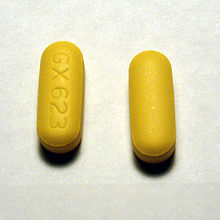
Abacavir (ABC) is a medication used to prevent and treat HIV/AIDS.[1][2] Similar to other nucleoside analog reverse-transcriptase inhibitors (NRTIs), abacavir is used together with other HIV medications, and is not recommended by itself.[3] It is taken by mouth as a tablet or solution and may be used in children over the age of three months.[1][4]
Abacavir is generally well tolerated.[4] Common side effects include vomiting, trouble sleeping, fever, and feeling tired.[1] More severe side effects include hypersensitivity, liver damage, and lactic acidosis.[1] Genetic testing can indicate whether a person is at higher risk of developing hypersensitivity.[1] Symptoms of hypersensitivity include rash, vomiting, and shortness of breath.[4] Abacavir is in the NRTI class of medications, which work by blocking reverse transcriptase, an enzyme needed for HIV virus replication.[5] Within the NRTI class, abacavir is a carbocyclic nucleoside.[1]
Abacavir was patented in 1988 and approved for use in the United States in 1998.[6][7] It is on the World Health Organization’s List of Essential Medicines, the most effective and safe medicines needed in a health system.[8] It is available as a generic medication.[1] The wholesale cost in the developing world as of 2014 is between US$0.36 and US$0.83 per day.[9] As of 2016 the wholesale cost for a typical month of medication in the United States is US$70.50.[10] Commonly, abacavir is sold together with other HIV medications, such as abacavir/lamivudine/zidovudine, abacavir/dolutegravir/lamivudine, and abacavir/lamivudine.[4][5]
Medical uses
Abacavir tablets and oral solution, in combination with other antiretroviral agents, are indicated for the treatment of HIV-1 infection.
Abacavir should always be used in combination with other antiretroviral agents. Abacavir should not be added as a single agent when antiretroviral regimens are changed due to loss of virologic response.
Side effects
Common adverse reactions include nausea, headache, fatigue, vomiting, diarrhea, loss of appetite and trouble sleeping. Rare but serious side effects include hypersensitivity reaction or rash, elevated AST and ALT, depression, anxiety, fever/chills, URI, lactic acidosis, hypertriglyceridemia, and lipodystrophy.[11]
People with liver disease should be cautious about using abacavir because it can aggravate the condition. Signs of liver problems include nausea and vomiting, abdominal pain, dark-colored urine and yellowing of the skin or whites of the eyes. The use of nucleosidedrugs such as abacavir can very rarely cause lactic acidosis. Signs of lactic acidosis include fast or irregular heartbeat, unusual muscle pain, fatigue, difficulty breathing and stomach pain with nausea and vomiting.[12] Abacavir can also lead to immune reconstitution inflammatory syndrome, a change in body fat as well as an increased risk of heart attack.
Resistance to abacavir has developed in laboratory versions of HIV which are also resistant to other HIV-specific antiretrovirals such as lamivudine, didanosine, and zalcitabine. HIV strains that are resistant to protease inhibitors are not likely to be resistant to abacavir.
Abacavir is contraindicated for use in infants under 3 months of age.
Little is known about the effects of Abacavir overdose. Overdose victims should be taken to a hospital emergency room for treatment.
Hypersensitivity syndrome
Hypersensitivity to abacavir is strongly associated with a specific allele at the human leukocyte antigen B locus namely HLA-B*5701.[13][14][15] There is an association between the prevalence of HLA-B*5701 and ancestry. The prevalence of the allele is estimated to be 3.4 to 5.8 percent on average in populations of European ancestry, 17.6 percent in Indian Americans, 3.0 percent in Hispanic Americans, and 1.2 percent in Chinese Americans.[16][17] There is significant variability in the prevalence of HLA-B*5701 among African populations. In African Americans, the prevalence is estimated to be 1.0 percent on average, 0 percent in the Yorubafrom Nigeria, 3.3 percent in the Luhya from Kenya, and 13.6 percent in the Masai from Kenya, although the average values are derived from highly variable frequencies within sample groups.[18]
Common symptoms of abacavir hypersensitivity syndrome include fever, malaise, nausea, and diarrhea. Some patients may also develop a skin rash.[19] Symptoms of AHS typically manifest within six weeks of treatment using abacavir, although they may be confused with symptoms of HIV, immune reconstitution syndrome, hypersensitivity syndromes associated with other drugs, or infection.[20] The U.S. Food and Drug Administration (FDA) released an alert concerning abacavir and abacavir-containing medications on July 24, 2008,[21] and the FDA-approved drug label for abacavir recommends pre-therapy screening for the HLA-B*5701 allele and the use of alternative therapy in subjects with this allele.[22] Additionally, both the Clinical Pharmacogenetics Implementation Consortium and the Dutch Pharmacogenetics Working Group recommend use of an alternative therapy in individuals with the HLA-B*5701 allele.[23][24]
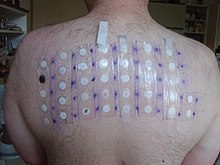
Skin-patch testing may also be used to determine whether an individual will experience a hypersensitivity reaction to abacavir, although some patients susceptible to developing AHS may not react to the patch test.[25]
The development of suspected hypersensitivity reactions to abacavir requires immediate and permanent discontinuation of abacavir therapy in all patients, including patients who do not possess the HLA-B*5701 allele. On March 1, 2011, the FDA informed the public about an ongoing safety review of abacavir and a possible increased risk of heart attack associated with the drug. A meta-analysis of 26 studies conducted by the FDA, however, did not find any association between abacavir use and heart attack [26][27]
Immunopathogenesis
The mechanism underlying abacavir hypersensitivity syndrome is related to the change in the HLA-B*5701 protein product. Abacavir binds with high specificity to the HLA-B*5701 protein, changing the shape and chemistry of the antigen-binding cleft. This results in a change in immunological tolerance and the subsequent activation of abacavir-specific cytotoxic T cells, which produce a systemic reaction known as abacavir hypersensitivity syndrome.[28]
Interaction
Abacavir, and in general NRTIs, do not undergo hepatic metabolism and therefore have very limited (to none) interaction with the CYP enzymes and drugs that effect these enzymes. That being said there are still few interactions that can affect the absorption or the availability of abacavir. Below are few of the common established drug and food interaction that can take place during abacavir co-administration:
- Protease inhibitors such as tipranavir or ritonovir may decrease the serum concentration of abacavir through induction of glucuronidation. Abacavir is metabolized by both alcohol dehydrogenase and glucuronidation.[29][30]
- Ethanol may result in increased levels of abacavir through the inhibition of alcohol dehydrogenase. Abacavir is metabolized by both alcohol dehydrogenase and glucuronidation.[29][31]
- Methadone may diminish the therapeutic effect of Abacavir. Abacavir may decrease the serum concentration of Methadone.[32][33]
- Orlistat may decrease the serum concentration of antiretroviral drugs. The mechanism of this interaction is not fully established but it is suspected that it is due to the decreased absorption of abacavir by orlistat.[34]
- Cabozantinib: Drugs from the MPR2 inhibitor (Multidrug resistance-associated protein 2 inhibitors) family such as abacavir could increase the serum concentration of Cabozantinib.[35]
Mechanism of action
Abacavir is a nucleoside reverse transcriptase inhibitor that inhibits viral replication. It is a guanosine analogue that is phosphorylated to carbovir triphosphate (CBV-TP). CBV-TP competes with the viral molecules and is incorporated into the viral DNA. Once CBV-TP is integrated into the viral DNA, transcription and HIV reverse transcriptase is inhibited.[36]
Pharmacokinetics
Abacavir is given orally and is rapidly absorbed with a high bioavailability of 83%. Solution and tablet have comparable concentrations and bioavailability. Abacavir can be taken with or without food.
