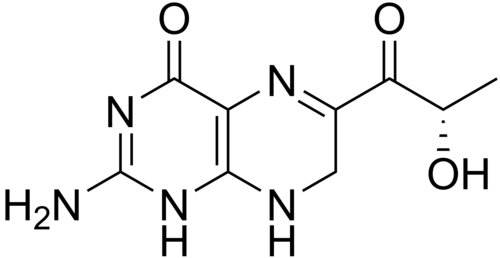
Sepiapterin
- Sepiapterine
- CNSA-001
- CJQ26KO7HP
| Molecular Weight | 237.22 |
|---|---|
| Formula | C9H11N5O3 |
2-amino-6-[(2S)-2-hydroxypropanoyl]-7,8-dihydro-3H-pteridin-4-one
(S)-2-Amino-6-(2-hydroxypropanoyl)-7,8-dihydropteridin-4(3H)-one
- 1-(2-amino-7,8-dihydro-4-hydroxy-6-pteridinyl)-2-hydroxy-1-Propanone
- 2-amino-6-[(2S)-2-hydroxypropanoyl]-7,8-dihydropteridin-4(1H)-one
- 2-amino-7,8-dihydro-6-[(2S)-2-hydroxy-1-oxopropyl]-4(1H)Pteridinone
- S(-)-2-Amino-7,8-dihydro-6-(2-hydroxy-1-oxopropyl)-4(1H)-pteridione
- S-(-)-2-Amino-7,8-dihydro-6-(2-hydroxy-1-oxopropyl)-4(1H)-pteridinone
- 2-AMINO-7,8-DIHYDRO-6-((2S)-2-HYDROXY-1-OXOPROPYL)-4(3H)-PTERIDINONE
- 4(1H)-Pteridinone, 2-amino-7,8-dihydro-6-(2-hydroxy-1-oxopropyl)-, (S)-
7/28/2025 fda approved, Sephience, To treat hyperphenylalaninemia in patients with sepiapterin-responsive phenylketonuria, in conjunction with a phenylalanine-restricted diet
Sepiapterin, sold under the brand name Sephience, is a medication used for the treatment of hyperphenylalaninemia.[2][3] Sepiapterin is a phenylalanine hydroxylase activator.[1]
The most common side effects are upper respiratory tract infection, headache, diarrhea, abdominal pain, hyperphenylalaninemia and discoloration of feces.[2]
Syn
https://patents.google.com/patent/WO2013168693A1/en
Sepiapterin is synthesized by a method of reacting 7,8-dihydropterin and α-keto-β-hydroxybutyric acid in the presence of zinc chloride (Non-patent Document 1), and a method of oxidizing BH4 in air for 6 days. (Non-Patent Document 2) is known.
As a method for synthesizing lactoylpterin, it is known that it can be obtained by oxidizing sepiapterin (Non-patent Documents 3 and 4).International Publication No. 2011/132435
However, the method described in Non-Patent Document 1 produces only a trace amount of sepiapterin and cannot be a stable supply method. Further, in the method of Non-Patent Document 2, very expensive BH4 is used as a raw material, and this cannot be a method that can be industrially stably supplied. Further, the method of Non-Patent Document 2 has a problem that the reaction time is long and many by-products such as biopterin in which BH4 is oxidized and deoxysepiapterin from which the β-position hydroxyl group of the side chain is eliminated are also generated. . In addition, the methods for synthesizing lactoylpterin of Non-Patent Documents 3 and 4 use sepiapterin, which is difficult to obtain industrially, as a raw material, and the yield is low, which cannot be a stable supply method.
Accordingly, an object of the present invention is to provide a novel production method capable of stably supplying sepiapterin, lactoylpterin and tetrahydrolactoylpterin, which have recently been found to be useful as pharmaceuticals.
Therefore, the present inventor has studied a method for synthesizing sepiapterin, lactoylpterin, and tetrahydrolactoylpterin using available raw materials. As a starting material, the compound of the following formula (1) or the compound of formula (7) is used. As a result, it was found that sepiapterin, lactoylpterin and tetrahydrolactoylpterin can be obtained in good yield, and these compounds can be stably supplied as a medicine for the first time, thereby completing the present invention.
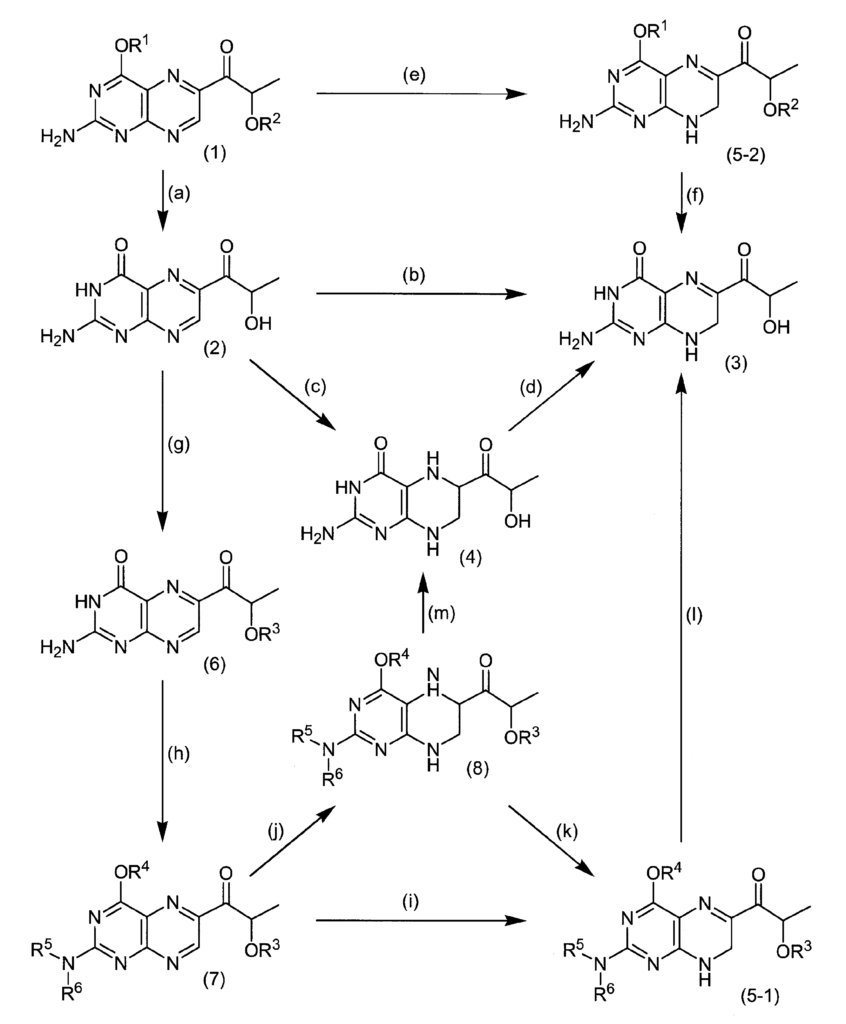
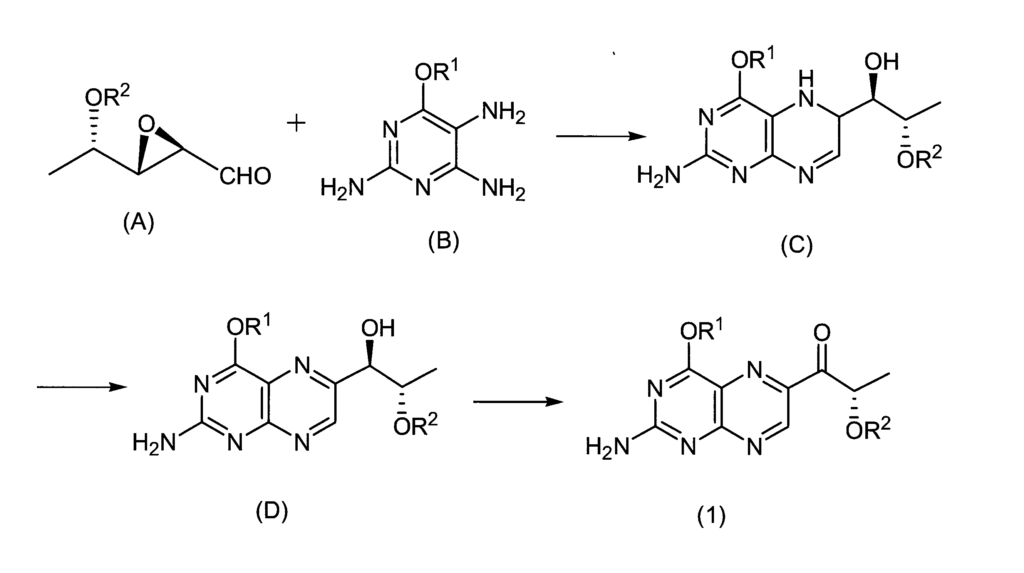
Example 1
Synthesis of S-lactoylpterin (2)

1- (2-Amino-4-cyclohexyloxypteridin-6-yl) -2S-methoxymethoxypropan-1-one (compound (1)) 24.7 g (68.2 mmol) in methanol 50 mL, 3 mol / L hydrochloric acid 250 mL And stirred at 50 ° C. for 3 hours. The reaction solution was adjusted to pH = 7 with an aqueous sodium hydroxide solution, collected by filtration, and dried under reduced pressure to obtain 15.1 g (64.2 mmol, 94% yield) of S-lactoylpterin.
(S-lactoylpterin: (2))
1 H NMR (DMSO-d 6 ): δ / ppm = 1.32 (d, 3H, J = 6.8Hz), 5.16 (br, 1H), 5.32 (q, 1H, J = 6.8Hz), 9.09 (s, 1H )
Example 2 (Synthesis of 1- (2-amino-4-cyclohexyloxypteridin-6-yl) -2S-hydroxypropan-1-one)

1- (2-Amino-4-cyclohexyloxypteridin-6-yl) -2St-butyldimethylsilanoxypropan-1-one (compound (1)) (4.0 g, 9.27 mmol) was added to THF 40 mL, 70% 6.92 g (18.5 mmol) of tetrabutylammonium fluoride was added and stirred at 10 ° C. or lower for 2 hours. Water was added to the reaction mixture, and the mixture was extracted with chloroform. The organic layer was dehydrated and concentrated under reduced pressure. The crude product was purified by flash chromatography to give 2.09 g (6.59 mmol, 71% yield) of 1- (2-amino-4-cyclohexyloxypteridin-6-yl) -2S-hydroxypropan-1-one. Got.
1 H NMR (DMSO-d 6 ): δ / ppm = 1.38 (d, 3H, J = 6.6Hz), 1.37-1.79 (m, 8H), 1.98-1.99 (m, 2H), 5.20 (d, 1H, J = 6.3Hz), 5.34 (dq, 1H, J = 6.6Hz), 5.29-5.37 (m, 1H), 7.68 (br, 1H), 7.82 (br, 1H), 9.22 (s, 1H)
Example 3
Synthesis of S-lactoylpterin hydrochloride

To 500 mg (2.13 mmol) of S-lactoylpterin were added 1.25 mL of 6 mol / L hydrochloric acid and 10 mL of ethanol, and the mixture was stirred for 30 minutes. The crystals were collected by filtration and dried under reduced pressure, and 465 mg of S-lactoylpterin hydrochloride (1. 71 mmol, yield 80%).
(S-lactoylpterin hydrochloride)
1 H NMR (DMSO-d 6 ): δ / ppm = 1.34 (d, 3H, J = 6.9Hz), 3.91 (br, 3H), 5.34 (q, 1H, J = 6.9Hz), 9.12 (s, 1H )
Example 4 Synthesis of 2-amino-6- (2S-hydroxypropionyl) -7,8-dihydro-3H-pteridin-4-one (S-sepiapterin)

To 500 mg (2.13 mol) of S-lactoylpterin were added 125 mL of methanol, 2.08 mL (14.9 mmol) of triethylamine, 250 mg of 8.4% Pd / C (Ph 2 S) (containing 50% water), and an external temperature of 40 ° C. The hydrogenation reaction was carried out for 3 hours. After completion of the reaction, the reaction solution was stirred in air at room temperature for 1 hour, and then the catalyst was filtered off from the reaction solution and concentrated under reduced pressure. The crude product was separated and purified by flash chromatography and 296 mg (1.25 mmol) of S-sepiapterin. Yield 59%).
(S-sepiapterin: (3))
1 H NMR (DMSO-d 6 ): δ / ppm = 1.21 (d, 3H, J = 6.6Hz), 4.11 (s, 2H), 4.89 (d, 1H, J = 6.6Hz), 5, 10 (quin ., 1H, J = 6.6Hz), 6.81 (br-s, 2H), 7.51 (s, 1H), 10.26 (s, 1H)
Example 5
To 20 mg (0.085 mmol) of S-lactoylpterin were added 2 mL of saturated aqueous sodium hydrogen carbonate and 76 mg (0.44 mmol) of sodium dithionite, and the mixture was stirred at room temperature for 2 hours to give S-sepiapterin as a mixture.
Example 6
A reaction was carried out in the same manner as in Example 5 except that 20% (0.085 mmol) of S-lactoylpterin was used and the saturated aqueous sodium bicarbonate solution was changed to an aqueous sodium borate solution to give S-sepiapterin as a mixture.
Example 7
Synthesis of S-sepiapterin hydrochloride

To 620 mg (2.61 mmol) of S-sepiapterin were added 2.5 mL of 6 mol / L hydrochloric acid and 5.0 mL of ethanol, and the mixture was stirred at 0 ° C. for 30 minutes. The crystals were collected by filtration and dried under reduced pressure to obtain 650 mg (2.38 mmol, yield 91%) of S-sepiapterin hydrochloride.
(S-sepiapterin hydrochloride)
1 H NMR (DMSO-d 6 ): δ / ppm = 1.22 (d, 3H, J = 6.9Hz), 4.14 (s, 2H), 4.89 (d, 1H, J = 6.6Hz), 5.11 (q, 1H , J = 6.9Hz), 7.40 (br-s, 4H), 7.80 (br-s, 1H)
Example 8 (Synthesis of 2-amino-6- (2S-hydroxypropionyl) -5,6,7,8-tetrahydro-3H-pteridin-4-one (S-tetrahydrolactoylpterin) dihydrochloride)

Methanol 50 mL, 6 mol / L hydrochloric acid 5 mL, and borane pyridine complex 593 mg (6.38 mmol) were added to 1.00 g (4.25 mmol) of S-lactoylpterin, and the mixture was stirred at an external temperature of 0 ° C. for 1 hour. After completion of the reaction, 5 mL of acetone was added, concentrated under reduced pressure, azeotropically dehydrated with ethanol, ethanol was added, the crystals were filtered and dried under reduced pressure, and a mixture 1 of S-tetrahydrolactoylpterin dihydrochloride (4a) and (4b) 1 .12 g (3.59 mmol, 85% yield) was obtained.
(6S—S-tetrahydrolactoylpterin dihydrochloride: (4a))
1 H NMR (DMSO-d 6 ): δ / ppm = 1.24 (d, 3H, J = 6.9Hz), 3.45 (dd, 1H, J = 7.2, 13.5Hz), 3.87 (dd, 1H, J = 3.3, 13.5Hz), 4.34 (q, 1H, J = 6.9Hz), 4.53 (dd, 1H, J = 3.3, 7.2Hz), 7.03 (br-s, 4H), 7.67 (br-s, 1H)
(6R-S-tetrahydrolactoylpterin dihydrochloride: (4b))
1 H NMR (DMSO-d 6 ): δ / ppm = 1.24 (d, 3H, J = 6.9Hz), 3.45 (dd, 1H, J = 6.9, 13.5Hz), 3.91 (dd, 1H, J = 3.3, 13.5Hz), 4.31 (q, 1H, J = 6.6Hz), 4.55 (dd, 1H, J = 3.3, 6.9Hz), 7.12 (br-s, 3H), 7.71 (br-s, 2H)
Example 9
To 3.00 g (12.8 mmol) of S-lactoylpterin was added 150 mL of methanol, 15 mL of 6 mol / L hydrochloric acid, and 1.78 g (19.1 mmol) of borane pyridine complex, and the mixture was stirred at an external temperature of 0 ° C. for 1 hour. After completion of the reaction, 45 mL of concentrated hydrochloric acid was added, and the mixture was stirred overnight at the same temperature. The crystals were collected by filtration and dried under reduced pressure, and 1.63 g (5.2 mmol, yield) of 6S-S-tetrahydrolactoylpterin dihydrochloride (4a) 41%). The filtrate was concentrated under reduced pressure, azeotropically dehydrated with ethanol, ethanol was added, the crystals were collected by filtration and dried under reduced pressure, and 1.38 g (4.4 mmol) of 6R-S-tetrahydrolactoylpterin dihydrochloride (4b) was collected. Yield 35%). It was confirmed that the obtained compound was consistent with the spectrum data described in Example 8.
Example 10
Methanol 5 mL, 6 mol / L hydrochloric acid 0.5 mL, and borane pyridine complex 59 mg (0.64 mmol) were added to 100 mg (0.43 mmol) of S-lactoylpterin, and the mixture was stirred overnight at an external temperature of 0 ° C. The precipitated crystals were collected by filtration and dried under reduced pressure to obtain 46 mg (0.15 mmol, yield 35%) of 6S—S-tetrahydrolactoylpterin dihydrochloride (4a). It was confirmed that the obtained compound was consistent with the spectrum data described in Example 8.
Example 11
To 200 mg (0.85 mol) of S-lactoylpterin, 50 mL of methanol, 0.62 mL (5.95 mmol) of diethylamine and 100 mg of 8.4% Pd / C (Ph 2 S) (containing 50% water) were added, and the external temperature was 40 ° C. The hydrogenation reaction was carried out for 2.5 hours. After completion of the reaction, concentrated hydrochloric acid is added, the catalyst is filtered off, concentrated under reduced pressure, azeotropically dehydrated with ethanol, ethanol is added, the crystals are filtered and dried under reduced pressure, and S-tetrahydrolactoylpterin dihydrochloride (4a) and 122 mg (0.39 mmol, 46% yield) of a mixture of (4b) was obtained. It was confirmed that the obtained compound was consistent with the spectrum data described in Example 8.
Example 12 (Synthesis of 2-amino-6- (2S-hydroxypropionyl) -5,6,7,8-tetrahydro-3H-pteridin-4-one (S-tetrahydrolactoylpterin) ditoluenesulfonate)

