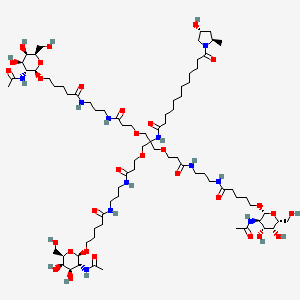
Givosiran
N-[1,3-bis[3-[3-[5-[(2R,3R,4R,5R,6R)-3-acetamido-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxypentanoylamino]propylamino]-3-oxopropoxy]-2-[[3-[3-[5-[(2R,3R,4R,5R,6R)-3-acetamido-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxypentanoylamino]propylamino]-3-oxopropoxy]methyl]propan-2-yl]-12-[(2R,4R)-4-hydroxy-2-methylpyrrolidin-1-yl]-12-oxododecanamide
Treatment of Acute Hepatic Porphyria (AHP)
| Formula |
C524H694F16N173O316P43S6
|
|---|---|
| CAS |
1639325-43-1
|
| Mol weight |
16300.3229
|
Treatment of acute hepatic porphyria, RNA interference (RNAi) drug
FDA APPROVED, Givlaari, 2019/11/20
ギボシラン;
Givosiran, sold under the brand name Givlaari, is for the treatment of adults with acute hepatic porphyria, a genetic disorder resulting in the buildup of toxic porphyrin molecules which are formed during the production of heme (which helps bind oxygen in the blood).[1][2]
History
The U.S. Food and Drug Administration (FDA) granted the application for givosiran breakthrough therapy designation, priority reviewdesignation, and orphan drug designation.[1] The FDA granted the approval of Givlaari to Alnylam Pharmaceuticals.[1]
The full ENVISION results demonstrated a 74 percent mean and 90 percent median reduction in the primary endpoint measure of annualized rate of composite attacks in patients on givosiran relative to placebo during the six-month double-blind period. In addition, givosiran achieved statistically significant positive results for five of nine secondary endpoints, with an overall safety and tolerability profile that the Company believes is encouraging, especially in this high unmet need disease. Adverse events (AEs) were reported in 89.6 percent of givosiran patients and 80.4 percent of placebo patients; serious adverse events (SAEs) were reported in 20.8 percent of givosiran patients and 8.7 percent of placebo patients. Ninety-three of 94 patients, or 99 percent, enrolled in the open-label extension (OLE) period of the study. Based on the ENVISION results, the Company plans to complete its rolling submission of a New Drug Application (NDA) and file a Marketing Authorisation Application (MAA) in mid-2019.
“Given the high unmet need in this disease setting, we are very pleased for the patients and families living with acute hepatic porphyria for whom these results signal hope for a potential new therapeutic option,” said Akshay Vaishnaw, M.D., Ph.D., President of R&D at Alnylam. “Givosiran substantially reduced the frequency of attacks, providing strong support for a treatment benefit, with a consistent effect across all components of the primary endpoint and all subgroups analyzed. In this disease with high burden and associated comorbidities, we’re encouraged by the overall tolerability profile. We firmly believe givosiran has the potential to be a transformative medicine for patients living with AHP.”
“Currently, there are no approved therapies aimed at preventing the painful, often incapacitating attacks and chronic symptoms associated with AHP,” said Manisha Balwani, M.D., M.S, Associate Professor of the Department of Genetics and Genomic Sciences and Department of Medicine at the Icahn School of Medicine at Mount Sinai and principal investigator of the ENVISION study. “The results from ENVISION are promising and demonstrate a strong treatment effect for givosiran, with reduction of attacks and improvement in patient-reported measures of overall health status and quality of life. Thus, givosiran represents a novel and targeted treatment approach that has the potential to make a significant impact on the lives of patients who are struggling with the disabling symptoms of this disease.”
