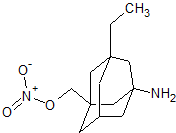
1-Amino-3-ethyl-5-nitratemethyladamantane hydrochloride (MN-05)
Med. Chem. Commun., 2017, 8,135-147
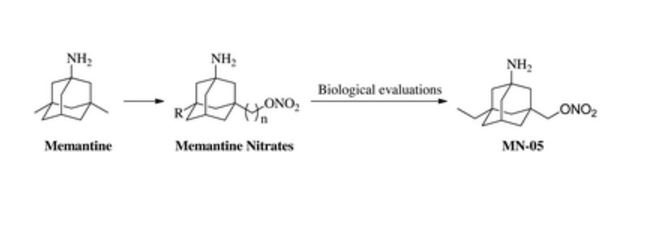
A series of memantine nitrate derivatives, as dual functional compounds with neuroprotective and vasodilatory activity for neurodegenerative diseases, was designed and synthesized
Synthesis and biological evaluation of memantine nitrates as a potential treatment for neurodegenerative diseases
E-mail: yxy0723@163.com, zaijunzhang@163.com
Fax: +86 20 8522 4766
Tel: +86 20 8522 5030
A series of memantine nitrate derivatives, as dual functional compounds with neuroprotective and vasodilatory activity for neurodegenerative diseases, was designed and synthesized. These compounds combined the memantine skeleton and a nitrate moiety, and thus inhibited the N-methyl-D-aspartic acid receptor and released NO in the central nervous system. The biological evaluation results revealed that the new memantine nitrates were effective in protecting neurons against glutamate-induced injury in vitro. Moreover, memantine nitrates dilated aortic rings against phenylephrine-induced contraction. The structure–activity relationships of neuroprotection and vasodilation were both analyzed. In further studies, compound MN-05 significantly protected cortical neurons by inhibiting Ca2+ influx, reducing free radical production and maintaining the mitochondrial membrane potential. Further research on MN-05 is warranted.
1-Amino-3-ethyl-5-nitratemethyladamantane hydrochloride (MN-05).
Compound MN- 05 was synthesized using a similar method to that as described for synthesis of compound MN-01 from compound 16. White solid, 65.5% yield. ESI-MS: m/z 255.1 [M + H]+ .
1H NMR (300 MHz, DMSO-d6) δ 0.75-0.80 (t, J = 7.5 Hz, 3H, CH3), 1.16-1.24 (q, J = 7.5 Hz, 2H, CH2), 1.24-1.25 (m, 2H), 1.30-1.39 (m, 2H), 1.43 (s, 2H), 1.45-1.57 (dd, J = 12 Hz, 6 Hz, 2H), 1.57-1.63 (dd, J = 12 Hz, 6 Hz, 2H), 1.71 (s, 2H), 2.23 (m, 1H, CH), 4.30 (s, 2H, CH2O), 8.21 (s, 3H, NH2HCl).
13C NMR (75 MHz, DMSO-d6) δ 7.4, 28.6, 34.5, 35.0, 36.8, 40.3, 41.6, 43.9, 52.3, 80.9. Anal. Calcd for C13H23N2O3Cl·0.3 H2O: C, 52.72%; H, 8.03%; N, 9.46%. Found: C, 52.72%; H, 7.92%; N, 9.51%
“ALL FOR DRUGS” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This article is a compilation for educational purposes only.
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent