime is an antidepressant which functions as a selective serotonin reuptake inhibitor (SSRI). Fluvoxamine is used for the treatment of major depressive disorder (MDD), obsessive compulsive disorder (OCD), and anxiety disorders such as panic disorder and post-traumatic stress disorder (PTSD). Fluvoxamine CR (controlled release) is approved to treat social anxiety disorder.
Fluvoxamine maleate and compounds were first disclosed in U.S. Pat. No. 4,085,225. According to said patent, Fluvoxamine maleate prepared by alkylation reaction of 5-methoxy-4′-trifluoromethylvalerophenone oxime, compound of formula III with 2-chloroethylamine hydrochloride in dimethylformamide in the presence of a base such as potassium hydroxide powder for two days at 25° C.
Subsequently the solvent is removed under vacuum then the residue is acidified and extracted with ether to remove the unreacted oxime followed by basification. The obtained fluvoxamine base in ether extract is washed with sodium bicarbonate solution. The fluvoxamine base is then treated with maleic acid in absolute ethanol and the residue obtained by concentration under vacuum is recrystallized from acetonitrile to obtain fluvoxamine maleate. The process is very much tedious, time consuming as it requires two days for the reaction completion. Operations like removal of dimethylformamide, ether, ethanol makes process cumbersome at plant level. Requirement of various solvents lead the process to be non-eco-friendly. Moreover the patent is silent about yield and purity of the product.
In an alternate route described in U.S. Pat. No. 4,085,225, the mine of formula III is converted to formula I in a five step process i.e. alkylation of formula III with ethylene oxide. The reaction solvent is ethanol in which lithium is already dissolved. The reaction further involves addition of acetic acid to give the hydroxyethyl compound of formula A as oil. The compound of formula A is purified chromatographically over the silica gel, which is converted to a mesylate compound of formula B by treating with methanesulfonyl chloride and triethylamine at −5 to 0° C., then aminated with ammonia in methanol at 100° C. using autoclave for 16 hours followed by removal of methanol and extraction in ether to give fluvoxamine base.
The base is then converted to the maleate salt formula I, which is finally purified by recrystallization from acetonitrile.
There are lots of disadvantages in like more unit operations, use of various solvents and handling of ethylene oxide which is also known for its carcinogen effect. More unit operations lead to long occupancy of reactors in the plant as well as man power, high energy consumption and require bigger plant. These all parameters make the process commercially unviable as well as environmentally non-feasible. Further, purification of the compound of formula A requires cumbersome technique i.e chromatography over silica gel as well as lengthy work-up procedure in U.S. Pat. No. 4,085,225 requires complete removal of organic solvents at various stages.
U.S. Pat. No. 6,433,225 discloses the process for preparing fluvoxamine maleate, prepared by alkylating 5-methoxy-4′-trifluoromethylvalerophenone oxime compound of formula III with 2-chloroethylamine hydrochloride in toluene and PEG-400 (polyethyleneglycol-400) as facilitator in the presence of a base potassium hydroxide powder at 30-35°C. to obtain fluvoxamine base in toluene layer is then treated with maleic acid in water. The precipitated fluvoxamine maleate is filtered and washed with toluene and dried. The obtained dried cake recrystallized with water to get fluvoxamine maleate. The process disclosed in the patent is silent about actual purity of the product. As per our scientist’s observation alkylation reaction at the temperature of 30-35° C. may lead to non completion of reaction and results lower yield. Additional step of purification may further lead to loss of yield.
