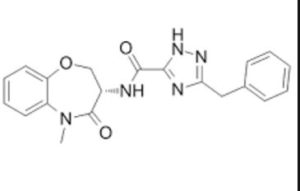
CAS: 1622848-92-3 (free base), 1987858-31-0 (hydrate)
Chemical Formula: C20H19N5O3
Molecular Weight: 377.404
5-Benzyl-N-[(3S)-5-methyl-4-oxo-2,3,4,5-tetrahydro-1,5-benzoxazepin-3-yl]-4H-1,2,4-triazole-3-carboxamide
(S)-5-benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][l,4]oxazepin-3-yl)-4H-l,2,4- triazole-3-carboxamide
- 3-(Phenylmethyl)-N-[(3S)-2,3,4,5-tetrahydro-5-methyl-4-oxo-1,5-benzoxazepin-3-yl]-1H-1,2,4-triazole-5-carboxamide
- (S)-5-Benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-4H-1,2,4-triazole-3-carboxamide
GSK2982772 is a potent and selective receptor Interacting Protein 1 (RIP1) Kinase Specific Clinical Candidate for the Treatment of Inflammatory Diseases. GSK2982772 is, currently in phase 2a clinical studies for psoriasis, rheumatoid arthritis, and ulcerative colitis. GSK2982772 potently binds to RIP1 with exquisite kinase specificity and has excellent activity in blocking many TNF-dependent cellular responses. RIP1 has emerged as an important upstream kinase that has been shown to regulate inflammation through both scaffolding and kinase specific functions.
GSK-2982772, an oral receptor-interacting protein-1 (RIP1) kinase inhibitor, is in phase II clinical development at GlaxoSmithKline for the treatment of active plaque-type psoriasis, moderate to severe rheumatoid arthritis, and active ulcerative colitis. A phase I trial was also completed for the treatment of inflammatory bowel disease using capsule and solution formulations.
- Originator GlaxoSmithKline
- Class Antipsoriatics
- Mechanism of Action Receptor-interacting protein serine-threonine kinase inhibitors
Highest Development Phases
- Phase II Plaque psoriasis; Rheumatoid arthritis; Ulcerative colitis
- Phase I Inflammatory bowel diseases
Most Recent Events
- 15 Dec 2016 Biomarkers information updated
- 01 Nov 2016 Phase-II clinical trials in Ulcerative colitis (Adjunctive treatment) in USA (PO) (NCT02903966)
- 01 Oct 2016 Phase-II clinical trials in Rheumatoid arthritis in Poland (PO) (NCT02858492)
PHASE 2 Psoriasis, plaque GSK
| Inflammatory Bowel Disease, Agents for Rheumatoid Arthritis, Treatment of Antipsoriatics |

Data Science and Informatics Leader | Innovation Advocate
GSK
University of North Carolina at Chapel Hill
He is a data scientist and innovator with experience in both early and late stages of drug development. his current role involves the late stage of drug product development. I’m leading a project to bring GSK’s large molecule process and analytical data onto our big data platform and develop new data analysis and modeling capabilities. Also, working within GSK’s Advanced Manufacturing Technology (AMT) initiative provides plenty of other opportunities to impact how we make medicines.
Previously as a computational chemist (i.e. a data scientist in drug discovery), he worked with scientists from many domains, including chemists, biologists, and other informaticians. he enjoys digging into all the computational aspects of life science research, and solving data challenges by exploiting adjacencies and connections – between diverse fields of knowledge, and the equally diverse scientists trained in them.
He has supported multiple drug discovery projects at GSK starting from target identification (“how should we modulate disease X?”) through to candidate selection and early clinical development (“let’s see if what we discovered can become a medicine”). Deriving insight by custom data integration is one of my specialties; recently he designed and implemented a platform for integrating data sets from multiple experiments that will be used by GSK screening scientists to find and combine hits.
A trained computer scientist and cheminformatician, he is an active member of the algorithms, data science and internal innovation communities at GSK, leading many of these efforts.
His Ph.D. work introduced new computational geometry techniques for structural bioinformatics and protein function prediction. I have touched on several other subject areas:
* data mining/machine learning (predictive modeling and graph mining),
* computer graphics and augmented reality (one of the pioneers of projection mapping)
* robotics (keen current interest and future aspiration)
Receptor-interacting protein- 1 (RIP1) kinase, originally referred to as RIP, is a TKL family serine/threonine protein kinase involved in innate immune signaling. RIPl kinase is a RHIM domain containing protein, with an N-terminal kinase domain and a C-terminal death domain ((2005) Trends Biochem. Sci. 30, 151-159). The death domain of RIPl mediates interaction with other death domain containing proteins including Fas and TNFR-1 ((1995) Cell 81 513-523), TRAIL-Rl and TRAIL-R2 ((1997) Immunity 7, 821-830) and TRADD ((1996) Immunity 4, 387-396), while the RHIM domain is crucial for binding other RHFM domain containing proteins such as TRIF ((2004) Nat Immunol. 5, 503-507), DAI ((2009) EMBO Rep. 10, 916-922) and RIP3 ((1999) J. Biol. Chem. 274, 16871-16875); (1999) Curr. Biol. 9, 539-542) and exerts many of its effects through these interactions. RIPl is a central regulator of cell signaling, and is involved in mediating both pro-survival and programmed cell death pathways which will be discussed below.
The role for RIPl in cell signaling has been assessed under various conditions
[including TLR3 ((2004) Nat Immunol. 5, 503-507), TLR4 ((2005) J. Biol. Chem. 280,
36560-36566), TRAIL ((2012) J .Virol. Epub, ahead of print), FAS ((2004) J. Biol. Chem. 279, 7925-7933)], but is best understood in the context of mediating signals downstream of the death receptor TNFRl ((2003) Cell 114, 181-190). Engagement of the TNFR by TNF leads to its oligomerization, and the recruitment of multiple proteins, including linear K63-linked polyubiquitinated RIPl ((2006) Mol. Cell 22, 245-257), TRAF2/5 ((2010) J. Mol. Biol. 396, 528-539), TRADD ((2008) Nat. Immunol. 9, 1037-1046) and cIAPs ((2008) Proc. Natl. Acad. Sci. USA. 105, 1 1778-11783), to the cytoplasmic tail of the receptor. This complex which is dependent on RIPl as a scaffolding protein (i.e. kinase
independent), termed complex I, provides a platform for pro-survival signaling through the activation of the NFKB and MAP kinases pathways ((2010) Sci. Signal. 115, re4).
Alternatively, binding of TNF to its receptor under conditions promoting the
deubiquitination of RIPl (by proteins such as A20 and CYLD or inhibition of the cIAPs) results in receptor internalization and the formation of complex II or DISC (death-inducing signaling complex) ((2011) Cell Death Dis. 2, e230). Formation of the DISC, which contains RIPl, TRADD, FADD and caspase 8, results in the activation of caspase 8 and the onset of programmed apoptotic cell death also in a RIPl kinase independent fashion ((2012) FEBS J 278, 877-887). Apoptosis is largely a quiescent form of cell death, and is involved in routine processes such as development and cellular homeostasis.
Under conditions where the DISC forms and RJP3 is expressed, but apoptosis is inhibited (such as FADD/caspase 8 deletion, caspase inhibition or viral infection), a third RIPl kinase-dependent possibility exists. RIP3 can now enter this complex, become phosphorylated by RIPl and initiate a caspase-independent programmed necrotic cell death through the activation of MLKL and PGAM5 ((2012) Cell 148, 213-227); ((2012) Cell 148, 228-243); ((2012) Proc. Natl. Acad. Sci. USA. 109, 5322-5327). As opposed to apoptosis, programmed necrosis (not to be confused with passive necrosis which is not programmed) results in the release of danger associated molecular patterns (DAMPs) from the cell.
These DAMPs are capable of providing a “danger signal” to surrounding cells and tissues, eliciting proinflammatory responses including inflammasome activation, cytokine production and cellular recruitment ((2008 Nat. Rev. Immunol 8, 279-289).