To 100 mg (0.43 mmol) of S-lactoylpterin was added 5 mL of methanol, 0.5 mL of water, 566 mg (2.98 mmol) of p-toluenesulfonic acid monohydrate, and 59 mg (0.64 mmol) of borane pyridine complex. Stir at 0 ° C. for 1 hour. After completion of the reaction, 0.5 mL of acetone was added and concentrated under reduced pressure. After azeotropic dehydration with ethanol, acetone was added, the crystals were collected by filtration and dried under reduced pressure, and S-tetrahydrolactoylpterin ditoluenesulfonate 158 mg (0.27 mmol, Yield 63%) was obtained.
(S-tetrahydrolactoylpterin ditoluenesulfonate)
1 H NMR (DMSO-d 6 ): δ / ppm = 1.25 (d, 3H, J = 7.2Hz), 2.29 (S, 6H), 3.35 (dd, 1H, J = 7.5, 13.5Hz), 3.84 (dd , 1H, J = 3.0, 13.5Hz), 4.35 (q, 1H, J = 6.9Hz), 4.49 (dd, 1H, J = 3.0, 7.5Hz), 6.72 (br-s, 2H), 7.13 (d, 4H, J = 8.1Hz), 7.49 (d, 4H, J = 8.1Hz), 7.62 (br-s, 1H), 10.66 (br-s, 1H)
1 H NMR (DMSO-d 6 ): δ / ppm = 1.25 (d, 3H, J = 7.2Hz), 2.29 (S, 6H), 3.33 (dd, 1H, J = 7.5, 13.5Hz), 3.84 (dd , 1H, J = 3.0, 13.5Hz), 4.32 (q, 1H, J = 6.9Hz), 4.49 (dd, 1H, J = 3.0, 7.5Hz), 6.72 (br-s, 2H), 7.13 (d, 4H, J = 8.1Hz), 7.49 (d, 4H, J = 8.1Hz), 7.62 (br-s, 1H), 10.66 (br-s, 1H)
Example 13 Synthesis of 2-amino-6- (2S-hydroxypropionyl) -7,8-dihydro-3H-pteridin-4-one (S-sepiapterin)

6 mL of water and 6 mL of ethanol were added to 1.00 g (3.20 mmol) of S-tetrahydrolactoylpterin dihydrochloride, and 363 mg (3.20 mmol) of 30% aqueous hydrogen peroxide was added at an external temperature of −10 ° C. at the same temperature. Stir for 2 hours. A sodium sulfite aqueous solution was added to the reaction solution, and the crystals were collected by filtration and dried under reduced pressure to obtain 676 mg (2.85 mmol, yield 89%) of S-sepiapterin. It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 14
46 mg of S-sepiapterin was prepared in the same manner as in Example 13 except that 100 mg (0.32 mmol) of S-tetrahydrolactoylpterin dihydrochloride was changed to 68 mg (0.32 mmol) of 36% peracetic acid with 30% hydrogen peroxide. 0.19 mmol, 61% yield). It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 15
S-Sepia was prepared in the same manner as in Example 13 except that 100 mg (0.32 mmol) of S-tetrahydrolactoylpterin dihydrochloride was changed to 85 mg of m-CPBA (content 65%, 0.32 mmol) with 30% hydrogen peroxide. 35 mg (0.15 mmol, 46% yield) of pterin was obtained. It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 16
20 mL of methanol and 0.89 mL (6.40 mmol) of triethylamine were added to 200 mg (0.64 mmol) of S-tetrahydrolactoylpterin dihydrochloride, and the mixture was stirred at room temperature for 1 hour in air. The reaction mixture was concentrated under reduced pressure, water was added, the crystals were collected by filtration and dried under reduced pressure to obtain 105 mg (0.44 mmol, yield 69%) of S-sepiapterin. It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 17
Add 20 mL of methanol to 200 mg (0.64 mmol) of S-tetrahydrolactoylpterin dihydrochloride, neutralize with 0.16 mL (1.28 mmol) of 8 mol / L aqueous sodium hydroxide solution, and stir in air at room temperature for 1 hour did. The reaction mixture was concentrated under reduced pressure, water was added, the crystals were collected by filtration and dried under reduced pressure to obtain 87 mg (0.37 mmol, yield 58%) of S-sepiapterin. It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 18 (Synthesis of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxymethoxypropan-1-one)

1- (2-Amino-4-cyclohexyloxypteridin-6-yl) -2S-methoxymethoxypropan-1-one (1.00 g, 2.77 mmol), ethyl acetate 60 mL, 10% Pd—C 500 mg, potassium carbonate 3 .82 g (27.6 mmol) was added, and the hydrogenation reaction was performed at an external temperature of 50 ° C. for 3 hours. After the catalyst was filtered off, the reaction solution was concentrated under reduced pressure. The crude product was separated and purified by flash chromatography to obtain 257 mg (0.71 mmol) of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxymethoxypropan-1-one. Yield 26%).
1 H NMR (CDCl 3 ): δ / ppm = 1.33-1.47 (m, 3H), 1.44 (d, 3H, J = 6.9Hz), 1.54-1.63 (m, 3H), 1.79 (m, 2H), 1.91 (m, 2H), 3.37 (s, 3H), 4.36 (d, 1H, J = 15.6), 4.43 (d, 1H, J = 15.6), 4.71 (d, 1H, J = 6.6Hz), 4.74 (d , 1H, J = 6.6Hz), 4.90 (br-s, 2H), 5.00 (br-s, 1H), 5.05-5.11 (m, 1H), 5.34 (q, 1H, J = 6.9Hz)
Example 19 (Synthesis of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxyethoxymethoxypropan-1-one)

