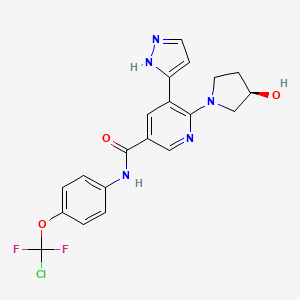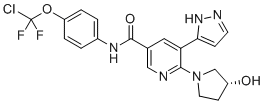
ABL001 / Asciminib
Cas 1492952-76-7
Chemical Formula: C20H18ClF2N5O3
Molecular Weight: 449.8428
Elemental Analysis: C, 53.40; H, 4.03; Cl, 7.88; F, 8.45; N, 15.57; O, 10.67
N-[4-[Chloro(difluoro)methoxy]phenyl]-6-[(3R)-3-hydroxypyrrolidin-1-yl]-5-(1H-pyrazol-5-yl)pyridine-3-carboxamide
3-Pyridinecarboxamide, N-[4-(chlorodifluoromethoxy)phenyl]-6-[(3R)-3-hydroxy-1-pyrrolidinyl]-5-(1H-pyrazol-3-yl)-
PHASE 3, Chronic Myeloid Leukemia, NOVARTIS
Asciminib is an orally bioavailable, allosteric Bcr-Abl tyrosine kinase inhibitor with potential antineoplastic activity. Designed to overcome resistance, ABL001 binds to the Abl portion of the Bcr-Abl fusion protein at a location that is distinct from the ATP-binding domain. This binding results in the inhibition of Bcr-Abl-mediated proliferation and enhanced apoptosis of Philadelphia chromosome-positive (Ph+) hematological malignancies. The Bcr-Abl fusion protein tyrosine kinase is an abnormal enzyme produced by leukemia cells that contain the Philadelphia chromosome.
- Originator Novartis
- Developer Novartis; Novartis Oncology
- Class Antineoplastics; Pyrazoles; Pyrrolidines; Small molecules
- Mechanism of Action Bcr-abl tyrosine kinase inhibitors
Highest Development Phases
- Phase III Chronic myeloid leukaemia
- No development reported Precursor cell lymphoblastic leukaemia-lymphoma
Most Recent Events
- 04 Nov 2017 No recent reports of development identified for phase-I development in Acute-lymphoblastic-leukaemia(Second-line therapy or greater) in Australia (PO)
- 04 Nov 2017 No recent reports of development identified for phase-I development in Acute-lymphoblastic-leukaemia(Second-line therapy or greater) in France (PO)
- 04 Nov 2017 No recent reports of development identified for phase-I development in Acute-lymphoblastic-leukaemia(Second-line therapy or greater) in Germany (PO)
- The tyrosine kinase activity of the ABLl protein is normally tightly regulated, with the N-terminal cap region of the SH3 domain playing an important role. One regulatory mechanism involves the N-terminal cap glycine-2 residue being myristoylated and then interacting with a myristate binding site within the SHI catalytic domain. A hallmark of chronic myeloid leukemia (CML) is the Philadelphia chromosome (Ph), formed by the t(9,22) reciprocal chromosome translocation in a haematopoietic stem cell. This chromosome carries the BCR-ABL1 oncogene which encodes the chimeric BCR-ABL1 protein, that lacks the N-terminal cap and has a constitutively active tyrosine kinase domain.Although drugs that inhibit the tyrosine kinase activity of BCR-ABL1 via an ATP-competitive mechanism, such as Gleevec® / Glivec® (imatinib), Tasigna® (nilotinib) and Sprycel® (dasatinib), are effective in the treatment of CML, some patients relapse due to the emergence of drug-resistant clones, in which mutations in the SHI domain compromise inhibitor binding. Although Tasigna® and Sprycel® maintain efficacy towards many Gleevec-resistant mutant forms of BCR-ABLl, the mutation in which the threonine-315 residue is replaced by an isoleucine (T315I) remains insensitive to all three drugs and can result in CML patients developing resistance to therapy. Therefore, inhibiting BCR-ABLl mutations, such as T315I, remains an unmet medical need. In addition to CML, BCR-ABLl fusion proteins are causative in a percentage of acute lymphocytic leukemias, and drugs targeting ABL kinase activity also have utility in this indication.Agents targeting the myristoyl binding site (so-called allosteric inhibitors) have potential for the treatment of BCR-ABLl disorders (J. Zhang, F. J. Adrian, W. Jahnke, S. W. Cowan- Jacob, A. G. Li, R. E. Iacob4, T. Sim, J. Powers, C. Dierks, F. Sun, G.