Abacavir can cross the blood-brain barrier. Abacavir is metabolized primarily through the enzymes alcohol dehydrogenase and glucuronyl transferase to an inactive carboxylate and glucuronide metabolites. It has a half-life of approximately 1.5-2.0 hours. If a person has liver failure, abacavir’s half life is increased by 58%.
Abacavir is eliminated via excretion in the urine (83%) and feces (16%). It is unclear whether abacavir can be removed by hemodialysis or peritoneal dialysis.[36]
History
Robert Vince and Susan Daluge along with Mei Hua, a visiting scientist from China, developed the medication in the ’80s.[37][38][39]
Abacavir was approved by the Food and Drug Administration (FDA) on December 18, 1998, and is thus the fifteenth approved antiretroviral drug in the United States. Its patent expired in the United States on 2009-12-26.
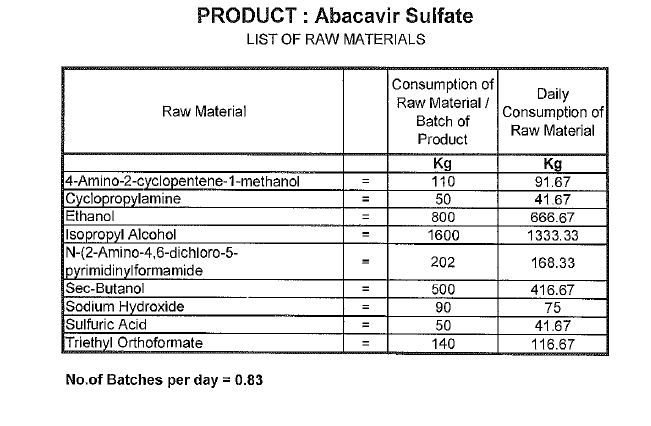
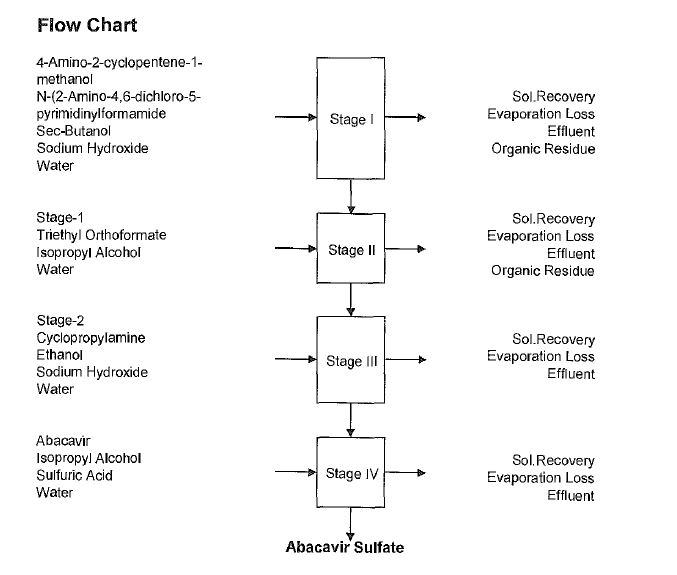
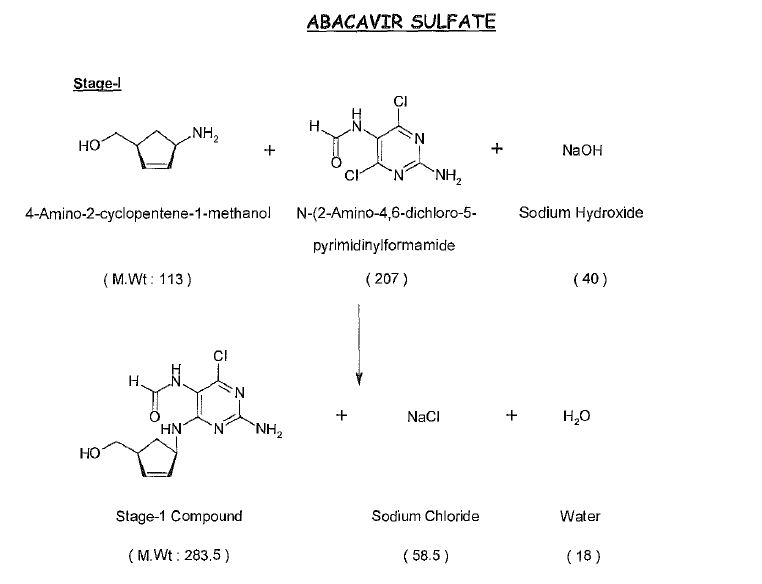
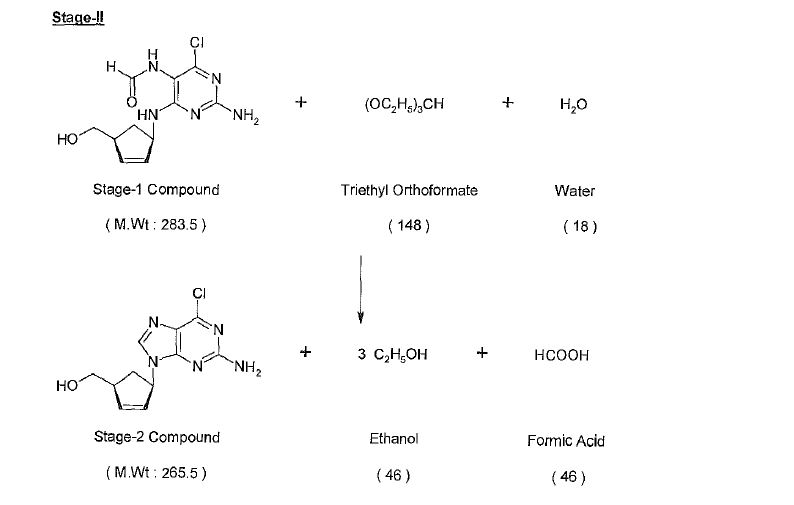
Synthesis
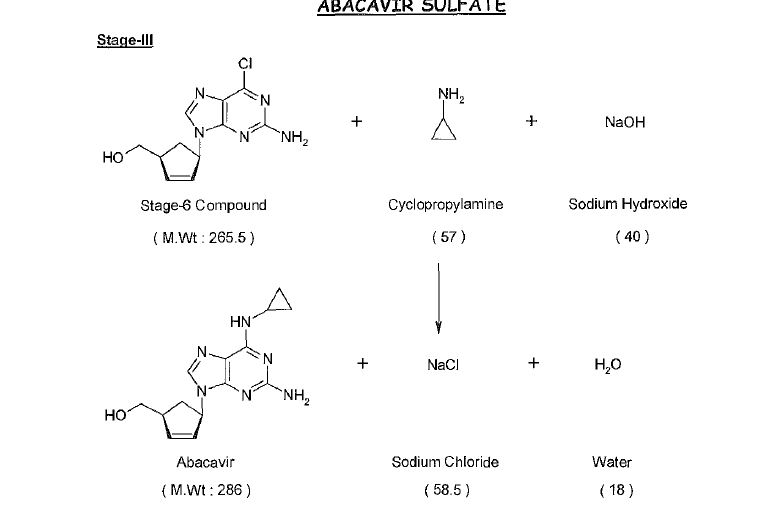
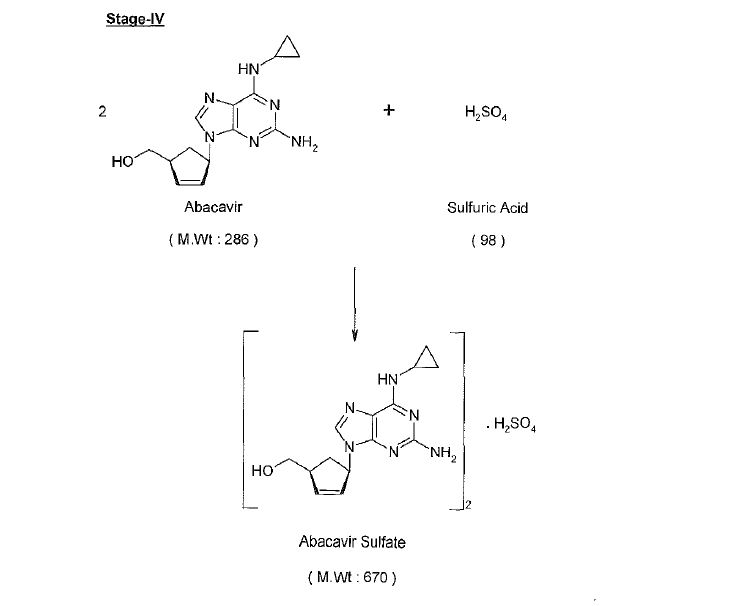
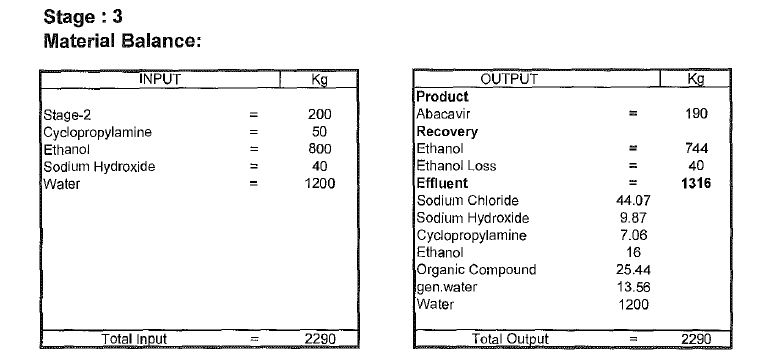
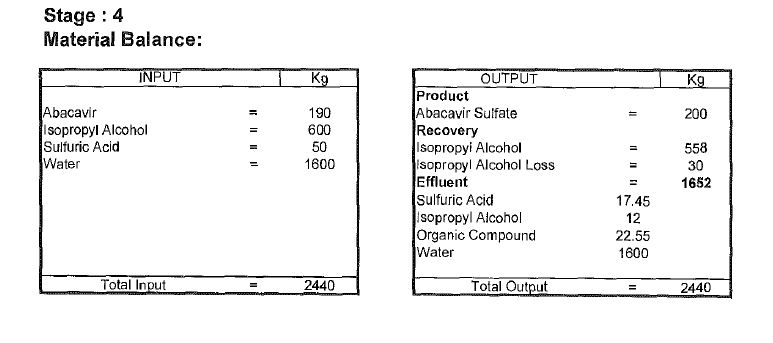
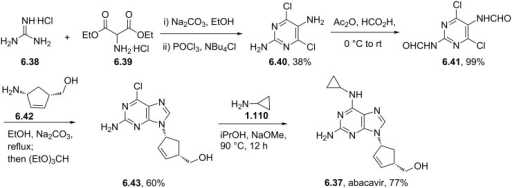
Abacavir synthesis:[40]
References



ABOVE From internet free resources
- Crimmins, M. T.; King, B. W. (1996). “An Efficient Asymmetric Approach to Carbocyclic Nucleosides: Asymmetric Synthesis of 1592U89, a Potent Inhibitor of HIV Reverse Transcriptase”. The Journal of Organic Chemistry. 61 (13): 4192–4193. doi:10.1021/jo960708p. PMID 11667311.


| AU 8937025; EP 0349242; JP 1990045486; JP 1999139976; US 5034394; US 5089500
|
SYN 2
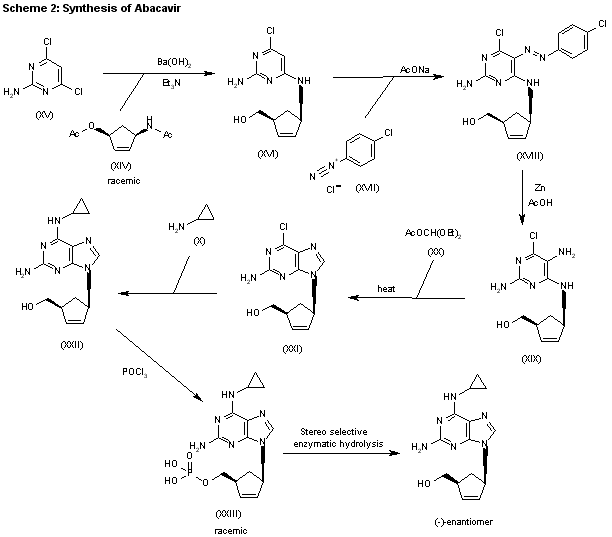
The condensation of (?-cis-4-acetamido-2-cyclopentenylmethyl acetate (XIV) with 2-amino-4,6-dichloropyrimidine (XV) by means of Ba(OH)2 and triethylamine in refluxing butanol gives the expected condensation product (XVI), which is treated with 4-chlorophenyldiazonium chloride (XVII) in water/acetic acid to yield the corresponding azo-compound (XVIII). The reduction of (XVIII) with Zn/acetic acid in ethanol affords the diamine (XIX), which is cyclized with refluxing diethoxymethyl acetate (XX) to afford the corresponding purine (XXI). The reaction of (XXI) with cyclopropylamine (X) in refluxing ethanol affords racemic abacavir (XXII), which is phosphorylated with POCl3 giving the racemic 4′-O-phosphate (XXIII). Finally, this compound is submitted to stereoselective enzymatic dephosphorylation using snake venom 5′-nucleotidase (EC 3.1.3.5) from Crotalus atrox yielding the (-)-enantiomer, abacavir.