Dysregulation of RIPl kinase-mediated programmed cell death has been linked to various inflammatory diseases, as demonstrated by use of the RIP3 knockout mouse (where RIPl -mediated programmed necrosis is completely blocked) and by Necrostatin-1 (a tool inhibitor of RIPl kinase activity with poor oral bioavailability). The RIP3 knockout mouse has been shown to be protective in inflammatory bowel disease (including Ulcerative colitis and Crohn’s disease) ((2011) Nature 477, 330-334), Psoriasis ((2011) Immunity 35, 572-582), retinal-detachment-induced photoreceptor necrosis ((2010) PNAS 107, 21695-21700), retinitis pigmentosa ((2012) Proc. Natl. Acad. Sci., 109:36, 14598-14603), cerulein-induced acute pancreatits ((2009) Cell 137, 1100-1111) and Sepsis/systemic inflammatory response syndrome (SIRS) ((2011) Immunity 35, 908-918). Necrostatin-1 has been shown to be effective in alleviating ischemic brain injury ((2005) Nat. Chem. Biol. 1, 112-119), retinal ischemia/reperfusion injury ((2010) J. Neurosci. Res. 88, 1569-1576), Huntington’s disease ((2011) Cell Death Dis. 2 el 15), renal ischemia reperfusion injury ((2012) Kidney Int. 81, 751-761), cisplatin induced kidney injury ((2012) Ren. Fail. 34, 373-377) and traumatic brain injury ((2012) Neurochem. Res. 37, 1849-1858). Other diseases or disorders regulated at least in part by RIPl -dependent apoptosis, necrosis or cytokine production include hematological and solid organ malignancies ((2013) Genes
Dev. 27: 1640-1649), bacterial infections and viral infections ((2014) Cell Host & Microbe 15, 23-35) (including, but not limited to, tuberculosis and influenza ((2013) Cell 153, 1-14)) and Lysosomal storage diseases (particularly, Gaucher Disease, Nature Medicine Advance Online Publication, 19 January 2014, doi: 10.1038/nm.3449).
