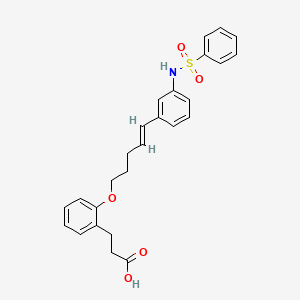
ONO-2910
CAS 2410177-35-2
3- [2-[(E) -5- [3- (benzenesulfonamide) phenyl] penta-4-enoxy] phenyl] propanoic acid
3- [2-[(E) -5- [3- (benzenesulfonamido) phenyl] penta-4-enoxy] phenyl] propanoic acid
ONO Pharmaceuticals is developing ONO-2910 , the lead from a program of novel transient receptor potential cation channel 4/5 inhibitors, for treating peripheral neuropathy. In April 2021, a phase II trial in patients with diabetic polyneuropathy was initiated.
PATENT
CN112513011-BENZENE DERIVATIVE
https://patentscope.wipo.int/search/en/detail.jsf?docId=CN321133877&_cid=P10-KS7EQ7-28392-1
| Example 84: 3-[2-[(E)-5-[3-(Benzenesulfonamido)phenyl]pent-4-enyloxy]phenyl]propionic acid |
| [Chemical formula 52] |
| |
| To a solution of the compound (146 mg) produced in Example 83 in THF (0.5 mL) and methanol (0.1 mL), 1M aqueous lithium hydroxide solution (0.5 mL) was added, and the mixture was stirred at 50°C for 8 hours. 1M hydrochloric acid was added to make it acidic, and it was extracted with ethyl acetate. After drying the organic layer over sodium sulfate, it was concentrated under reduced pressure to obtain the title compound (105 mg) having the following physical properties. |
| HPLC retention time (min): 1.10 |
| 1 H-NMR(CD 3 OD): δ 1.95-2.03, 2.41-2.46, 2.57-2.61,2.92-2.95, 4.03-4.06, 6.24, 6.36, 6.86, 6.90-6.95, 7.06-7.08, 7.11-7.19, 7.45-7.49, 7.55, 7.75 -7.78. |
PATENT
WO-2021153690
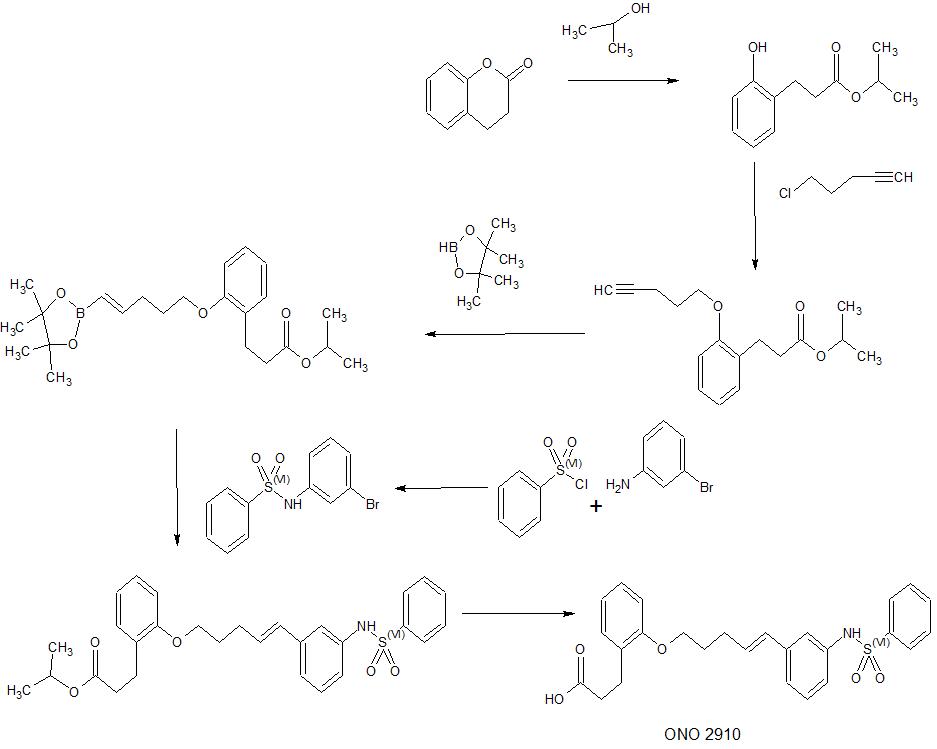
Novel crystalline forms of 3-[2-[(E)-5-[3-(benzenesulfonamide) phenyl] penta-4-enoxy] phenyl] propanoic acid act as neuroprotective, useful for treating neurological disorders eg chronic inflammatory demyelinating polyneuritis, Guillain-Barre syndrome and allergic angiitis.