SYN 3
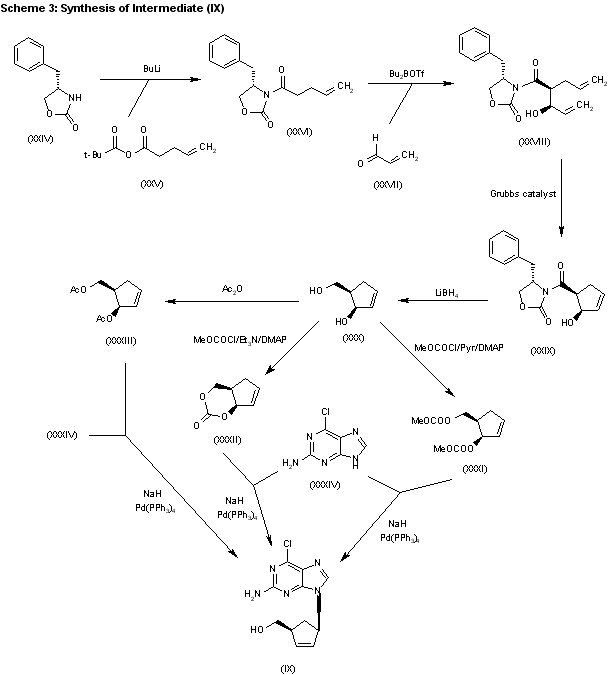
The acylation of 4(S)-benzyloxazolidin-2-one (XXIV) with 4-pentenoyl pivaloyl anhydride (XXV) by means of NaH in THF gives 4(S)-benzyl-3-(4-pentenoyl)oxazolidin-2-one (XXVI), which is submitted to a diastereoselective syn aldol condensation with acrolein (XXVII), using dibutylboron triflate as catalyst, affording the aldol (XXVIII). The cyclization of (XXVIII) by means of the Grubbs catalyst in dichloromethane yields the cyclopentenol (XXIX), which is reduced with LiBH4 in THF/methanol to give the key intermediate 5(R)-(hydroxymethyl)-2-cyclopenten-1(R)-ol (XXX). The reaction of (XXX) with methyl chloroformate/pyridine/DMAP or methyl chloroformate/triethylamine/DMAP or acetic anhydride gives the diols (XXXI), (XXXII) and (XXXIII), respectively, each of which coupled with 2-amino-6-chloropurine (XXXIV) in the presence of NaH and palladium tetrakis(triphenylphosphine) in THF/DMSO, affords the purine intermediate (IX) already reported.
SYN
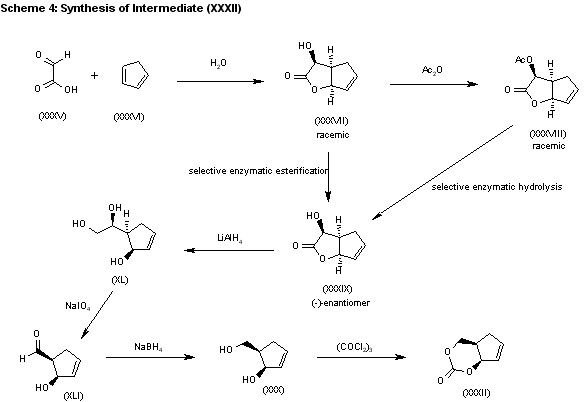
The water promoted condensation of glyoxylic acid (XXXV) with cyclopentadiene (XXXVI) gives the racemic cis-hydroxylactone (XXXVII), which is acetylated with acetic anhydride to the acetate (XXXVIII). The selective enzymatic hydrolysis of (XXXVIII) with Pseudomonas fluorescens lipase yields the pre (-)-enantiomer (XXXIX), which is reduced with LiAlH4 in refluxing THF, affording triol (XL). The oxidation of the vicinal glycol of (XL) with NaIO4 in ethyl ether/water yields the hydroxyaldehyde (XLI), which is reduced with NaBH4 in ethanol to give the key intermediate 5(R)-(hydroxymethyl)-2-cyclopenten-1(R)-ol (XXX). This compound, by reaction with triphosgene and triethylamine in dichloromethane, results in the cyclic carbonate intermediate (XXXII), already reported.
SYN
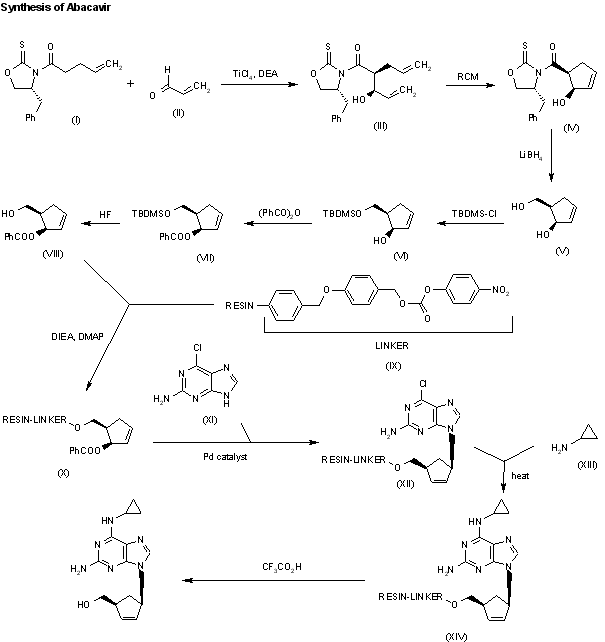
A new solid phase synthesis of abacavir has been reported: Condensation of the chiral 4(R)-benzyl-3-(4-pentenoyl)oxazolidin-2-thione (I) with acrolein (II) by means of TiCl4 and DIEA gives the adduct (III), which was transformed into the chiral cyclopentene (IV) by catalytic ring-closing metathesis. The reductive removal of the chiral auxiliary with LiBH4 affords the chiral diol (V), which is selectively silylated with TBDMSCl providing the primary silyl ether (VI). Acylation of the secondary alcohol of (VI) with benzoic anhydride gives the benzoate (VII), which is desilylated with HF in acetonitrile yielding the allylic benzoate (VIII). Benzoate (VIII) is condensed with a p-nitrophenyl Wang carbonate resin (IX) by means of DIEA and DMAP affording the solid phase resin (X) which is condensed with 2-amino-6-chloropurine (XI) by means of a Pd catalyst furnishing the adduct (XII). Thermal condensation of (XII) with cyclopropylamine (XIII) yields the diaminopurine resin (XIV) which, after cleavage from the resin by a treatment with TFA in dichloromethane, gives directly abacavir.