A potent, selective, small molecule inhibitor of RIP1 kinase activity would block RIP 1 -dependent cellular necrosis and thereby provide a therapeutic benefit in diseases or events associated with DAMPs, cell death, and/or inflammation.
Patent
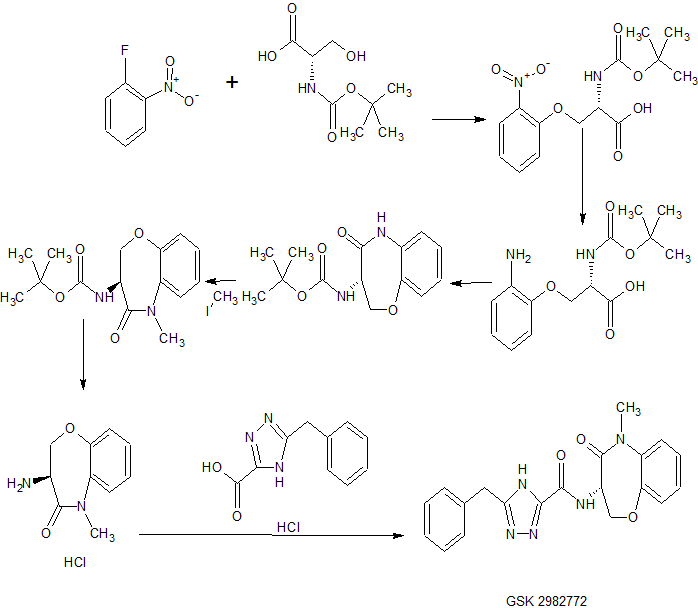
Example 12
Method H
(S)-5-benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][l,4]oxazepin-3-yl)-4H-l,2,4- triazole-3-carboxamide

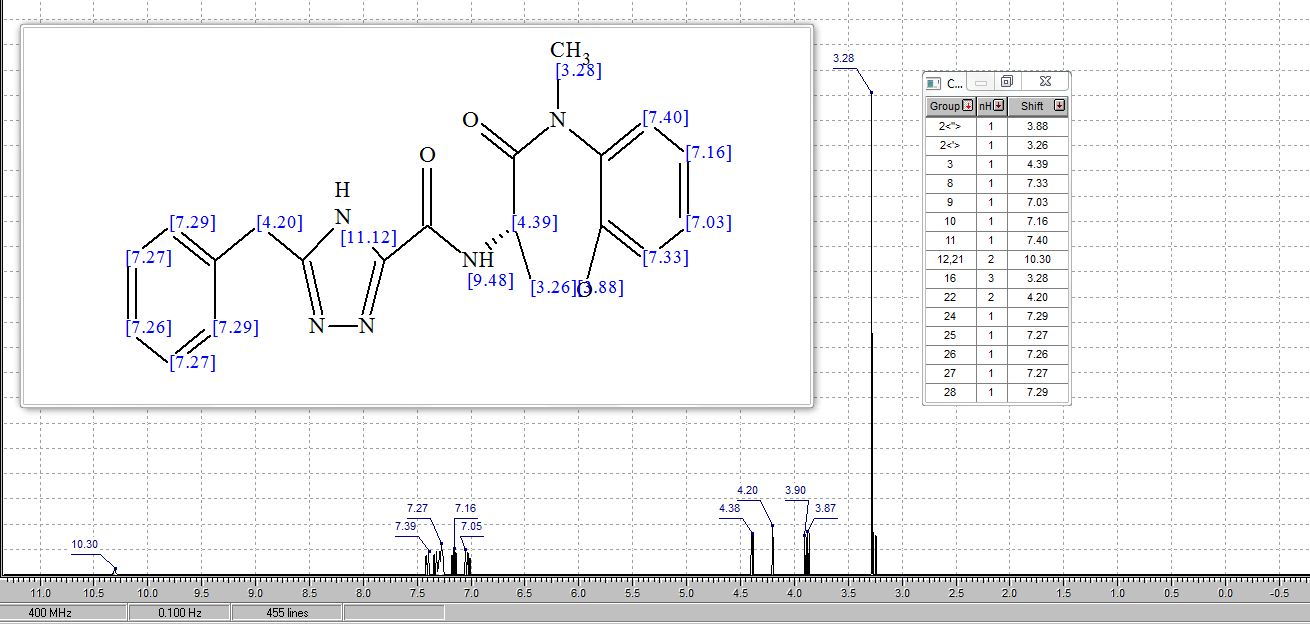
A mixture of (S)-3-amino-5-methyl-2,3-dihydrobenzo[b][l,4]oxazepin-4(5H)-one, hydrochloride (4.00 g, 16.97 mmol), 5-benzyl-4H-l,2,4-triazole-3-carboxylic acid, hydrochloride (4.97 g, 18.66 mmol) and DIEA (10.37 mL, 59.4 mmol) in isopropanol (150 mL) was stirred vigorously for 10 minutes and then 2,4,6-tripropyl-l,3,5,2,4,6-trioxatriphosphinane 2,4,6-trioxide (T3P) (50% by wt. in EtOAc) (15.15 mL, 25.5 mmol) was added. The mixture was stirred at rt for 10 minutes and then quenched with water and concentrated to remove isopropanol. The resulting crude material is dissolved in EtOAc and washed with 1M HC1, satd. NaHC03 and brine. Organics were concentrated and purified by column chromatography (220 g silica column; 20-90% EtOAc/hexanes, 15 min.; 90%, 15 min.) to give the title compound as a light orange foam (5.37 g, 83%). 1H NMR (MeOH-d4) δ: 7.40 – 7.45 (m, 1H), 7.21 – 7.35 (m, 8H), 5.01 (dd, J = 11.6, 7.6 Hz, 1H), 4.60 (dd, J = 9.9, 7.6 Hz, 1H), 4.41 (dd, J = 11.4, 9.9 Hz, 1H), 4.17 (s, 2H), 3.41 (s, 3H); MS (m/z) 378.3 (M+H+).