Efficacy Results
Givosiran met the primary efficacy endpoint with a 74 percent mean reduction relative to placebo in the annualized rate of composite porphyria attacks, defined as those requiring hospitalization, urgent healthcare visit, or hemin administration, in patients with acute intermittent porphyria (AIP) over six months (p equal to 6.04×10-9). There was a corresponding 90 percent median reduction in composite annualized attack rate (AAR), with a median AAR of 1.0 in givosiran patients compared with a median AAR of 10.7 in placebo patients. Fifty percent of givosiran-treated patients were attack-free during the six-month treatment period as compared to 16.3 percent of placebo-treated patients. The reductions in attack rates were observed across all components of the primary endpoint. The treatment benefit for givosiran compared to placebo was maintained across all pre-specified patient subgroups, including age, race, geography, historical attack rates, prior hemin prophylaxis status, disease severity, and other baseline characteristics.
Givosiran also demonstrated statistically significant differences in five of nine hierarchically tested secondary endpoints relative to placebo. These included mean reductions of:
- 91 percent in urinary aminolevulinic acid (ALA) in patients with AIP at three months (p equal to 8.74×10-14).
- 83 percent in urinary ALA in patients with AIP at six months (p equal to 6.24×10-7).
- 73 percent in urinary levels of porphobilinogen (PBG) in patients with AIP at six months (p equal to 8.80×10-7).
- 77 percent in the number of annualized days on hemin in patients with AIP (p equal to 2.35×10-5).
- 73 percent in composite AAR for patients with any AHP (p equal to 1.35×10-8).
The remaining four secondary endpoints did not meet the prespecified criteria for statistical significance in hierarchical testing.

About Acute Hepatic Porphyria
Acute hepatic porphyria (AHP) refers to a family of rare, genetic diseases characterized by potentially life-threatening attacks and for some patients chronic debilitating symptoms that negatively impact daily functioning and quality of life. AHP is comprised of four subtypes, each resulting from a genetic defect leading to deficiency in one of the enzymes of the heme biosynthesis pathway in the liver: acute intermittent porphyria (AIP), hereditary coproporphyria (HCP), variegate porphyria (VP), and ALAD-deficiency porphyria (ADP). These defects cause the accumulation of neurotoxic heme intermediates aminolevulinic acid (ALA) and porphobilinogen (PBG), with ALA believed to be the primary neurotoxic intermediate responsible for causing both attacks and ongoing symptoms between attacks. Common symptoms of AHP include severe, diffuse abdominal pain, weakness, nausea, and fatigue. The nonspecific nature of AHP signs and symptoms can often lead to misdiagnoses of other more common conditions such as irritable bowel syndrome, appendicitis, fibromyalgia, and endometriosis, and consequently, patients afflicted by AHP often remain without a proper diagnosis for up to 15 years. In addition, long-term complications of AHP and its treatment can include chronic neuropathic pain, hypertension, chronic kidney disease and liver disease, including iron overload, fibrosis, cirrhosis and hepatocellular carcinoma. Currently, there are no treatments approved to prevent debilitating attacks or to treat the chronic manifestations of the disease.
About Givosiran
Givosiran is an investigational, subcutaneously administered RNAi therapeutic targeting aminolevulinic acid synthase 1 (ALAS1) in development for the treatment of acute hepatic porphyria (AHP). Monthly administration of givosiran has the potential to significantly lower induced liver ALAS1 levels in a sustained manner and thereby decrease neurotoxic heme intermediates, aminolevulinic acid (ALA) and porphobilinogen (PBG), to near normal levels. By reducing accumulation of these intermediates, givosiran has the potential to prevent or reduce the occurrence of severe and life-threatening attacks, control chronic symptoms, and decrease the burden of the disease. Givosiran utilizes Alnylam’s Enhanced Stabilization Chemistry ESC-GalNAc conjugate technology, which enables subcutaneous dosing with increased potency and durability and a wide therapeutic index. Givosiran has been granted Breakthrough Therapy Designation by the U.S. Food and Drug Administration (FDA) and PRIME Designation by the European Medicines Agency (EMA). Givosiran has also been granted Orphan Drug Designations in both the U.S. and the EU for the treatment of AHP. The safety and efficacy of givosiran were evaluated in the ENVISION Phase 3 trial with positive results; these results have not been evaluated by the FDA, the EMA or any other health authority.