EXAMPLES
Stage-1: Preparation of (1 E)-N-hydroxy-5-methoxy-1-(4-trifluoromethyl phenyl)pentan-1-imine Formula III
To a stirred solution of 5-methoxy-1-(4-trifluoromethylphenyl)pentan-1one (150 gm) in methanol (750 ml), sodium carbonate (granule) (72 gm) and hydroxylamine hydrochloride (59.64 gm) were added at temperature 25-30° C. The reaction mass was heated 45-50° C. for 10-15 minutes followed by maintaining the reaction mass at temperature 45-50° C. for 8-9 hours under stirring. The reaction mass was cooled to 25-30° C. and filtered under vacuum to remove unreacted inorganic matter, then distilled out the methanol completely from the collected filtrate under vacuum at temperature below 50° C. The obtained slurry was cooled to 25-30° C. and water (300 ml) was added into the residue followed by the addition of hexane (300×2 ml) and stirred for 30 minutes. The layers were separated. The collected organic layer was stirred for 5-10 minutes at temperature 25-30° C. followed by cooling the mass at temperature −5° C. to −10° C., stirred for 30-40 minutes and filtered at the same temperature. The product was suck dried at −5 to −10° C. and further in vacuum at 25-30° C. for 2-3 hours to give 138-142 gm of title compound. HPLC purity: >98.5%
Stage-2: Preparation of Crude Fluvoxamine Maleate Formula I
To a prepared solution of dimethyl sulphoxide (575 ml), potassium hydroxide flakes (114.64 gm) and water (69 ml), stage-1 (115 gm) was added at temperature 40-45° C. The reaction mixture was stirred to get clear solution followed by adding 2-chloroethylamine hydrochloride (8636 gm) drop wise into the reaction mixture at temperature 40-45° C. and maintained for 1-2 hour. Water (1150 ml) was added in to the reaction mixture at temperature 25-30° C. and stirred for 20-25 minutes. Then toluene (575 ml×2) was added and stirred for 30 minutes and preceded for separation of layers followed by washing the toluene layer with water (1150×5 ml). The solution of maleic acid (48.47 gm) dissolved in water (98 ml) was added into above obtained toluene layer and stirred at temperature 25-30° C. for 2-3 hours. The reaction mixture was cooled to 0-5° C. and maintained for 30-40 minutes at the same temperature. The obtained material was washed with toluene, filtered and such dried. The wet cake was then added hexane (600 ml) and stirred for 30 minutes at temperature 25-30° C., filtered, washed with hexane and dried to get 161 gm of title compound. HPLC purity: >98.5%
Stage-3: Preparation of Pure Fluvoxamine Maleate Formula I
In to the reaction assembly, water (600 ml) was added and heated to 40-45° C. Stage-2 (150 gm) was added into the hot water under stirring. The reaction mixture was stirred for 5-10 minutes, filtered and cooled to 25° C. Toluene (68 ml) was added into the reaction mixture at temperature 25° C. and stirred for 30 minutes. Filtered the solid, washed with 10-15° C. chilled water and dried to get the pure 127.5 gm fluvoxamine maleate. HPLC purity: >99.8%
Process for isolation of 5-methoxy-1-[4-(trifluoromethyl)phenyl]pentan-1-one Formula II
To a solution of conc. HCl (600 ml) and water (160 organic residue (250 gm) of (1 E)+(1 Z) of 1-N-hydroxy-5-methoxy-1-[4trifluoromethyl)phenyl]pentan-1-imine and traces of 5-methoxy-1-[4-(trifluoromethyl)phenyl]pentan-1-one (obtained after hexane recovery from stage-1 filtrate) was added at temperature 25-30° C. under stirring. The reaction mixture was heated to 67-75° C. and maintained for 13-14 hours followed by cooling the reaction mixture at temperature 25-30° C. Then after hexane (500×2 ml) was added into the reaction mixture and stirred for 15 minutes at 25-30° C. The organic layers were separated and sodium bicarbonate solution (25 gm sodium bicarbonate dissolved in 250 ml water) was added into the hexane layer and stirred for 15 minutes. The layers were separated and water (250 ml) was added into hexane layer and stirred for 15 minutes at temperature 25-30° C. Further the layers were separated and hexane layer was added activated charcoal (12.5 gm) and stirred for 20-30 minutes at temperature 30-35° C. The reaction mixture was filtered and stirred for 5-10 minutes at 25-30° C. followed by cooling at 0 to −5° C. and stirred for 30-40 minutes at 0 to −5° C. The reaction mixture was filtered and dried to get 150 to 175 gm of title compound. HPLC purity: >99%.