Alternative Preparation:
To a solution of (S)-3-amino-5-methyl-2,3-dihydrobenzo[b][l,4]oxazepin-4(5H)-one hydrochloride (100 g, 437 mmol), 5-benzyl-4H-l,2,4-triazole-3-carboxylic acid hydrochloride (110 g, 459 mmol) in DCM (2.5 L) was added DIPEA (0.267 L, 1531 mmol) at 15 °C. The reaction mixture was stirred for 10 min. and 2,4,6-tripropyl-l, 3, 5,2,4,6-trioxatriphosphinane 2,4,6-trioxide >50 wt. % in ethyl acetate (0.390 L, 656 mmol) was slowly added at 15 °C. After stirring for 60 mins at RT the LCMS showed the reaction was complete, upon which time it was quenched with water, partitioned between DCM and washed with 0.5N HCl aq (2 L), saturated aqueous NaHC03 (2 L), brine (2 L) and water (2 L). The organic phase was separated and activated charcoal (100 g) and sodium sulfate
(200 g) were added. The dark solution was shaken for 1 h before filtering. The filtrate was then concentrated under reduced pressure to afford the product as a tan foam (120 g). The product was dried under a high vacuum at 50 °C for 16 h. 1H MR showed 4-5% wt of ethyl acetate present. The sample was dissolved in EtOH (650 ml) and stirred for 30 mins, after which the solvent was removed using a rotavapor (water-bath T=45 °C). The product was dried under high vacuum for 16 h at RT (118 g, 72% yield). The product was further dried under high vacuum at 50 °C for 5 h. 1H NMR showed <1% of EtOH and no ethyl acetate. 1H NMR (400 MHz, DMSO-i¾) δ ppm 4.12 (s, 2 H), 4.31 – 4.51 (m, 1 H), 4.60 (t, J=10.36 Hz, 1 H), 4.83 (dt, 7=11.31, 7.86 Hz, 1 H), 7.12 – 7.42 (m, 8 H), 7.42 – 7.65 (m, 1 H), 8.45 (br. s., 1 H), 14.41 (br. s., 1 H). MS (m/z) 378 (M + H+).
Crystallization:
(S)-5-Benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][l,4]oxazepin-3-yl)-4H-l,2,4-triazole-3-carboxamide (100 mg) was dissolved in 0.9 mL of toluene and 0.1 mL of methylcyclohexane at 60 °C, then stirred briskly at room temperature (20 °C) for 4 days. After 4 days, an off-white solid was recovered (76 mg, 76% recovery). The powder X-ray diffraction (PXRD) pattern of this material is shown in Figure 7 and the corresponding diffraction data is provided in Table 1.
The PXRD analysis was conducted using a PANanalytical X’Pert Pro
diffractometer equipped with a copper anode X-ray tube, programmable slits, and
X’Celerator detector fitted with a nickel filter. Generator tension and current were set to 45kV and 40mA respectively to generate the copper Ka radiation powder diffraction pattern over the range of 2 – 40°2Θ. The test specimen was lightly triturated using an agate mortar and pestle and the resulting fine powder was mounted onto a silicon background plate.
Table 1.

Paper
Discovery of a first-in-class receptor interacting protein 1 (RIP1) kinase specific clinical candidate (GSK2982772) for the treatment of inflammatory diseases
J Med Chem 2017, 60(4): 1247

http://pubs.acs.org/doi/pdf/10.1021/acs.jmedchem.6b01751
RIP1 regulates necroptosis and inflammation and may play an important role in contributing to a variety of human pathologies, including immune-mediated inflammatory diseases. Small-molecule inhibitors of RIP1 kinase that are suitable for advancement into the clinic have yet to be described. Herein, we report our lead optimization of a benzoxazepinone hit from a DNA-encoded library and the discovery and profile of clinical candidate GSK2982772 (compound 5), currently in phase 2a clinical studies for psoriasis, rheumatoid arthritis, and ulcerative colitis. Compound 5 potently binds to RIP1 with exquisite kinase specificity and has excellent activity in blocking many TNF-dependent cellular responses. Highlighting its potential as a novel anti-inflammatory agent, the inhibitor was also able to reduce spontaneous production of cytokines from human ulcerative colitis explants. The highly favorable physicochemical and ADMET properties of 5, combined with high potency, led to a predicted low oral dose in humans.
J. Med. Chem. 2017, 60, 1247−1261
(S)-5-Benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b]- [1,4]oxazepin-3-yl)-4H-1,2,4-triazole-3-carboxamide (5).