SYN
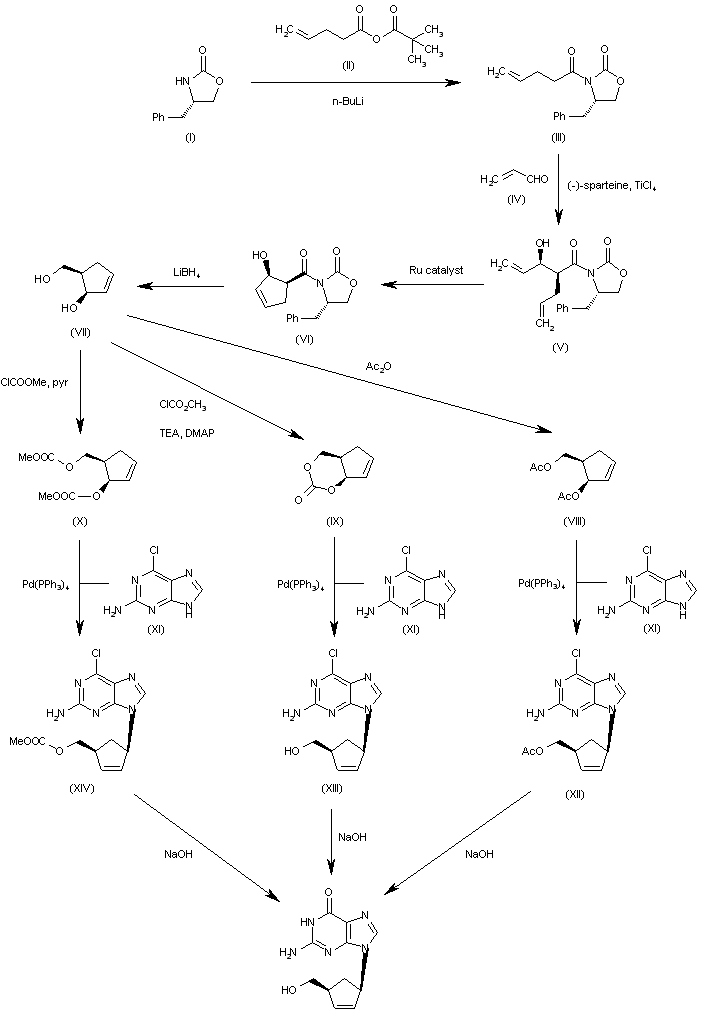 The condensation of the chiral oxazolidinone (I) with the pentenoic anhydride (II) by means of n-BuLi in THF gives the N-pentenoyloxazolidinone (III), which is condensed with acrolein (IV) catalyzed by TiCl4 and (-)-spartein in dichloromethane, yielding the chiral adduct (V). The ring-closing metathesis of (V) by means of a Ru catalyst in dichloromethane affords the chiral cyclopentenol derivative (VI), which is reduced to the (R,R)-5-(hydroxymethyl)-2-cyclopenten-1-ol (VII) by means of LiBH4 in THF. The reaction of diol (VII) with Ac2O; with methyl chloroformate, TEA and DMAP; or with ethyl chloroformate and pyridine gives the diacetate (VIII), the cyclic carbonate (IX) or the dicarbonate (X), respectively. The condensation of (VIII), (IX) or (X) with 2-amino-6-chloropurine (XI) by means of Pd(PPh3)4 yields the carbocyclic purines (XII), (XIII) or (XIV), respectively. Finally, these compounds are hydrolyzed with aqueous NaOH to the target carbocyclic guanine.
The condensation of the chiral oxazolidinone (I) with the pentenoic anhydride (II) by means of n-BuLi in THF gives the N-pentenoyloxazolidinone (III), which is condensed with acrolein (IV) catalyzed by TiCl4 and (-)-spartein in dichloromethane, yielding the chiral adduct (V). The ring-closing metathesis of (V) by means of a Ru catalyst in dichloromethane affords the chiral cyclopentenol derivative (VI), which is reduced to the (R,R)-5-(hydroxymethyl)-2-cyclopenten-1-ol (VII) by means of LiBH4 in THF. The reaction of diol (VII) with Ac2O; with methyl chloroformate, TEA and DMAP; or with ethyl chloroformate and pyridine gives the diacetate (VIII), the cyclic carbonate (IX) or the dicarbonate (X), respectively. The condensation of (VIII), (IX) or (X) with 2-amino-6-chloropurine (XI) by means of Pd(PPh3)4 yields the carbocyclic purines (XII), (XIII) or (XIV), respectively. Finally, these compounds are hydrolyzed with aqueous NaOH to the target carbocyclic guanine.
SYN
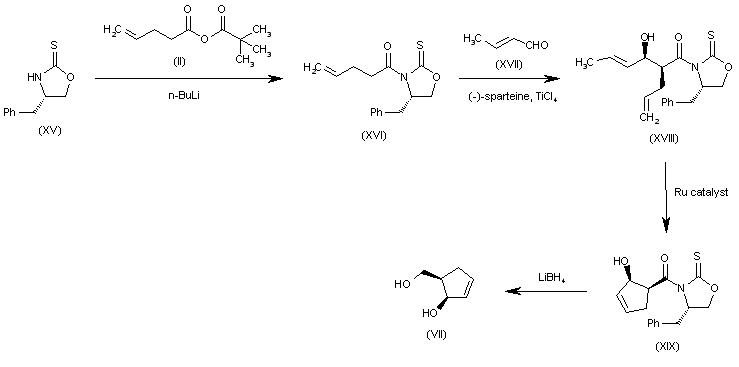
Alternatively, the (R,R)-5-(hydroxymethyl)-2-cyclopenten-1-ol (VII) can also be obtained as follows: The condensation of the chiral oxazolidinethione (XV) with the pentenoic anhydride (II) by means of n-BuLi in THF gives the N-pentenoyloxazolidinethione (XVI), which is condensed with crotonaldehyde (XVII) catalyzed by TiCl4 and (-)-spartein in dichloromethane, yielding the chiral adduct (XVIII). The ring-closing metathesis of (XVIII) by means of a Ru catalyst in dichloromethane affords the chiral cyclopentenol derivative (XIX), which is reduced to the target diol (VII) by means of LiBH4 in THF.