We claim:
2. The process according to claim 1, wherein in step a), said suitable solvent is selected from the group consisting of alcohol, ketone, nitrile, and hydrocarbons in any suitable proportion or mixtures thereof;
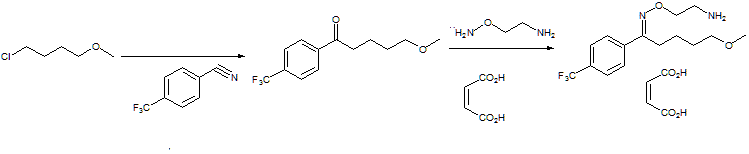
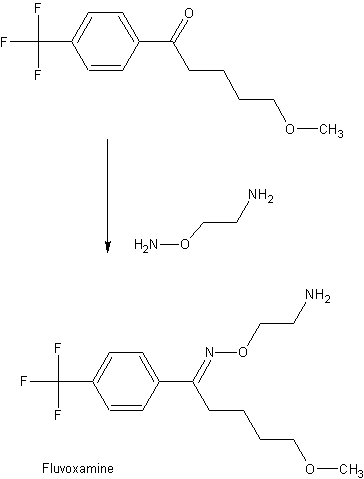
EXAMPLE 15-Methoxy-4′-trifluoromethylvalerophenone O-(2-aminoethyl) oxime maleate (1:1).
20.4 Mmol (5.3 g) of 5-methoxy-4′-trifluoromethylvalerophenone (melting point 43°-44° C), 20.5 mmol (3.1 g) of 2-aminooxyethylaminedihydrochloride and 10 ml of pyridine were refluxed for 15 hours in 20 ml of absolute ethanol. After evaporating the pyridine and the ethanol in vacuo, the residue was dissolved in water. This solution was washed with petroleum ether and 10 ml of 50% sodium hydroxide solution were then added. Then three extractions with 40 ml of ether were carried out. The ether extract was washed successively with 20 ml of 5% sodium bicarbonate solution and 20 ml of water. After drying on sodium sulphate, the ether layer was evaporated in vacuo. Toluene was then evaporated another three times (to remove the pyridine) and the oil thus obtained was dissolved in 15 ml of absolute ethanol. An equimolar quantity of maleic acid was added to said solution and the solution was then heated until a clear solution was obtained. The ethanol was then removed in vacuo and the residue was crystallized from 10 ml of acetonitrile at +5° C. After sucking off and washing with cold acetonitrile, it was dried in air. The melting point of the resulting title compound was 120°-121.5° C.
References
- ^ Jump up to:a b Use During Pregnancy and Breastfeeding
- ^ Jump up to:a b c d e f “Product Information Luvox”. TGA eBusiness Services. Abbott Australasia Pty Ltd. 15 January 2013. Retrieved 21 October 2013.
- ^ van Harten J (March 1993). “Clinical pharmacokinetics of selective serotonin reuptake inhibitors”. Clinical Pharmacokinetics. 24 (3): 203–20. doi:10.2165/00003088-199324030-00003. PMID 8384945. S2CID 84636672.
- ^ “Luvox”. ChemSpider. Royal Society of Chemistry. Archived from the original on 15 November 2013. Retrieved 21 October 2013.
- ^ “Fluvoxamine Maleate Information”. U.S. Food and Drug Administration(FDA). 15 July 2015. Archived from the original on 29 November 2019. Retrieved 28 November 2019.
- ^ Jump up to:a b McCain JA (July 2009). “Antidepressants and suicide in adolescents and adults: a public health experiment with unintended consequences?”. P T. 34(7): 355–78. PMC 2799109. PMID 20140100.
- ^ Figgitt DP, McClellan KJ (October 2000). “Fluvoxamine. An updated review of its use in the management of adults with anxiety disorders”. Drugs. 60 (4): 925–54. doi:10.2165/00003495-200060040-00006. PMID 11085201.
- ^ Irons J (December 2005). “Fluvoxamine in the treatment of anxiety disorders”. Neuropsychiatric Disease and Treatment. 1 (4): 289–99. PMC 2424117. PMID 18568110.
- ^ Jump up to:a b “Fluvoxamine Maleate tablet, coated prescribing information”. DailyMed. 14 December 2018. Retrieved 28 November 2019.
- ^ “Luvox CR approved for OCD and SAD”. MPR. 29 February 2008. Retrieved 2 March 2019.