EtOAc solvate. 1 H NMR (DMSO-d6) δ ppm 14.41 (br s, 1 H), 8.48 (br s, 1 H), 7.50 (dd, J = 7.7, 1.9 Hz, 1 H), 7.12−7.40 (m, 8 H), 4.83 (dt, J = 11.6, 7.9 Hz, 1 H), 4.60 (t, J = 10.7 Hz, 1 H), 4.41 (dd, J = 9.9, 7.8 Hz, 1 H), 4.12 (s, 2 H), 3.31 (s, 3 H). Anal. Calcd for C20H20N5O3·0.026EtOAc·0.4H2O C, 62.36; H, 5.17; N, 18.09. Found: C, 62.12; H, 5.05; N, 18.04.
Synthesis of (<it>S</it>)-3-amino-benzo[<it>b</it>][1,4]oxazepin-4-one via Mitsunobu and S<INF>N</INF>Ar reaction for a first-in-class RIP1 kinase inhibitor GSK2982772 in clinical trials
Tetrahedron Lett 2017, 58(23): 2306
Harris, P.A.
Identification of a first-in-class RIP1 kinase inhibitor in phase 2a clinical trials for immunoinflammatory diseases
ACS MEDI-EFMC Med Chem Front (June 25-28, Philadelphia) 2017, Abst
Harris, P.
Identification of a first-in-class RIP1 kinase inhibitor in phase 2a clinical trials for immuno-inflammatory diseases
253rd Am Chem Soc (ACS) Natl Meet (April 2-6, San Francisco) 2017, Abst MEDI 313
1H NMR AND 13C NMR PREDICT
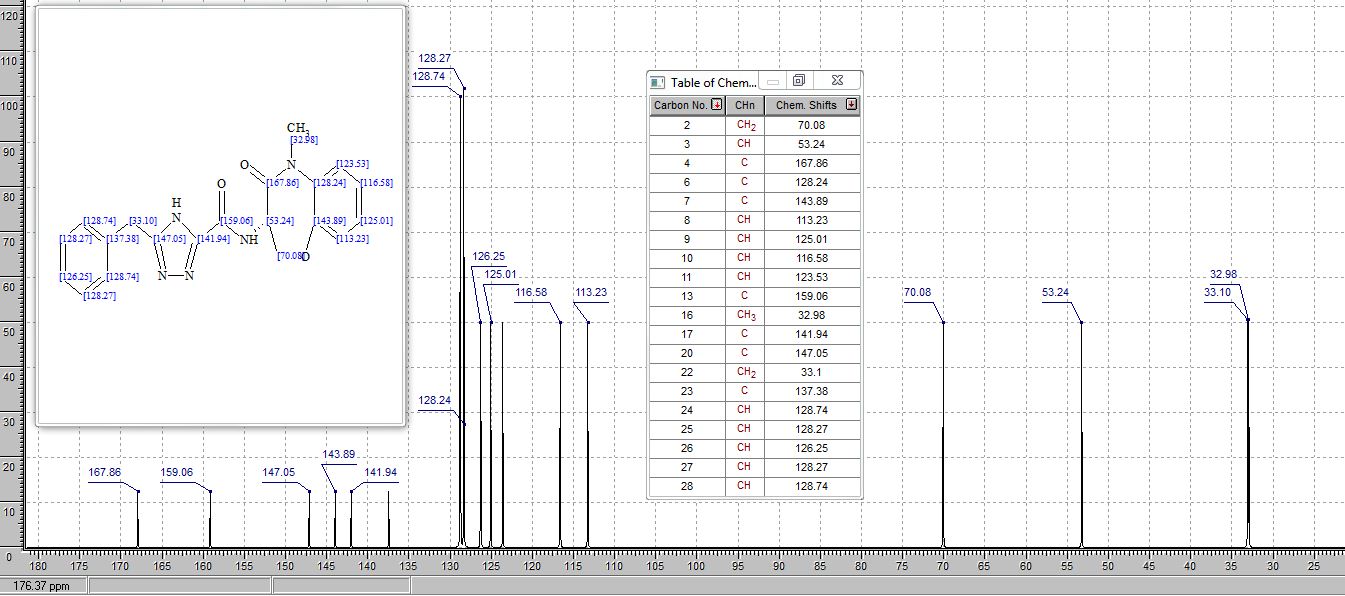
////////////GSK 2982772, phase 2, Plaque psoriasis, Rheumatoid arthritis, Ulcerative colitis
CN3c4ccccc4OC[C@H](NC(=O)c2nnc(Cc1ccccc1)n2)C3=O