SYN
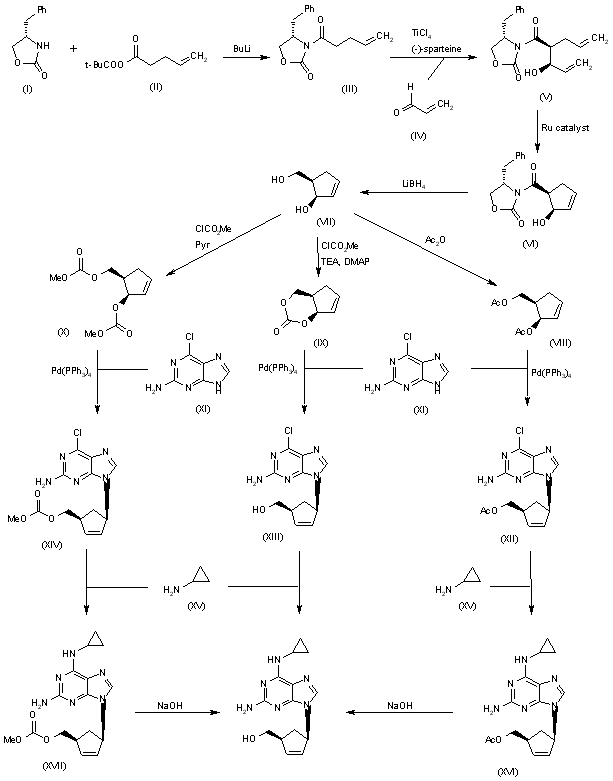
An efficient asymmetric synthesis of abacavir has been reported: Acylation of the chiral oxazolidinone (I) with the mixed anhydride (II) by means of BuLi in THF gives the N-pentenoyloxazolidinone (III), which by condensation with acrolein (IV) catalyzed by TiCl4 and (?-spartein in dichloromethane yields the chiral adduct (V). The ring-closing metathesis of adduct (V) by means of the ruthenium catalyst (Cy3P)Cl2Ru=CHPh in dichloromethane affords the chiral cyclopentenol (VI), which is reduced to 5(R)-(hydroxymethyl)-2-cyclopenten-1(R)-ol (VII) by means of LiBH4 in THF. Reaction of diol (VII) with a) Ac2O, TEA and DMAP, b) methyl chloroformate, TEA and DMAP or c) methyl chloroformate, pyridine and DMAP gives a) the diacetate (VIII), b) the cyclic carbonate (IX) or c) the dicarbonate (X), respectively. The condensation of diacetate (VIII), cyclic carbonate (IX) or dicarbonate (X) with 2-amino-6-chloropurine (XI) by means of Pd(PPh3)4 yields the carbocyclic purines (XII), (XIII) or (XIV), respectively. Treatment of these chloro-purines (XII), (XIII) and (XIV) with cyclopropylamine (XV) in hot DMSO provides the corresponding cyclopropylaminopurine carbonate (XVI), abacavir or cyclopropylaminopurine acetate (XVII), respectively. Finally, the protecting groups of purines (XVI) and (XVII) are hydrolyzed with aqueous NaOH.
SYN
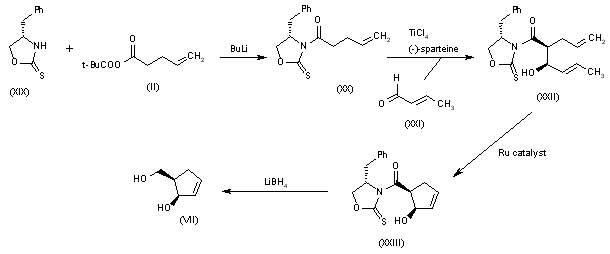
Alternatively, 5(R)-(hydroxymethyl)-2-cyclopenten-1(R)-ol (VII) can also be obtained as follows: Acylation of the chiral oxazolidinethione (XIX) with the mixed anhydride (II) by means of BuLi in THF gives the N-pentenoyl-oxazolidinethione (XX), which by condensation with crotonaldehyde (XXI) catalyzed by TiCl4 and (?-spartein in dichloromethane yields the chiral adduct (XXII). The ring-closing metathesis of (XXII) by means of the ruthenium catalyst in dichloromethane affords the chiral cyclopentenol derivative (XXIII), which is reduced to the target diol (VII) by means of LiBH4 in THF.
SYN
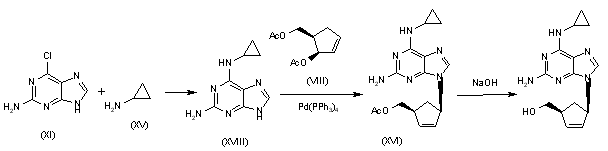
Alternatively, 2-amino-6-chloropurine (XI) is treated with cyclopropylamine (XV) in hot DMSO to give 2-amino-6-(cyclopropylamino)purine (XVIII), which is condensed with the chiral diacetate (VIII) by means of Pd(PPh3)4 to yield the carbocyclic purine acetate (XVI). Finally, purine (XVI) is deprotected by hydrolysis with aqueous NaOH.
CLIP

https://www.sciencedirect.com/science/article/pii/S0960894X15007581
CLIP
CLIP
CLIP
Production of Abacavir
030-8 1.0g (0.0053mol), in the reaction flask was added cesium carbonate 1.75 g (0.0054 mol) and dry DMSO 50ml, stirred under N2 protection, the temperature was raised to 60 °C and stirred at this temperature for 2 h the mixture wascooled to room temperature, then add tetrakis (triphenylphosphine) combined palladium (TTP) [0.85 (0.00074mol)] and compound 030-5 [0.79g (0.0034 mol), DMSO (10 ml) solution was stirred and heated to 65 °C held 65 °C and stirred reaction 2.25h. The you can get the mixture containing compounds 030-9.
To the mixture was added methanol 100ml and K2CO3 is 2.10g, the mixture reaction was stirred for 45min at 40 °C, a solid precipitate which was filtered through a Celite layer and the filtrate was evaporated to a small volume under vacuum at 90 °C, and the remaining gum pounding mill was extracted with dichloromethane (100ml * 2) to give a brown solid residue was purified by silica gel (Merck 9385) column chromatography [eluent: dichloromethane / methanol (volume ratio 9:1)] to give a yellow foam was 030 0.26 g, yield 26.8.

CLIP
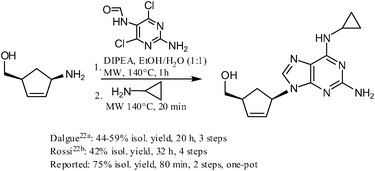
https://pubs.rsc.org/en/content/articlehtml/2012/ra/c2ra20842c
References
- ^ Jump up to:a b c d e f g h “Abacavir Sulfate”. The American Society of Health-System Pharmacists. Archived from the original on 8 September 2017. Retrieved 31 July 2015.
- ^ “Drug Name Abbreviations Adult and Adolescent ARV Guidelines”. AIDSinfo. Archived from the original on 2016-11-09. Retrieved 2016-11-08.
- ^ “What Not to Use Adult and Adolescent ARV Guidelines”. AIDSinfo. Archived from the original on 2016-11-09. Retrieved 2016-11-08.