- ^ Jump up to:a b Rossi S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ “Luvox Tablets”. NPS MedicineWise. Retrieved 22 October 2018.
- ^ Jump up to:a b Joint Formulary Committee (2013). British National Formulary (BNF)(65 ed.). London, UK: Pharmaceutical Press. ISBN 978-0-85711-084-8.
- ^ “Summary of Full Prescribing Information: Fluvoxamine”. Drug Registry of Russia (RLS) Drug Compendium (in Russian). Retrieved 21 March 2015.
- ^ “2005 News Releases”. Astellas Pharma. Retrieved 16 September 2018.
- ^ “International Approvals: Ebixa, Depromel/Luvox, M-Vax”. www.medscape.com. Retrieved 16 September 2018.
- ^ “US-FDA Fluvoxamine Product Insert”. March 2005.
- ^ Wilde MI, Plosker GL, Benfield P (November 1993). “Fluvoxamine. An updated review of its pharmacology, and therapeutic use in depressive illness”. Drugs. 46(5): 895–924. doi:10.2165/00003495-199346050-00008. PMID 7507038.
- ^ Kwasucki J, Stepień A, Maksymiuk G, Olbrych-Karpińska B (2002). “[Evaluation of analgesic action of fluvoxamine compared with efficacy of imipramine and tramadol for treatment of sciatica–open trial]”. Wiadomosci Lekarskie. 55 (1–2): 42–50. PMID 12043315.
- ^ Schreiber S, Pick CG (August 2006). “From selective to highly selective SSRIs: a comparison of the antinociceptive properties of fluoxetine, fluvoxamine, citalopram and escitalopram”. European Neuropsychopharmacology. 16 (6): 464–8. doi:10.1016/j.euroneuro.2005.11.013. PMID 16413173. S2CID 39278756.
- ^ Coquoz D, Porchet HC, Dayer P (September 1993). “Central analgesic effects of desipramine, fluvoxamine, and moclobemide after single oral dosing: a study in healthy volunteers”. Clinical Pharmacology and Therapeutics. 54 (3): 339–44. doi:10.1038/clpt.1993.156. PMID 8375130. S2CID 8229797.
- ^ Williams T, Hattingh CJ, Kariuki CM, Tromp SA, van Balkom AJ, Ipser JC, Stein DJ (October 2017). “Pharmacotherapy for social anxiety disorder (SAnD)”. The Cochrane Database of Systematic Reviews. 10 (10): CD001206. doi:10.1002/14651858.CD001206.pub3. PMC 6360927. PMID 29048739.
- ^ Cheer SM, Figgitt DP (2002). “Spotlight on fluvoxamine in anxiety disorders in children and adolescents”. CNS Drugs. 16 (2): 139–44. doi:10.2165/00023210-200216020-00006. PMID 11825104. S2CID 26774895.
- ^ Silver H (2001). “Fluvoxamine as an adjunctive agent in schizophrenia”. CNS Drug Reviews. 7 (3): 283–304. doi:10.1111/j.1527-3458.2001.tb00200.x. PMC 6741705. PMID 11607044.
- ^ Polcwiartek C, Nielsen J (March 2016). “The clinical potentials of adjunctive fluvoxamine to clozapine treatment: a systematic review”. Psychopharmacology. 233 (5): 741–50. doi:10.1007/s00213-015-4161-1. PMID 26626327. S2CID 12168939.
- ^ Lenze EJ, Mattar C, Zorumski CF, Stevens A, Schweiger J, Nicol GE, Miller JP, Yang L, Yingling M, Avidan MS, Reiersen AM (December 2020). “Fluvoxamine vs Placebo and Clinical Deterioration in Outpatients With Symptomatic COVID-19: A Randomized Clinical Trial”. JAMA. 324 (22): 2292–2300. doi:10.1001/jama.2020.22760. PMID 33180097.
- ^ Seymour CW, Bauchner H, Golub RM (December 2020). “COVID-19 Infection-Preventing Clinical Deterioration”. JAMA. 324 (22): 2300. doi:10.1001/jama.2020.21720. PMID 33180115.