100 mg (0.56 mmol) of ascorbic acid was weighed and 2 mL of water was added. 1- (2-amino-4-cyclohexyloxypteridin-6-yl) -2S-methoxyethoxymethoxypropane- dissolved in 2 mL of methanol after neutralizing the pH of the solution with 1 mol / L aqueous sodium hydroxide solution 20 mg (0.054 mmol) of 1-one was added. To this, 80 mg (0.46 mmol) of Na 2 S 2 O 4 was added and stirred at room temperature for 1 hour. Water was added to the reaction solution, and the mixture was extracted with ethyl acetate. After dehydrating the organic phase, the solvent was concentrated under reduced pressure. Separation and purification by silica gel column chromatography gave 4.4 mg (0.011 mmol) of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxyethoxymethoxypropan-1-one. Yield 20%).
1 H NMR (CDCl 3 ): δ / ppm = 1.13 (m, 1H), 1.44 (d, 3H, J = 6.8 Hz), 1.63 (m, 1H), 1.80 (m, 2H), 1.93 (m, 2H ), 2.06 (m, 2H), 3.37 (s, 3H), 3.52 (m, 2H), 3.70 (t, J = 4.6 Hz, 2H), 4.40 (m, 2H), 4.81 (m, 2H), 5.11 (tt, J = 3.9, 8.5 Hz, 1H), 5.35 (q, J = 6.8 Hz, 1H)
Example 20
Synthesis of S-sepiapterin (3)

To 10 mg of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxymethoxypropan-1-one (compound (5-2)) was added 0.1 mL of concentrated hydrochloric acid. , Warmed up. The reaction solution was diluted with water, neutralized to pH 6-7 with an aqueous sodium hydroxide solution, and the precipitated crystals were filtered off. The filtrate was concentrated under reduced pressure to obtain S-sepiapterin as a mixture. The resulting compound was consistent with the spectral data described in Example 4.
Example 21
Synthesis of S-sepiapterin (3)

4.0 mg (9.8 μmol) of 1- (2-amino-4-cyclohexyloxy-7,8-dihydropteridin-6-yl) -2S-methoxyethoxymethoxypropan-1-one in 2 mL of methanol, ascorbic acid 3 0.02 mg was added, 2 mL of 3 mol / L hydrochloric acid was added thereto, and the mixture was stirred at 50 ° C. for 6 hours while shielding light. The solution was adjusted to pH 7 with 28% aqueous ammonia, washed with ethyl acetate, and purified by Florisil column chromatography to obtain 2.0 mg (8.4 μmol, yield 86%) of S-sepiapterin. As a result of HPLC measurement, the retention time and the UV waveform of the peak coincided with the standard S-sepiapterin.
Example 22 (Synthesis of 2-amino-6- [2S- (tert-butyldimethylsilanyloxy) -propionyl] -3H-pteridin-4-one (6))

To 3.00 g (12.8 mmol) of S-lactoylpterin were added 30 mL of DMF, 2.61 g (38.3 mmol) of imidazole and 3.84 g (25.5 mmol) of TBSCl, and the mixture was stirred for 1 hour under ice cooling. Water was added to the reaction mixture, and the crystals were collected by filtration and dried under reduced pressure to give 3.91 g of 2-amino-6- [2S- (tert-butyldimethylsilanyloxy) -propionyl] -3H-pteridin-4-one (6). (11.2 mmol, 88% yield) was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 0.01 (s, 3H), 0.06 (s, 3H), 0.83 (s, 9H), 1.36 (d, 3H, J = 6.9Hz), 5.55 (q , 1H, J = 6.9Hz), 9.10 (s, 1H), 11.73 (br-s, 1H)
Example 23 (Synthesis of 2-amino-6- [2S- (triisopropylsilanyl) -propionyl] -3H-pteridin-4-one (6))
2-amino-6- [2S- (triisopropylsilanyl) -propionyl] -3H- was prepared in the same manner as in Example 22 except that TBSCl was changed to TIPSCl from 300 mg (1.28 mmol) of S-lactoylpterin. Pteridin-4-one (6) (339 mg, 0.87 mmol, yield 68%) was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 0.89-1.15 (m, 21H), 1.40 (d, 3H, J = 6.9Hz), 5.71 (q, 1H, J = 6.9Hz), 9.13 (s , 1H), 11.74 (br-s, 1H)
Example 24 (Synthesis of 2-amino-6- [2S- (tert-butyldiphenylsilanyl) -propionyl] -3H-pteridin-4-one (6))

2-amino-6- [2S- (triisopropylsilanyl) -propionyl] -3H- was prepared in the same manner as in Example 22 except that TBSCl was changed to TBDPSCl from 300 mg (1.28 mmol) of S-lactoylpterin. 498 mg (1.05 mmol, yield 82%) of pteridin-4-one (6) was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 1.03 (s, 9H), 1.38 (d, 3H, J = 6.9Hz), 5.71 (q, 1H, J = 6.9Hz), 7.23-7.33 (m , 3H), 7.37-7.45 (m, 3H), 7.50-7.59 (m, 2H), 7.61-7.71 (m, 2H), 8.96 (s, 1H), 11.67 (br-s, 1H)
Example 25 (1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one Synthesis of (7))

