
This paper demonstrates an efficient, mild, and chemoselective synthesis of various thioesters via a HOTf-catalyzed S-acetylation of aromatic and aliphatic thiols using isopropenyl acetate as a cheap and convenient acetylating agent. During our tests, we also investigated the differences between the activities of metal triflates and triflic acid as catalysts in the acetylation of thiols. Finally, the potential of our concept has been increased by the implementation of Nafion as a heterogeneous catalyst for S-acetylation of thiols.
S-Acetylation of Thiols Mediated by Triflic Acid: A Novel Route to Thioesters
Faculty of Chemistry, Adam Mickiewicz University in Poznań, ul. Umultowska 89b, 61-614 Poznań, Poland
Org. Process Res. Dev., Article ASAP
DOI: 10.1021/acs.oprd.7b00378
Publication Date (Web): March 12, 2018
Copyright © 2018 American Chemical Society
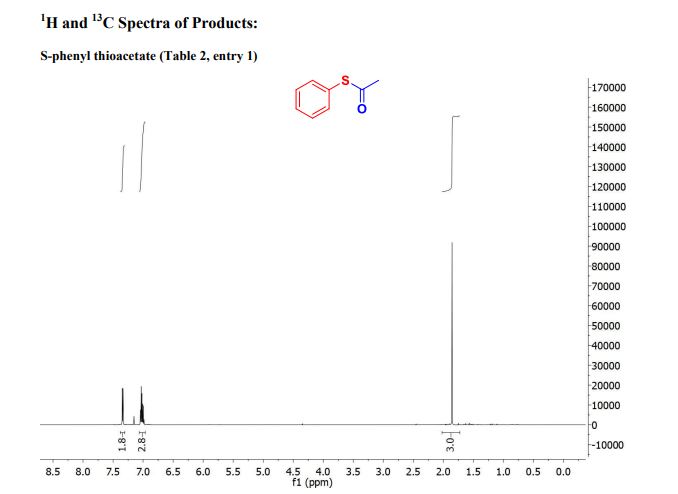
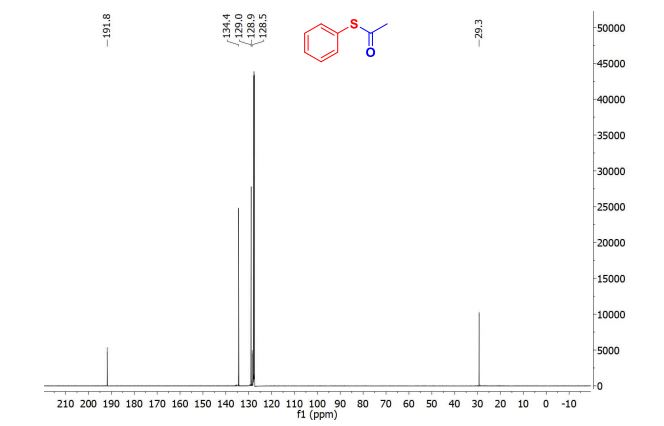
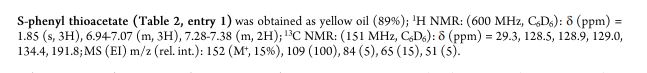
////////