- ^ [+https://scitechdaily.com/antidepressant-fluvoxamine-may-prevent-covid-19-infections-from-worsening/ “Antidepressant Fluvoxamine May Prevent COVID-19 Infections From Worsening”] Check
|url=value (help). - ^ https://academic.oup.com/ofid/advance-article/doi/10.1093/ofid/ofab050/6124100
- ^ Brayfield A, ed. (13 August 2013). Fluoxetine Hydrochloride. Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 24 November 2013.
- ^ Taylor D, Paton C, Shitij K (2012). The Maudsley prescribing guidelines in psychiatry. West Sussex: Wiley-Blackwell. ISBN 978-0-470-97948-8.
- ^ “Faverin 100 mg film-coated tablets – Summary of Product Characteristics (SPC)”. electronic Medicines Compendium. Abbott Healthcare Products Limited. 14 May 2013. Retrieved 21 October 2013.
- ^ “Top Ten Legal Drugs Linked to Violence”. Time. 7 January 2011. Retrieved 10 September 2014.
- ^ Jump up to:a b Ciraulo DA, Shader RI (2011). Ciraulo DA, Shader RI (eds.). Pharmacotherapy of Depression (2nd ed.). Springer. p. 49. doi:10.1007/978-1-60327-435-7. ISBN 978-1-60327-435-7.
- ^ Jump up to:a b Brunton L, Chabner B, Knollman B (2010). Goodman and Gilman’s The Pharmacological Basis of Therapeutics (12th ed.). New York: McGraw-Hill Professional. ISBN 978-0-07-162442-8.
- ^ Baumann P (December 1996). “Pharmacokinetic-pharmacodynamic relationship of the selective serotonin reuptake inhibitors”. Clinical Pharmacokinetics. 31 (6): 444–69. doi:10.2165/00003088-199631060-00004. PMID 8968657. S2CID 31923953.
- ^ DeVane CL, Gill HS (1997). “Clinical pharmacokinetics of fluvoxamine: applications to dosage regimen design”. The Journal of Clinical Psychiatry. 58 Suppl 5 (Suppl 5): 7–14. PMID 9184622.
- ^ DeVane CL (1998). “Translational pharmacokinetics: current issues with newer antidepressants”. Depression and Anxiety. 8 Suppl 1 (Suppl 1): 64–70. doi:10.1002/(SICI)1520-6394(1998)8:1+<64::AID-DA10>3.0.CO;2-S. PMID 9809216.
- ^ Bondy B, Spellmann I (March 2007). “Pharmacogenetics of antipsychotics: useful for the clinician?”. Current Opinion in Psychiatry. 20 (2): 126–30. doi:10.1097/YCO.0b013e328017f69f. PMID 17278909. S2CID 23859992.
- ^ Kroon LA (September 2007). “Drug interactions with smoking”. American Journal of Health-System Pharmacy. 64 (18): 1917–21. doi:10.2146/ajhp060414. PMID 17823102.
- ^ Waknine Y (13 April 2007). “Prescribers Warned of Tizanidine Drug Interactions”. Medscape News. Medscape. Retrieved 1 February 2008.
- ^ “Fluvoxamine (Oral Route) Precautions”. Mayo Clinic. Retrieved 2 November2018.
- ^ Hemeryck A, Belpaire FM (February 2002). “Selective serotonin reuptake inhibitors and cytochrome P-450 mediated drug-drug interactions: an update”. Current Drug Metabolism. 3 (1): 13–37. doi:10.2174/1389200023338017. PMID 11876575.
- ^ “Drug Development and Drug Interactions: Table of Substrates, Inhibitors and Inducers”.
- ^ Raouf M (2016). Fudin J (ed.). “Benzodiazepine Metabolism and Pharmacokinetics” (PDF).
- ^ Peppers MP (1996). “Benzodiazepines for alcohol withdrawal in the elderly and in patients with liver disease”. Pharmacotherapy. 16 (1): 49–57. doi:10.1002/j.1875-9114.1996.tb02915.x. PMID 8700792. S2CID 1389910.
- ^ “fluvoxamine maleate: PRODUCT MONOGRAPH” (PDF). 2016.
- ^ “Luvox Data Sheet” (PDF). Medsafe, New Zealand. 2017.