2-amino-6- [2S- (tert-butyldimethylsilanyloxy) -propionyl] -3H-pteridin-4-one 1.50 g (4.29 mmol) and 75 mL of ethyl acetate, di-tert-butyl dicarbonate 4 .68 g (21.4 mmol) and N, N-dimethylaminopyridine 52 mg (0.43 mmol) were added, and the mixture was heated to reflux for 1 hour. The reaction solution is washed with water, and the organic layer is dehydrated and concentrated under reduced pressure to give 2- (N, N-di-tert-butylcarbonyl) -amino-6- [2S- (tert-butyldimethylsilanyloxy) -propionyl]- 2.18 g (3.35 mmol, yield 78%) of 3H-pteridin-4-one (7) was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 0.01 (s, 3H), 0.08 (s, 3H), 0.75 (s, 9H), 1.40 (d, 3H, J = 6.6Hz), 1.48 (s , 18H), 1.71 (s, 9H), 5.59 (q, 1H, J = 6.6Hz), 9.53 (s, 1H)
Example 26 (1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butoxycarbonyloxypropan-1-one (7 )

To 1.00 g (4.25 mmol) of S-lactoylpterin was added 50 mL of THF, 4.64 g (21.3 mmol) of di-tert-butyl dicarbonate, and 30 mg (0.25 mmol) of N, N-dimethylaminopyridine. Heated to reflux for hours. The reaction solution was concentrated under reduced pressure, and the crude product was separated and purified by flash chromatography, and 1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] There was obtained 0.30 g (0.47 mmol, yield 11%) of -2S-tert-butoxycarbonyloxypropan-1-one (7).
1 H NMR (CDCl 3 ): δ / ppm = 1.26 (s, 9H), 1.27 (d, 3H, J = 7.2Hz), 1.45 (s, 18H), 1.71 (s, 9H), 6.11 (q, 1H , J = 7.2Hz), 6.73 (s, 1H)
Example 27 (Synthesis of 1- [4-cyclohexyloxy-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-methoxymethoxypropan-1-one)

1- (2-Amino-4-cyclohexyloxypteridin-6-yl) -2S-methoxymethoxypropan-1-one (1.00 g, 2.76 mmol) was added with 20 mL of THF and 1.27 g of di-tert-butyl dicarbonate ( 5.82 mmol) and 3.4 mg (0.03 mmol) of N, N-dimethylaminopyridine were added, and the mixture was heated to reflux for 1 hour. The reaction solution was concentrated under reduced pressure to give 1- [4-cyclohexyloxy-2- (N, N-di-tert-butylcarbonyl) aminopteridin-6-yl] -2S-methoxymethoxypropan-1-one (7) 1 Obtained .55 g (2.76 mmol, 100% yield).
1 H NMR (DMSO-d 6 ): δ / ppm = 1.45-1.88 (m, 8H), 1.53 (s, 18H), 1.58 (d, 3H, J = 6.9Hz), 2.10-2.14 (m, 2H) , 3.38 (s, 3H), 4.78 (d, 1H, J = 6.9Hz), 4.84 (d, 1H, J = 6.9Hz), 5.36-5.45 (m, 1H), 5.55 (q, 1H, J = 6.9 Hz), 9.65 (s, 1H)
Example 28 (1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-7,8-dihydropteridin-6-yl] -2S-tert-butyldimethylsilanyl Synthesis of oxypropan-1-one)

1- [4-tert-Butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one 1.31 g ( 2.02 mmol) was added with 130 mL of ethyl acetate, 655 mg of 10% Pd—C and 2.78 g (20.1 mmol) of potassium carbonate, and the hydrogenation reaction was carried out for 1 hour at an external temperature of 50 ° C. under normal pressure (H 2 balloon). . After the catalyst was filtered off, the reaction solution was stirred in air at room temperature overnight, and the reaction solution was concentrated under reduced pressure. The crude product was separated and purified by flash chromatography, and 1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-7,8-dihydropteridin-6-yl]- 684 mg (1.05 mmol, 66% yield) of 2S-tert-butyldimethylsilanyloxypropan-1-one was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 0.01 (s, 3H), 0.07 (s, 3H), 0.82 (s, 9H), 1.24 (d, 3H, J = 6.6Hz), 1.42 (s , 18H), 1.53 (s, 9H), 4.23 (d, 1H, J = 16.5Hz), 4.32 (d, 1H, J = 16.5Hz), 5.39 (q, 1H, J = 6.6Hz), 7.92 (s , 1H)
Example 29 (Synthesis of 2-amino-6S- (2S-hydroxypropionyl) -5,6,7,8-tetrahydro-3H-pteridin-4-one (6S-S-tetrahydrolactoylpterin) dihydrochloride)

1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one 4.92 g ( 7.57 mmol) was added 250 mL of ethyl acetate, 2.46 g of 10% Pd—C, and 10.5 g (76.0 mmol) of K 2 CO 3 , and the hydrogenation reaction was performed at an external temperature of 50 ° C. under normal pressure (H 2 balloon). It went for 1 hour. After the catalyst was filtered off, the reaction solution was concentrated under reduced pressure, 49 mL of concentrated hydrochloric acid was added, and the mixture was concentrated under reduced pressure. Ethanol was added to the concentrate, and the crystals were collected by filtration and dried under reduced pressure to obtain 1.79 g (5.73 mmol, yield 76%) of 6S—S-tetrahydrolactoylpterin dihydrochloride (4a). The compound obtained agreed with the spectral data described in Example 8.
Example 30
1- [4-tert-Butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one 500 mg (0. 164 mg (0.53 mmol, 68% yield) of 6S—S-tetrahydrolactoylpterin dihydrochloride (4a) was obtained in the same manner as in Example 29 except that the amount of 10% Pd / C was changed to 100 mg from 77 mmol). Obtained. It was confirmed that the obtained compound was consistent with the spectrum data described in Example 8.
Example 31 (1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-5,6,7,8-tetrahydropteridin-6-yl] -2S-tert- Synthesis of butyldimethylsilanyloxypropan-1-one)

