
Tucatinib
ツカチニブ;
N6-(4,4-dimethyl-4,5-dihydro-1,3-oxazol-2-yl)-N4-(3-methyl-4-{[1,2,4]triazolo[1,5-a]pyridin-7-yloxy}phenyl)quinazoline-4,6-diamine
| Formula |
C26H24N8O2
|
|---|---|
| CAS |
937263-43-9
|
| Mol weight |
480.5212
|
To treat advanced unresectable or metastatic HER2-positive breast cancer
Drug Trials Snapshot
FDA APPROVED 4/17/2020 Tukysa
- ARRY 380
- ARRY-380
- ONT 380
- ONT-380
Tucatinib (INN),[1] sold under the brand name Tukysa, is a small molecule inhibitor of HER2 for the treatment of HER2-positive breast cancer.[2][3] It was developed by Array BioPharma and licensed to Cascadian Therapeutics (formerly Oncothyreon, subsequently part of Seattle Genetics).[4]
Common side effects are diarrhea, palmar-plantar erythrodysesthesia (burning or tingling discomfort in the hands and feet), nausea, fatigue, hepatotoxicity (liver damage), vomiting, stomatitis (inflammation of the mouth and lips), decreased appetite, abdominal pain, headache, anemia and rash.[5][6] Pregnant or breastfeeding women should not take Tucatinib because it may cause harm to a developing fetus or newborn baby.[5]
Tucatinib was approved for medical use in Australia in August 2020.[7]
Medical uses
Tucatinib is a kinase inhibitor indicated in combination with trastuzumab and capecitabine for treatment of adults with advanced unresectable or metastatic HER2-positive breast cancer, including those with brain metastases, who have received one or more prior anti-HER2-based regimens in the metastatic setting.[8]
Clinical trials
Two early stage clinical trials have reported encouraging results, both of which had options to enroll subjects with central nervous system (CNS) metastases.[2][9][10][11][12][10] HER2CLIMB is a Phase 2 randomized, double-blinded, placebo-controlled study of tucatinib in combination with trastuzumab and capecitabine in patients with pretreated, unresectable locally advanced or metastatic HER2-positive breast cancer.[13]
History
In April 2020, the U.S. Food and Drug Administration (FDA) approved tucatinib in combination with chemotherapy (trastuzumab and capecitabine) for the treatment of adults with advanced forms of HER2-positive breast cancer that can’t be removed with surgery, or has spread to other parts of the body, including the brain, and who have received one or more prior treatments.[5][6][14]
The FDA collaborated with the Australian Therapeutic Goods Administration (TGA), Health Canada, Health Sciences Authority (HSA, Singapore) and Swissmedic (SMC, Switzerland) on the review.[5] This was the first Project Orbis partnership between the FDA, HSA and Swissmedic.[5] As of 17 April 2020, the application is still under review at the other agencies.[5]
Tucatinib is a kinase inhibitor meaning it blocks a type of enzyme (kinase) and helps prevent the cancer cells from growing.[5] Tucatinib is approved for treatment after adults have taken one or more anti-HER2-based regimens in the metastatic setting.[5] The FDA approved tucatinib based on the results of the HER2CLIMB trial (NCT02614794) enrolling 612 subjects who had HER2-positive advanced unresectable or metastatic breast cancer and had prior treatment with trastuzumab, pertuzumab and ado-trastuzumab emtansine (T-DM1).[5][6] Subjects with previously treated and stable brain metastases, as well as those with previously treated and growing or untreated brain metastases, were eligible for the clinical trial, and 48% of enrolled subjects had brain metastases at the start of the trial.[5]
Subjects received either tucatinib 300 mg twice daily plus trastuzumab and capecitabine (tucatinib arm, n=410) or placebo plus trastuzumab and capecitabine (control arm, n=202).[6] The primary endpoint was progression-free survival (PFS), or the amount of time when there was no growth of the tumor, assessed by a blinded independent central review, evaluated in the initial 480 randomized patients.[5][6] The median PFS in subjects who received tucatinib, trastuzumab, and capecitabine was 7.8 months (95% CI: 7.5, 9.6) compared to 5.6 months (95% CI: 4.2, 7.1) in those subjects who received placebo, trastuzumab, and capecitabine (HR 0.54; 95% CI: 0.42, 0.71; p<0.00001).[5][6] Overall survival and PFS in subjects with brain metastases at baseline were key secondary endpoints.[5] The median overall survival in subjects who received tucatinib, trastuzumab, and capecitabine was 21.9 months (95% CI: 18.3, 31.0) compared to 17.4 months (95% CI: 13.6, 19.9) in subjects who received placebo, trastuzumab, and capecitabine (HR: 0.66; 95% CI: 0.50, 0.87; p=0.00480).[5][6] The median PFS in subjects with brain metastases at baseline who received tucatinib, trastuzumab and capecitabine was 7.6 months (95% CI: 6.2, 9.5) compared to 5.4 months (95% CI: 4.1, 5.7) in subjects who received placebo, trastuzumab and capecitabine (HR: 0.48; 0.34, 0.69; p<0.00001).[5][6]
The FDA granted the application for tucatinib priority review, breakthrough therapy, fast track, and orphan drug designations.[5][6][15] The FDA granted approval of Tukysa to Seattle Genetics, Inc.[5]
SYN
Recently, the Mao team reported a new route for the efficient synthesis of Tucatinib.