- ^ Jump up to:a b c d Yuen, GJ; Weller, S; Pakes, GE (2008). “A review of the pharmacokinetics of abacavir”. Clinical Pharmacokinetics. 47 (6): 351–71. doi:10.2165/00003088-200847060-00001. PMID 18479171.
- ^ Jump up to:a b “Nucleoside reverse transcriptase inhibitors (NRTIs or ‘nukes’) – HIV/AIDS”. www.hiv.va.gov. Archived from the original on 2016-11-09. Retrieved 2016-11-08.
- ^ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 505. ISBN 9783527607495. Archived from the original on 2017-09-08.
- ^ Kane, Brigid M. (2008). HIV/AIDS Treatment Drugs. Infobase Publishing. p. 56. ISBN 9781438102078. Archived from the original on 2017-09-08.
- ^ “WHO Model List of Essential Medicines (19th List)” (PDF). World Health Organization. April 2015. Archived (PDF) from the original on 13 December 2016. Retrieved 8 December 2016.
- ^ “International Drug Price Indicator Guide”. ERC. Retrieved 20 November 2016.
- ^ “NADAC as of 2016-12-07 | Data.Medicaid.gov”. Centers for Medicare and Medicaid Services. Archived from the original on 21 December 2016. Retrieved 12 December 2016.
- ^ “Abacavir Adverse Reactions”. Epocrates Online.
- ^ “Abacavir | Dosage, Side Effects | AIDSinfo”. AIDSinfo. Archived from the original on 2017-03-06. Retrieved 2016-11-08.
- ^ Mallal, S., Phillips, E., Carosi, G.; et al. (2008). “HLA-B*5701 screening for hypersensitivity to abacavir”. New England Journal of Medicine. 358 (6): 568–579. doi:10.1056/nejmoa0706135. PMID 18256392.
- ^ Rauch, A., Nolan, D., Martin, A.; et al. (2006). “Prospective genetic screening decreases the incidence of abacavir hypersensitivity reactions in the Western Australian HIV cohort study”. Clinical Infectious Diseases. 43 (1): 99–102. doi:10.1086/504874. PMID 16758424.
- ^ Dean, Laura (2012), Pratt, Victoria; McLeod, Howard; Rubinstein, Wendy; Dean, Laura (eds.), “Abacavir Therapy and HLA-B*57:01 Genotype”, Medical Genetics Summaries, National Center for Biotechnology Information (US), PMID 28520363, retrieved 2019-01-14
- ^ Heatherington; et al. (2002). “Genetic variations in HLA-B region and hypersensitivity reactions to abacavir”. Lancet. 359 (9312): 1121–1122. doi:10.1016/s0140-6736(02)08158-8. PMID 11943262.
- ^ Mallal; et al. (2002). “Association between presence of HLA*B5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir”. Lancet. 359(9308): 727–732. doi:10.1016/s0140-6736(02)07873-x. PMID 11888582.
- ^ Rotimi, C. N.; Jorde, L. B. (2010). “Ancestry and disease in the age of genomic medicine”. New England Journal of Medicine. 363 (16): 1551–1558. doi:10.1056/nejmra0911564. PMID 20942671.
- ^ Phillips, E., Mallal, S. (2009). “Successful translation of pharmacogenetics into the clinic”. Molecular Diagnosis & Therapy. 13: 1–9. doi:10.1007/bf03256308.
- ^ Phillips, E., Mallal S. (2007). “Drug hypersensitivity in HIV”. Current Opinion in Allergy and Clinical Immunology. 7 (4): 324–330. doi:10.1097/aci.0b013e32825ea68a. PMID 17620824.
- ^ “Postmarket Drug Safety Information for Patients and Providers – Information for Healthcare Professionals: Abacavir (marketed as Ziagen) and Abacavir-Containing Medications”. Center for Drug Evaluation and Research at the US FDA. Archived from the original on 2013-12-11. Retrieved 2013-11-29.
- ^ “Archived copy”. Archived from the original on 2014-08-08. Retrieved 2014-07-31.
- ^ Swen JJ, Nijenhuis M, de Boer A, et al. (May 2011). “Pharmacogenetics: from bench to byte–an update of guidelines”. Clin Pharmacol Ther. 89 (5): 662–73. doi:10.1038/clpt.2011.34. PMID 21412232.
- ^ Martin MA, Hoffman JM, Freimuth RR, et al. (May 2014). “Clinical Pharmacogenetics Implementation Consortium Guidelines for HLA-B Genotype and Abacavir Dosing: 2014 update”. Clin Pharmacol Ther. 95 (5): 499–500. doi:10.1038/clpt.2014.38. PMC 3994233. PMID 24561393.
- ^ Shear, N.H., Milpied, B., Bruynzeel, D. P.; et al. (2008). “A review of drug patch testing and implications for HIV clinicians”. AIDS. 22 (9): 999–1007. doi:10.1097/qad.0b013e3282f7cb60. PMID 18520343.
- ^ “FDA Alert: Abacavir – Ongoing Safety Review: Possible Increased Risk of Heart Attack”. Drugs.com. Archived from the original on 2013-12-10. Retrieved 2013-11-29.
- ^ Ding X, Andraca-Carrera E, Cooper C, et al. (December 2012). “No association of abacavir use with myocardial infarction: findings of an FDA meta-analysis”. J Acquir Immune Defic Syndr. 61 (4): 441–7. doi:10.1097/QAI.0b013e31826f993c. PMID 22932321.
- ^ Illing, PT; et al. (2012). “Immune self-reactivity triggered by drug-modified HLA-peptide repertoire”. Nature. 486 (7404): 554–8. doi:10.1038/nature11147. PMID 22722860.
- ^ Jump up to:a b Prescribing information. Ziagen (abacavir). Research Triangle Park, NC: GlaxoSmithKline, July 2002
- ^ Vourvahis, M; Kashuba, AD (2007). “Mechanisms of Pharmacokinetic and Pharmacodynamic Drug Interactions Associated with Ritonavir-Enhanced Tipranavir”. Pharmacotherapy. 27 (6): 888–909. doi:10.1592/phco.27.6.888. PMID 17542771.
- ^ McDowell, JA; Chittick, GE; Stevens, CP; et al. (2000). ““, “Pharmacokinetic Interaction of Abacavir (1592U89) and Ethanol in Human Immunodeficiency Virus-Infected Adults”. Antimicrob Agents Chemother. 44 (6): 1686–90. doi:10.1128/aac.44.6.1686-1690.2000. PMC 89933. PMID 10817729.
- ^ Berenguer, J; Perez-Elias, MJ; Bellon, JM; et al. (2006). “Effectiveness and safety of abacavir, lamivudine, and zidovudine in antiretroviral therapy-naive HIV-infected patients: results from a large multicenter observational cohort”. J Acquir Immune Defic Syndr. 41 (2): 154–159. doi:10.1097/01.qai.0000194231.08207.8a. PMID 16394846.