About RNAi
RNAi (RNA interference) is a natural cellular process of gene silencing that represents one of the most promising and rapidly advancing frontiers in biology and drug development today. Its discovery has been heralded as “a major scientific breakthrough that happens once every decade or so,” and was recognized with the award of the 2006 Nobel Prize for Physiology or Medicine. By harnessing the natural biological process of RNAi occurring in our cells, a new class of medicines, known as RNAi therapeutics, is now a reality. Small interfering RNA (siRNA), the molecules that mediate RNAi and comprise Alnylam’s RNAi therapeutic platform, function upstream of today’s medicines by potently silencing messenger RNA (mRNA) – the genetic precursors – that encode for disease-causing proteins, thus preventing them from being made. This is a revolutionary approach with the potential to transform the care of patients with genetic and other diseases.
References
- ^ Jump up to:a b c “FDA approves first treatment for inherited rare disease”. U.S. Food and Drug Administration (FDA) (Press release). 20 November 2019. Archived from the original on 21 November 2019. Retrieved 20 November 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “FDA approves givosiran for acute hepatic porphyria”. U.S. Food and Drug Administration (FDA) (Press release). 20 November 2019. Archived from the original on 21 November 2019. Retrieved 20 November 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - The New England journal of medicine (2019), 380(6), 549-558.
- New England Journal of Medicine (2019), 380(6), 549-558.
- Toxicologic Pathology (2018), 46(7), 735-745.
External links
- “Givosiran”. Drug Information Portal. U.S. National Library of Medicine (NLM).
-
GIVLAARI
(givosiran) Injection, for Subcutaneous UseDESCRIPTION
GIVLAARI is an aminolevulinate synthase 1-directed small interfering RNA (siRNA), covalently linked to a ligand containing three N-acetylgalactosamine (GalNAc) residues to enable delivery of the siRNA to hepatocytes.
The structural formulas of the givosiran drug substance in its sodium form, and the ligand (L96), are presented below.

Abbreviations: Af = adenine 2′-F ribonucleoside; Cf = cytosine 2′-F ribonucleoside; Uf = uracil 2′-F ribonucleoside; Am = adenine 2′-OMe ribonucleoside; Cm = cytosine 2′-OMe ribonucleoside; Gf = guanine 2′-F ribonucleoside; Gm = guanine 2′-OMe ribonucleoside; Um = uracil 2′-OMe ribonucleoside; L96 = triantennary GalNAc (N-acetylgalactosamine)
GIVLAARI is supplied as a sterile, preservative-free, 1-mL colorless-to-yellow solution for subcutaneous injection containing 189 mg givosiran in a single-dose, 2-mL Type 1 glass vial with a TEFLON®-coated stopper and a flip-off aluminum seal. GIVLAARI is available in cartons containing one single-dose vial each. Water for injection is the only excipient used in the manufacture of GIVLAARI.
The molecular formula of givosiran sodium is C524 H651 F16 N173 Na43 O316 P43 S6 with a molecular weight of 17,245.56 Da.
The molecular formula of givosiran (free acid) is C524 H694 F16 N173 O316 P43 S6 with a molecular weight of 16,300.34 Da.
| Clinical data | |
|---|---|
| Trade names | Givlaari |
| Routes of administration |
Subcutaneous injection |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C524H694F16N173O316P43S6 |
| Molar mass | 16300.42 g·mol−1 |
| 3D model (JSmol) | |
/////////Givosiran, ギボシラン , FDA 2019, Acute Hepatic Porphyria,