The results were published on Synthesis (DOI: 10.1055/s-0037-1610706).
Previously, the synthesis report route of Tucatinib was published by Array BioPharma in a patent document (WO 2007059257, 2007). The synthetic route reported in the patent is shown in the figure below:
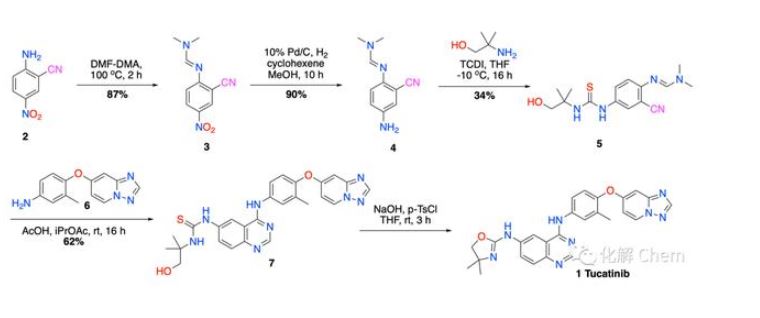
Using 4-nitro-2-cyanoaniline as the raw material, the first step is to condense with DMF-DMA to prepare imine 3 (yield 87%); subsequent catalytic hydrogenation of palladium on carbon to reduce the nitro group to obtain the amine 4 (90% yield); followed by 1,1&39;-thiocarbonyldiimidazole (TCDI) and The amino alcohol undergoes condensation to prepare the thiourea derivative 5 (yield is only 34%); further with the intermediate 6 to undergo ring-closure reaction to obtain the key intermediate 7 (yield 62%) ; Finally, under the action of p-toluenesulfonic acid, intramolecular dehydration and ring closure to form oxazoline, complete the synthesis of the target compound tucatinib.
Reverse synthesis analysis
The author broke the bond of Tucatinib from two points a and b and split them into three fragments. : Thioether oxazoline 17, nitrobenzene 3 and the key fragment of the original research route 6.
Preparation of key fragment 6
4-nitro-3-methylphenol 8 as a starting point The material, with pyridine derivative 9, undergoes aromatic affinity substitution reaction to prepare aryl ether 10 (yield 64%); then it is condensed with DMF-DMA, and then treated with hydroxylamine hydrochloride. The step yield was 81% to obtain the oxime derivative 12; subsequently, the ring was closed under the treatment of trifluoroacetic anhydride, the mostAfter palladium-catalyzed hydrogenation to reduce the nitro group, the key aniline triazole 6 was successfully prepared, with a total yield of 32.8%.
aromatic ring skeleton construction
fragment 3 was synthesized according to the method reported in the literature. The estimated aromatic ring fragment was then constructed with the aniline triazole 6 prepared above:
Compound 6 and fragment 3 were cyclized in acetic acid , 14 was successfully prepared, and finally the nitro group was reduced by palladium-catalyzed hydrogenation to obtain the key arylamine 15 with a two-step yield of 76.4%.
Fragment 17 and Tucatinib synthesis
amino alcohol and 1,1&39;-thiocarbonyl diimidazole (TCDI) The ring is closed to obtain 16, which is then treated with methyl trifluoromethanesulfonate to obtain oxazoline 17, with a total yield of 67.23% in the two steps.
oxazoline17 and arylamine 15 in the presence of cesium carbonate, heated in DMF for 20 hours, and finally completed the synthesis of Tucatinib with a yield of 76%.
Comparison of the new route and the patent route
The yield of the last step of the patent is unknown, starting with key intermediates 3 and 6, total income The rate is less than 19%.
The overview of the new route is as follows:
Correspondingly, starting from the intermediate 3 and 6, the total yield of the new route There is a significant improvement to 39%. Moreover, the purity of the product and other aspects also meet the requirements of API.
Comment
Tucatinib (Tukysa) Tucatinib/Tucatinib as a small-molecule oral tyrosine kinase (TKI) inhibitor for HER2 Positive breast cancer has highly specific targeting selectivity. The study of the new synthetic route
effectively improves the production efficiency in terms of ensuring the purity of the compound, and the raw materials used are relatively simple and easy to obtain.
Medicinal chemists have completed the research and development and synthesis of compounds (from 0 to 1), while process chemists have optimized the synthetic routes and processes, so that the compounds can be prepared more simply, efficiently, economically and environmentally.