- ^ Dolophine(methadone) [prescribing information]. Columbus, OH: Roxane Laboratories, Inc.; March 2015.
- ^ Gervasoni, C; Cattaneo, D; Di Cristo, V; et al. (2016). “Orlistat: weight lost at cost of HIV rebound”. J Antimicrob Chemother. 71 (6): 1739–1741. doi:10.1093/jac/dkw033. PMID 26945709.
- ^ Cometriq (cabozantinib) [prescribing information]. South San Francisco, CA: Exelixis, Inc.; May 2016.
- ^ Jump up to:a b Product Information: ZIAGEN(R) oral tablets, oral solution, abacavir sulfate oral tablets, oral solution. ViiV Healthcare (per Manufacturer), Research Triangle Park, NC, 2015.
- ^ “Dr. Robert Vince – 2010 Inductee”. Minnesota Inventors Hall of Fame. Minnesota Inventors Hall of Fame. Archived from the original on 15 February 2016. Retrieved 10 February 2016.
- ^ “Robert Vince, PhD (faculty listing)”. University of Minnesota. University of Minnesota. Archived from the original on 2016-02-17.
- ^ Daluge SM, Good SS, Faletto MB, Miller WH, St Clair MH, Boone LR, Tisdale M, Parry NR, Reardon JE, Dornsife RE, Averett DR (May 1997). “1592U89, a novel carbocyclic nucleoside analog with potent, selective anti-human immunodeficiency virus activity”. Antimicrobial Agents and Chemotherapy. 41 (5): 1082–1093. doi:10.1128/AAC.41.5.1082. PMC 163855. PMID 9145874.
- ^ Crimmins, M. T.; King, B. W. (1996). “An Efficient Asymmetric Approach to Carbocyclic Nucleosides: Asymmetric Synthesis of 1592U89, a Potent Inhibitor of HIV Reverse Transcriptase”. The Journal of Organic Chemistry. 61 (13): 4192–4193. doi:10.1021/jo960708p. PMID 11667311.
External links
- Full Prescribing Information
- Abacavir pathway on PharmGKB
- Abacavir dosing guidelines from the Clinical Pharmacogenetics Implementation Consortium (CPIC)
- Abacavir dosing guidelines from the Dutch Pharmacogenetics Working Group (DPWG)
References
-
- Crimmins, M.T. et al.: J. Org. Chem. (JOCEAH) 61 4192 (1996).
- b Olivo, H.F. et al.: J. Chem. Soc., Perkin Trans. 1 (JCPRB4) 1998, 391.
- US 5 089 500 (Burroughs Wellcome; 18.2.1992; GB-prior. 27.6.1988).
- a EP 434 450 (Wellcome Found.; 26.6.1991; appl. 21.12.1990; USA-prior. 22.12.1989).
- EP 1 857 458 (Solmag; appl. 5.5.2006).
- aa EP 424 064 (Enzymatix; appl. 24.4.1991; GB-prior. 16.10.1989).
- US 6 340 587 (SmithKline Beecham; 22.1.2002; appl. 20.8.1998; GB-prior. 22.8.1997).
- c US 5 034 394 (Welcome Found.; 23.7.1991; appl. 22.12.1989; GB-prior. 27.6.1988).
- d WO 9 924 431 (Glaxo; appl. 12.11.1998; WO-prior. 12.11.1997).
-
Alternative syntheses:
- EP 878 548 (Lonza; appl. 13.5.1998; CH-prior. 13.5.1997).
-
Preparation of chloropyrimidine intermediate V:
- US 6 448 403 (SmithKline Beecham; 10.9.2002; appl. 3.2.1995; GB-prior. 4.2.1994).
-
Condensation of pyrimidines with cyclopentylamine IV:
- Vince, R.; Hua, M.: J. Med. Chem. (JMCMAR) 33 (1), 17 (1990).
- Grumam, A. et al.: Tetrahedron Lett. (TELEAY) 36 (42), 7767 (1995).
- EP 349 242 (Wellcome Found.; appl. 26.6.1989; GB-prior. 27.6.1988).
- EP 366 385 (Wellcome Found.; appl. 23.10.1989; GB-prior. 24.10.1988).
- US 6 646 125 (SmithKline Beecham; 11.11.2003; appl. 14.10.1998; GB-prior. 14.10.1997).
- JP 1 022 853 (Asahi Glass Co.; appl. 17.7.1987).
-
Alternative preparation of 4-amino-2-cyclopentene-1-methanol:
- EP 926 131 (Lonza; appl. 24.11.1998; CH-prior. 27.11.1997).
- WO 9 745 529 (Lonza; appl. 30.5.1997; CH-prior. 30.5.1996).
- WO 9 910 519 (Glaxo; 4.3.1999; GB-prior. 20.8.1998).
- WO 9 824 741 (Glaxo; 11.6.1998; GB-prior. 7.12.1996).
- WO 2 001 017 952 (Chirotech; 15.3.2001; GB-prior. 9.9.1999).
-
Abacavir hemisulfate salt:
- US 6 294 540 (Glaxo Wellcome; 25.9.2001; appl. 14.5.1998; GB-prior. 17.5.1997).
-
Abacavir succinate as antiviral agent:
- WO 9 606 844 (Wellcome; 7.3.1996; appl. 25.8.1995; GB-prior. 26.8.1994).
-
Pharmaceutical formulations:
- US 6 641 843 (SmithKline Beecham; 4.11.2003; appl. 4.2.1999; GB-prior. 6.2.1998).
-
Synergistic combinations for treatment of HIV infection:
- WO 9 630 025 (Wellcome; 3.10.1996; appl. 28.3.1996; GB-prior. 30.3.1995).
 |
|
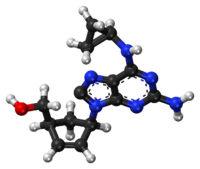
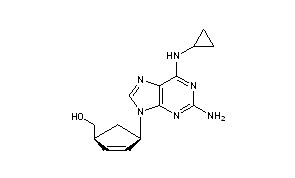
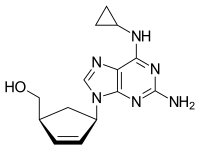
Chemical structure of abacavir
|
|
| Clinical data | |
|---|---|
| Pronunciation | /əˈbækəvɪər/ ( |
| Trade names | Ziagen, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699012 |
| License data | |
| Pregnancy category |
|
| Routes of administration |
By mouth (solution or tablets) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 83% |
| Metabolism | Liver |
| Elimination half-life | 1.54 ± 0.63 h |
| Excretion | Kidney (1.2% abacavir, 30% 5′-carboxylic acid metabolite, 36% 5′-glucuronide metabolite, 15% unidentified minor metabolites). Fecal (16%) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| ECHA InfoCard | 100.149.341 |
| Chemical and physical data | |
| Formula | C14H18N6O |
| Molar mass | 286.332 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 165 °C (329 °F) |