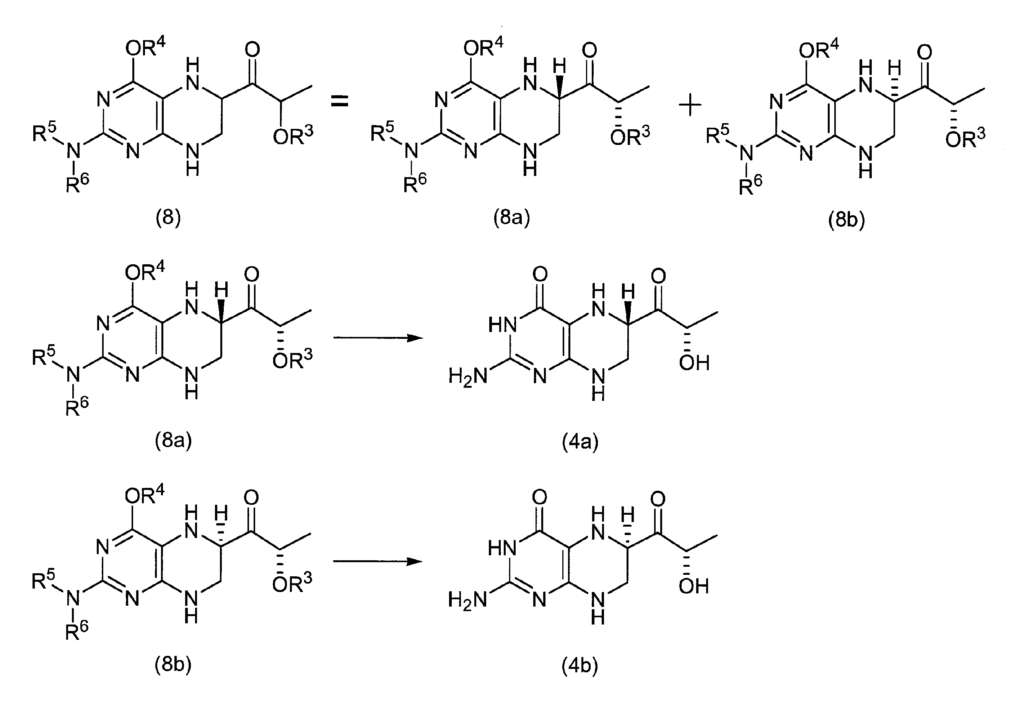
1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one 100 mg (0. 15 mmol), 10 mL of ethyl acetate, 20 mg of 10% Pd—C and 156 mg (1.54 mmol) of triethylamine were added, and the hydrogenation reaction was carried out for 1 hour at an external temperature of 50 ° C. under normal pressure (H 2 balloon). After the catalyst was filtered off, the reaction solution was concentrated under reduced pressure, and the crude product was separated and purified by flash chromatography to give 1- [4-tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino. -5,6,7,8-tetrahydropteridin-6S-yl] -2S-tert-butyldimethylsilanyloxypropan-1-one (8a) 30 mg (0.045 mmol, 30% yield) and 1- [4 -Tert-butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-5,6,7,8-tetrahydropteridin-6R-yl] -2S-tert-butyldimethylsilanyloxypropane-1 -30 mg (0.045 mmol, 30% yield) of ONE (8b) was obtained.
(8a)
1 H NMR (DMSO-d 6 ): δ / ppm = 0.08 (s, 3H), 0.09 (s, 3H), 0.89 (s, 9H), 1.21 (d, 3H, J = 6.6Hz), 1.37 (s , 18H), 1.49 (s, 9H), 3.56-3.67 (m, 2H), 4.39 (m, 1H), 4.42 (q, 1H, J = 6.6Hz), 4.79 (s, 1H), 7.00 (s, 1H)
(8b)
1 H NMR (DMSO-d 6 ): δ / ppm = 0.08 (s, 3H), 0.09 (s, 3H), 0.89 (s, 9H), 1.23 (d, 3H, J = 6.6Hz), 1.37 (s , 18H), 1.49 (s, 9H), 3.40-3.53 (m, 2H), 4.35 (m, 1H), 4.44 (q, 1H, J = 6.6Hz), 4.93 (s, 1H), 7.09 (s, 1H)
Example 32 (1- [4-cyclohexyloxy-2- (N, N-di-tert-butoxycarbonyl) amino-5,6,7,8-tetrahydropteridin-6-yl] -2S-methoxymethoxypropane- Synthesis of 1-one)

From Example 1 from 200 mg (0.36 mmol) of 1- [4-cyclohexyloxy-2- (N, N-di-tert-butoxycarbonyl) aminopteridin-6-yl] -2S-methoxymethoxypropan-1-one In a similar manner, 1- [4-cyclohexyloxy-2- (N, N-di-tert-butoxycarbonyl) amino-5,6,7,8-tetrahydropteridin-6-yl] -2S-methoxymethoxypropane- 76 mg (0.13 mmol, yield = 38%) of 1-one was obtained.
1 H NMR (DMSO-d 6 ): δ / ppm = 1.21 (d, 3H, J = 6.9Hz), 1.32-1.37 (m, 3H), 1.38 (s, 18H), 1.43-1.51 (m, 3H) , 1.73 (m, 2H), 1.89-1.91 (m, 2H), 3.27 (s, 3H), 3.51-3.56 (m, 2H), 4.33-4.35 (m, 1H), 4.41 (q, 1H, J = 6.9Hz), 4.59 (d, 1H, J = 6.9Hz), 4.67 (d, 1H, J = 6.9Hz), 4.86-4.89 (m, 1H), 4.95 (d, 1H, J = 2.7Hz), 7.08 (s, 1H)
1 H NMR (DMSO-d 6 ): δ / ppm = 1.24 (d, 3H, J = 6.9Hz), 1.32-1.37 (m, 3H), 1.38 (s, 18H), 1.43-1.51 (m, 3H) , 1.73 (m, 2H), 1.89-1.91 (m, 2H), 3.32 (s, 3H), 3.51-3.56 (m, 2H), 4.33-4.35 (m, 1H), 4.39 (q, 1H, J = 6.9Hz), 4.59 (d, 1H, J = 6.9Hz), 4.67 (d, 1H, J = 6.9Hz), 4.86-4.89 (m, 1H), 5.01 (d, 1H, J = 2.4Hz), 7.08 (s, 1H)
Example 33 Synthesis of 2-amino-6- (2S-hydroxypropionyl) -7,8-dihydro-3H-pteridin-4-one (S-sepiapterin)