- ^ Suzuki Y, Shioiri T, Muratake T, Kawashima Y, Sato S, Hagiwara M, Inoue Y, Shimoda K, Someya T (April 2003). “Effects of concomitant fluvoxamine on the metabolism of alprazolam in Japanese psychiatric patients: interaction with CYP2C19 mutated alleles”. European Journal of Clinical Pharmacology. 58 (12): 829–33. doi:10.1007/s00228-003-0563-9. PMID 12698310. S2CID 32559753.
- ^ Gerlach M, Warnke A, Greenhill L (2014). Psychiatric Drugs in Children and Adolescents: Basic Pharmacology and Practical Applications. Springer-Verlag Wien. p. 131. ISBN 978-3-7091-1500-8.
- ^ Fleishaker JC, Hulst LK (1994). “A pharmacokinetic and pharmacodynamic evaluation of the combined administration of alprazolam and fluvoxamine”. European Journal of Clinical Pharmacology. 46 (1): 35–9. doi:10.1007/bf00195913. PMID 8005185. S2CID 2161450.
- ^ Obach RS, Ryder TF (August 2010). “Metabolism of ramelteon in human liver microsomes and correlation with the effect of fluvoxamine on ramelteon pharmacokinetics”. Drug Metabolism and Disposition. 38 (8): 1381–91. doi:10.1124/dmd.110.034009. PMID 20478852. S2CID 8421997.
- ^ Pandi-Perumal SR, Spence DW, Verster JC, Srinivasan V, Brown GM, Cardinali DP, Hardeland R (12 April 2011). “Pharmacotherapy of insomnia with ramelteon: safety, efficacy and clinical applications”. Journal of Central Nervous System Disease. 3: 51–65. doi:10.4137/JCNSD.S1611. PMC 3663615. PMID 23861638.
- ^ Jump up to:a b Anttila AK, Rasanen L, Leinonen EV (October 2001). “Fluvoxamine augmentation increases serum mirtazapine concentrations three- to fourfold”. The Annals of Pharmacotherapy. 35 (10): 1221–3. doi:10.1345/aph.1A014. PMID 11675851. S2CID 44807359.
- ^ Granfors MT, Backman JT, Neuvonen M, Ahonen J, Neuvonen PJ (April 2004). “Fluvoxamine drastically increases concentrations and effects of tizanidine: a potentially hazardous interaction”. Clinical Pharmacology and Therapeutics. 75(4): 331–41. doi:10.1016/j.clpt.2003.12.005. PMID 15060511. S2CID 25781307.
- ^ Ishikawa M, Ishiwata K, Ishii K, Kimura Y, Sakata M, Naganawa M, et al. (October 2007). “High occupancy of sigma-1 receptors in the human brain after single oral administration of fluvoxamine: a positron emission tomography study using [11C]SA4503”. Biological Psychiatry. 62 (8): 878–83. doi:10.1016/j.biopsych.2007.04.001. PMID 17662961. S2CID 728565.
- ^ Schatzberg AF, Nemeroff CB (2009). The American Psychiatric Publishing textbook of psychopharmacology (4th ed.). Arlington, VA: American Psychiatric Pub. p. 354. ISBN 978-1-585-62386-0. OCLC 320111564.
- ^ Yahata M, Chiba K, Watanabe T, Sugiyama Y (September 2017). “Possibility of Predicting Serotonin Transporter Occupancy From the In Vitro Inhibition Constant for Serotonin Transporter, the Clinically Relevant Plasma Concentration of Unbound Drugs, and Their Profiles for Substrates of Transporters”. Journal of Pharmaceutical Sciences. 106 (9): 2345–2356. doi:10.1016/j.xphs.2017.05.007. PMID 28501470.
- ^ Jump up to:a b Hashimoto K (September 2009). “Sigma-1 receptors and selective serotonin reuptake inhibitors: clinical implications of their relationship”. Central Nervous System Agents in Medicinal Chemistry. 9 (3): 197–204. doi:10.2174/1871524910909030197. PMID 20021354.
- ^ Westenberg HG, Sandner C (April 2006). “Tolerability and safety of fluvoxamine and other antidepressants”. International Journal of Clinical Practice. 60 (4): 482–91. doi:10.1111/j.1368-5031.2006.00865.x. PMC 1448696. PMID 16620364.