Sulfuric acid (0.26 mL) is added to a solution of isopropyl 3- (2-hydroxyphenyl) propanoate 3,4-dihydrocoumarin (50.0 g) in isopropyl alcohol (500 mL), and the reaction mixture is mixed at room temperature for 2 hours. Stirred. The reaction mixture was concentrated under reduced pressure, and the obtained residue was diluted with ethyl acetate. The mixture was washed with saturated aqueous sodium hydrogen carbonate solution, water and saturated brine, dried over sodium sulfate, and concentrated under reduced pressure to give the title compound (73.2 g) having the following physical properties.
1 1 H-NMR (CDCl 3 ): δ 1.20, 2.66-2.70, 2.87-2.91, 4.95-5.08, 6.86-6.91, 7.06-7.15, 7.35.
Example 2: Isopropyl 3- (2- (pent-4-in-1-yloxy) phenyl) propanoate In a
HPLC retention time (minutes): 1.13.
a heptane (2 mL) solution of the compound (1.00 g) prepared in Example 2, 4,4,5,5-tetramethyl-1,3,2-dioxaborolane (1. 17 g) and 4-dimethylaminobenzoic acid (60.2 mg) were added, and the mixture was stirred at 100 ° C. for 4 hours. The reaction solution was cooled to room temperature and then concentrated. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 20: 1 → 4: 1) to give the title compound (503 mg) having the following physical characteristics.
HPLC retention time (minutes): 1.38.
Pyridine (0.95 mL), N, N-dimethyl in a solution of N- (3-bromophenyl) benzenesulfonamide 3-bromoaniline (1.02 g) in dichloromethane (20 mL) at 0 ° C. Aminopyridine (hereinafter abbreviated as DMAP) (72.4 mg) and benzenesulfonyl chloride (1.10 g) were added, and the mixture was stirred at room temperature for 2 hours. After concentrating the reaction solution, the obtained residue is purified by silica gel column chromatography (hexane: ethyl acetate = 9: 1 → 2: 1) to give the title compound (1.96 g) having the following physical properties. rice field.
HPLC retention time (minutes): 0.98.
compound prepared in Example 3. In a solution of (180 mg) in THF (3 mL), the compound (168 mg) prepared in Example 3 (1), chloro (2-dicyclohexylphosphino-2′, 4′, 6′-triisopropyl-1,1′- Biphenyl) [2- (2′-amino-1,1′-biphenyl)] palladium (II) (0.035 g) and a 2M tripotassium phosphate aqueous solution (0.67 mL) were added, and the mixture was stirred at 60 ° C. for 1 hour. .. The reaction solution was cooled to room temperature, water was added, and the mixture was extracted with ethyl acetate. The organic layer was dried over sodium sulfate and concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 7: 1 → 2: 1) to give the title compound (113 mg) having the following physical characteristics.
HPLC retention time (minutes): 1.24
Form: Amorphous
HPLC retention time (minutes): 1.101
1 H-NMR (CD 3 OD): δ 1.95-2.03, 2.41-2.46, 2.57-2.61, 2.92-2.95, 4.03-4.06, 6.24, 6.36, 6.86, 6.90-6.95, 7.06-7.08, 7.11-7.19, 7.45-7.49, 7.55, 7.75-7.78.
PATENT
WO2020027150
https://patents.google.com/patent/WO2020027150A1/en
Example 83: Isopropyl (E) -3- (2-((5- (3- (phenylsulfonamido) phenyl) penta-4-en-1-yl) oxy) phenyl) propanoate The compound prepared in Example 82 Compound (168 mg) prepared in Example 9 and chloro (2-dicyclohexylphosphino-2 ′, 4 ′, 6′-triisopropyl-1,1′-biphenyl) [180 mg) in THF (3 mL) solution were added. 2- (2′-Amino-1,1′-biphenyl)] palladium (II) (0.035 g) and a 2M aqueous solution of tripotassium phosphate (0.67 mL) were added, and the mixture was stirred at 60 ° C. for 1 hour. After cooling the reaction solution to room temperature, water was added, and the mixture was extracted with ethyl acetate. The organic layer was dried over sodium sulfate and concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography (hexane: ethyl acetate = 7: 1 → 2: 1) to give the title compound (113 mg) having the following physical data.
HPLC retention time (min): 1.24.
HPLC retention time (min): 1.10
1 H-NMR (CD 3 OD): δ 1.95-2.03, 2.41-2.46, 2.57-2.61, 2.92-2.95, 4.03-4.06, 6.24, 6.36, 6.86, 6.90-6.95, 7.06-7.08, 7.11-7.19, 7.45 -7.49, 7.55, 7.75-7.78.
///////////ONO-2910, ONO 2910, PHASE 2,
O=S(=O)(Nc1cc(\C=C\CCCOc2ccccc2CCC(=O)O)ccc1)c1ccccc1