SYN PATENT
CN 111825604
PAPER
Synthesis (2019), 51(13), 2660-2664
Abstract
A new and improved synthetic route to tucatinib is described that involves three key intermediates. The first of these, 4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylaniline, was prepared on a 100 g scale in 33% yield over five steps and 99% purity. Next, N 4-(4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylphenyl)quinazoline-4,6-diamine was isolated in 67% yield over three steps and >99% purity. Then, 4,4-dimethyl-2-(methylthio)-4,5-dihydrooxazole trifluoromethanesulfonate was prepared under mild conditions in 67% yield over two steps. Finally, tucatinib was obtained in 17% yield over nine steps and in >99% purity (HPLC). Purification methods used to isolate the product and the intermediates involved in the route are also reported.

References
- ^ World Health Organization (2016). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 75”. WHO Drug Information. 30 (1): 161. hdl:10665/331046.
- ^ Jump up to:a b “ONT-380 Active Against CNS Mets in HER2-Positive Breast Cancer”. Cancer Network. 15 December 2015. Retrieved 17 April 2020.
- ^ Martin M, López-Tarruella S (October 2018). “Emerging Therapeutic Options for HER2-Positive Breast Cancer”. American Society of Clinical Oncology Educational Book. American Society of Clinical Oncology. Annual Meeting. 35 (36): e64–70. doi:10.1200/EDBK_159167. PMID 27249772.
- ^ “Tucatinib” (PDF). Statement on a Nonproprietary Name Adopted by the USAN Council.
- ^ Jump up to:a b c d e f g h i j k l m n o p q “FDA Approves First New Drug Under International Collaboration, A Treatment Option for Patients with HER2-Positive Metastatic Breast Cancer”. U.S. Food and Drug Administration (FDA) (Press release). 17 April 2020. Retrieved 17 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g h i “FDA approves tucatinib for patients with HER2-positive metastatic brea”. U.S. Food and Drug Administration (FDA). 17 April 2020. Retrieved 20 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Tukysa”. Therapeutic Goods Administration (TGA). 21 August 2020. Retrieved 22 September 2020.
- ^ “Tukysa (tucatinib) tablets, for oral use” (PDF). Seattle Genetics. Retrieved 17 April2020.
- ^ “Oncothyreon Inc. Announces Data For ONT-380 In HER2-Positive Breast Cancer Patients With And Without Brain Metastases At The San Antonio Breast Cancer Symposium”. BioSpace (Press release). 9 December 2015. Retrieved 18 April 2020.
- ^ Jump up to:a b Borges VF, Ferrario C, Aucoin N, Falkson CI, Khan QJ, Krop IE, et al. “Efficacy results of a phase 1b study of ONT-380, a CNS-penetrant TKI, in combination with T-DM1 in HER2+ metastatic breast cancer (MBC), including patients (pts) with brain metastases”. Journal of Clinical Oncology. 2016 ASCO Annual Meeting.
- ^ “SABCS15: Promising phase 1 results lead to phase 2 for ONT-380 in HER2+ breast cancer”. Colorado Cancer Blogs. Retrieved 10 June 2016.
- ^ “A Study of Tucatinib (ONT-380) Combined With Capecitabine and/or Trastuzumab in Patients With HER2+ Metastatic Breast Cancer”. ClinicalTrials.gov. 31 December 2013. Retrieved 18 April 2020.
- ^ “A Study of Tucatinib vs. Placebo in Combination With Capecitabine & Trastuzumab in Patients With Advanced HER2+ Breast Cancer (HER2CLIMB)”. ClinicalTrials.gov. Retrieved 18 April 2020.
- ^ “Tukysa: FDA-Approved Drugs”. U.S. Food and Drug Administration (FDA). Retrieved 20 April 2020.
- ^ “Tucatinib Orphan Drug Designation and Approval”. U.S. Food and Drug Administration(FDA). 24 December 1999. Retrieved 20 April 2020.
External links
- “Tucatinib”. Drug Information Portal. U.S. National Library of Medicine.
- “Tucatinib”. National Cancer Institute.
- Clinical trial number NCT02614794 for “A Study of Tucatinib vs. Placebo in Combination With Capecitabine & Trastuzumab in Patients With Advanced HER2+ Breast Cancer (HER2CLIMB)” at ClinicalTrials.gov
 |
|
| Clinical data | |
|---|---|
| Trade names | Tukysa |
| Other names | ONT-380, ARRY-380 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620032 |
| License data | |
| Pregnancy category |
|
| Routes of administration |
By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C26H24N8O2 |
| Molar mass | 480.532 g·mol−1 |
| 3D model (JSmol) | |
| NAME | DOSAGE | STRENGTH | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Tukysa | Tablet | 150 mg/1 | Oral | Seattle Genetics, Inc. | 2020-04-17 | Not applicable | ||
| Tukysa | Tablet | 150 mg | Oral | Seattle Genetics, Inc. | 2020-08-27 | Not applicable | ||
| Tukysa | Tablet | 50 mg/1 | Oral | Seattle Genetics, Inc. | 2020-04-17 | Not applicable | ||
| Tukysa | Tablet | 50 mg | Oral | Seattle Genetics, Inc. | 2020-10-08 | Not applicable |
///////tucatinib, FDA 2020, TUKSYA, 2020 APROVALS, ARRY 380, ONT 380, ツカチニブ ,