- ^ Hindmarch I, Hashimoto K (April 2010). “Cognition and depression: the effects of fluvoxamine, a sigma-1 receptor agonist, reconsidered”. Human Psychopharmacology. 25 (3): 193–200. doi:10.1002/hup.1106. PMID 20373470. S2CID 26491662.
- ^ Hrdina PD (July 1991). “Pharmacology of serotonin uptake inhibitors: focus on fluvoxamine”. Journal of Psychiatry & Neuroscience. 16 (2 Suppl 1): 10–8. PMC 1188307. PMID 1931931.
- ^ Jump up to:a b Sittig’s Pharmaceutical Manufacturing Encyclopedia (PDF) (3rd ed.). William Andrew. 2008. p. 1699. ISBN 978-0-8155-1526-5. Retrieved 17 October2013.
- ^ Leslie LK, Newman TB, Chesney PJ, Perrin JM (July 2005). “The Food and Drug Administration’s deliberations on antidepressant use in pediatric patients”. Pediatrics. 116 (1): 195–204. doi:10.1542/peds.2005-0074. PMC 1550709. PMID 15995053.
- ^ “Brand Index―Fluvoxamine India”. Archived from the original on 19 October 2013. Retrieved 18 October 2013.
- ^ Omori IM, Watanabe N, Nakagawa A, Cipriani A, Barbui C, McGuire H, Churchill R, Furukawa TA (March 2010). “Fluvoxamine versus other anti-depressive agents for depression”. The Cochrane Database of Systematic Reviews (3): CD006114. doi:10.1002/14651858.CD006114.pub2. PMC 4171125. PMID 20238342.
- ^ “OCD Medication”. Archived from the original on 14 October 2013. Retrieved 17 October 2013.
- ^ “Fluvoxamine Product Monograph” (PDF). 1999.
- ^ “Luvox Approved For Obsessive Compulsive Disorder in Children and Teens”. Archived from the original on 16 January 2009. Retrieved 8 February 2014.
- ^ Higuchi T, Briley M (February 2007). “Japanese experience with milnacipran, the first serotonin and norepinephrine reuptake inhibitor in Japan”. Neuropsychiatric Disease and Treatment. 3 (1): 41–58. doi:10.2147/nedt.2007.3.1.41. PMC 2654524. PMID 19300537.
- ^ “Human Metabolome Database: Showing metabocard for Fluvoxamine (HMDB0014322)”. www.hmdb.ca. Retrieved 15 September 2018.
- ^ “Solvay’s Fluvoxamine maleate is first drug approved for the treatment of social anxiety disorder in Japan”.
- ^ Walker R, Whittlesea C, eds. (2007) [1994]. Clinical Pharmacy and Therapeutics (4th ed.). Edinburgh: Churchill Livingstone Elsevier. ISBN 978-0-7020-4293-5.
- ^ “Fluvoxamine”. www.drugbank.ca. Retrieved 22 October 2019.
External links
- “Fluvoxamine”. Drug Information Portal. U.S. National Library of Medicine.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Luvox, Faverin, Fluvoxin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695004 |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
By mouth |
| Drug class | Selective serotonin reuptake inhibitor (SSRI) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 53% (90% confidence interval: 44–62%)[2] |
| Protein binding | 77-80%[2][3] |
| Metabolism | Hepatic (via cytochrome P450 enzymes. Mostly via oxidative demethylation)[2] |
| Elimination half-life | 12–13 hours (single dose), 22 hours (repeated dosing)[2] |
| Excretion | Renal (98%; 94% as metabolites, 4% as unchanged drug)[2] |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.125.476 |
| Chemical and physical data | |
| Formula | C15H21F3N2O2 |
| Molar mass | 318.335 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
/////////DU23000, Fevarin, Fluvoxamine maleate, Luvox, Luvox CR, SME 3110, UNII-5LGN83G74V, Fluvoxamine, sme 3110, DU 23000
#DU23000, #Fevarin, #Fluvoxamine maleate, #Luvox, #Luvox CR, #SME 3110, #UNII-5LGN83G74V, #Fluvoxamine, #sme 3110, #DU 23000