1- [4-tert-Butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-7,8-dihydropteridin-6-yl] -2S-tert-butyldimethylsilanyloxypropane-1 -To 300 mg (0.46 mmol) of ON was added 3 mL of acetonitrile and 6 mL of 2 mol / L hydrochloric acid, and the mixture was stirred at an external temperature of 40 ° C for 3 hours. The reaction solution was adjusted to pH = 7 with an aqueous sodium hydroxide solution, and the crystals were collected by filtration and dried under reduced pressure to obtain 96 mg (0.40 mmol, yield 88%) of S-sepiapterin. It was confirmed that the obtained compound was consistent with the spectral data described in Example 4.
Example 34 (Synthesis of 2-amino-6R- (2S-hydroxypropionyl) -5,6,7,8-tetrahydro-3H-pteridin-4-one (6R-S-tetrahydrolactoylpterin) dihydrochloride)

1- [4-tert-Butoxycarbonyl-2- (N, N-di-tert-butoxycarbonyl) amino-5,6,7,8-tetrahydropteridin-6R-yl] -2S-tert-butyldimethylsilanyl To 393 mg (0.60 mmol) of oxypropan-1-one (8b) was added 10 mL of concentrated hydrochloric acid, and the mixture was concentrated under reduced pressure. Ethanol was added to the concentrate, and the crystals were collected by filtration and dried under reduced pressure to obtain 106 mg (0.34 mmol, yield 56%) of 6R—S-tetrahydrolactoylpterin dihydrochloride (4b). The compound obtained agreed with the spectral data described in Example 8.



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
Medical uses
Sepiapterin is indicated for the treatment of hyperphenylalaninemia in people with phenylketonuria.[1][2]
Side effects
The most common side effects are upper respiratory tract infection, headache, diarrhea, abdominal pain, hyperphenylalaninemia and discoloration of feces.[2]
Society and culture
Legal status
In April 2025, the Committee for Medicinal Products for Human Use of the European Medicines Agency adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Sephience, intended for the treatment of hyperphenylalaninemia in adults and children with phenylketonuria.[2] The applicant for this medicinal product is PTC Therapeutics International Limited.[2] Sepiapterin was authorized for medical use in the European Union in June 2025.[2][3]
Sepiapterin was approved for medical use in the United States in July 2025.[1]
Research
Deficiency of tetrahydrobiopterin can cause toxic buildup of phenylalanine (phenylketonuria) as well as deficiencies of dopamine, norepinephrine, and epinephrine, leading to dystonia and other neurological illnesses. This has led to clinical study of sepiapterin in humans to treat tetrahydrobiopterin deficiency.[4]
Since atherosclerosis and other circulatory diseases associated with diabetes are also associated with tetrahydrobiopterin deficiency, animal studies of the value of sepiaterin in these vascular diseases have been done. These studies show that relaxation of the blood vessels studied was impaired after animals were given sepiapterin, even though their levels of tetrahydrobiopterin were replenished.[5]
References
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/219666s000lbl.pdf
- “Sephience EPAR”. European Medicines Agency (EMA). 25 April 2025. Retrieved 2 May 2025. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- “Sephience Product information”. Union Register of medicinal products. 25 June 2025. Retrieved 27 June 2025.
- Smith N, Longo N, Levert K, Hyland K, Blau N (April 2019). “Phase I clinical evaluation of CNSA-001 (sepiapterin), a novel pharmacological treatment for phenylketonuria and tetrahydrobiopterin deficiencies, in healthy volunteers”. Molecular Genetics and Metabolism. 126 (4): 406–412. doi:10.1016/j.ymgme.2019.02.001. ISSN 1096-7192. PMID 30922814. S2CID 85564348.
- Vasquez-Vivar J, Duquiane D, Whitsett J, Kalyanaraman B, Rajagopalan S (October 2002). “Altered Tetrahydrobiopterin Metabolism in Atherosclerosis”. Arteriosclerosis, Thrombosis, and Vascular Biology. 22 (10): 1655–1661. doi:10.1161/01.ATV.0000029122.79665.D9. PMID 12377745.
| Names | |
|---|---|
| IUPAC name2-amino-6-[(2S)-2-hydroxypropanoyl]-7,8-dihydro-1H-pteridin-4-one | |
| Other namesSephience | |
| Identifiers | |
| CAS Number | 17094-01-8 |
| 3D model (JSmol) | Interactive image |
| ChEMBL | ChEMBL1255653 |
| ChemSpider | 58746 |
| KEGG | C00835 |
| PubChem CID | 65253 |
| UNII | CJQ26KO7HP |
| InChI | |
| SMILES | |
| Properties | |
| Chemical formula | C9H11N5O3 |
| Molar mass | 237.22 g/mol |
| Pharmacology | |
| ATC code | None |
| Routes of administration | By mouth |
| Legal status | US: ℞-only[1]EU: Rx-only[2][3] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
- [1]. Pannirselvam M, et al. Chronic oral supplementation with sepiapterin prevents endothelial dysfunction and oxidative stress in small mesenteric arteries from diabetic (db/db) mice. Br J Pharmacol. 2003;140(4):701‐706. [Content Brief][2]. Cho YR, et al. Sepiapterin inhibits cell proliferation and migration of ovarian cancer cells via down-regulation of p70S6K-dependent VEGFR-2 expression. Oncol Rep. 2011;26(4):861‐867. [Content Brief]
//////////Sepiapterin, approvals 2025, fda 2025, Sephience, Sepiapterine, CNSA 001, CJQ26KO7HP, PTC 923, WHO 11848,