-R. Guo, Q. Ding, B. Okram, Y. Choi, A. Wojciechowski, X. Deng, G. Liu, G. Fendrich, A. Strauss, N. Vajpai, S. Grzesiek, T. Tuntland, Y. Liu, B. Bursulaya, M. Azam, P. W. Manley, J. R. Engen, G. Q. Daley, M. Warmuth., N. S. Gray. Targeting BCR-ABL by combining allosteric with ATP -binding-site inhibitors. Nature 2010;463:501-6). To prevent the emergence of drug resistance from ATP inhibitor and/or allosteric inhibitor use, a combination treatment using both types of inhibitor can be developed for the treatment of BCR-ABLl related disorders. In particular, the need exists for small molecules, or combinations thereof, that inhibit the activity of BCR-ABLl and BCR-ABLl mutations via the ATP binding site, the myristoyl binding site or a combination of both sites.Further, inhibitors of ABL 1 kinase activity have the potential to be used as therapies for the treatment of metastatic invasive carcinomas and viral infections such as pox and Ebola viruses.The compounds from the present invention also have the potential to treat or prevent diseases or disorders associated with abnormally activated kinase activity of wild-type ABL1, including non-malignant diseases or disorders, such as CNS diseases in particular neurodegenerative diseases (for example Alzheimer’s, Parkinson’s diseases), motoneuroneuron diseases (amyotophic lateral sclerosis), muscular dystrophies, autoimmune and inflammatory diseases (diabetes and pulmonary fibrosis), viral infections, prion diseases.
Asciminib is an allosteric inhibitor of BCR-ABL kinase in phase III clinical development at Novartis for the treatment of patients with chronic myelogenous leukemia (CML) in chronic phase who have been previously treated with ATP-binding site tyrosine kinase inhibitors. Early clinical trials are also under way in patients with Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) and as first-line threapy of CML.
PATENT
To illustrate tautomerism with the following specific examples, (R)-N-(4- (chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-l-yl)-5-(lH-pyrazol-5-yl)nicotinamide
(right structure, below) is a tautomer of (R)-N-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-l-yl)-5-(lH-pyrazol-3-yl)nicotinamide (left structure, below) and vice versa:

[0045] Where the plural form (e.g. compounds, salts) is used, this includes the singular
Example 9
(R)-N-(4-(Chlorodifluoromethoxy)phenyl)-6-(3-hvdroxypyrrolidin-l-yl)-5-(lH-pyrazol-5- vDnicotinamide

[00365] A mixture of (R)-5-Bromo-N-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-l-yl)nicotinamide (Stage 9.2, 100 mg, 0.216 mmol) and 5-(4 ,4,5,5-tetramethyl- 1 ,3 ,2-dioxaborolan-2-yl)- 1 -((2-(trimethylsilyl)ethoxy)methyl)- IH-pyrazole (215 mg, 0.663 mmol), Pd(PPh3)2Cl2 (17 mg, 0.024 mmol), Na2C03 (115 mg, 1.081 mmol), DME (917 μί), water (262 μΕ) and EtOH (131 μί) in a MW vial was sealed, evacuated / purged 3 times with argon and subjected to MW irradiation at 125°C for 20 min. The RM was diluted with 2 mL
of DME, stirred with Si-Thiol (Silicycle 1.44 mmol/g, 90 mg, 0.130 mmol) for 3 h. The mixture was centrifuged and the supernatant was filtered through a 0.45 μηι PTFE filter and the solvent was evaporated off under reduced pressure. The crude product was purified by flash
chromatography (RediSep® Silica gel column, 12 g, cyclohexane / EtOAc from 40% to 100% EtOAc) to afford the protected intermediate as a colorless oil. Ethylene diamine (96 μί, 1.428 mmol) and TBAF 1 M in THF (1.428 mL, 1.428 mmol) were then added and the RM was stirred at 80-85°C for 5 days. The solvent was evaporated off under reduced pressure and the residue was dissolved in EtOAc (40 mL), washed 3 times with sat. aq. NaHCC and brine, dried over Na2S04 and The solvent was evaporated off under reduced pressure to give a residue which was purified by preparative SFC (Column DEAP, from 25% to 30% in 6 min) to yield the title compound as a white solid.
[00366] Alternatively, Example 9 was prepared by adding TFA (168 mL, 2182 mmol) to a solution of N-(4-(chlorodifluoromethoxy)phenyl)-6-((R)-3-hydroxypyrrolidin-l-yl)-5-(l-(tetrahydro-2H-pyran-2-yl)-lH-pyrazol-5-yl)nicotinamide (Stage 9.1, 31.3 g, 54.6 mmol) in DCM (600 mL). The mixture was stirred at RT for 2.5 h. The solvent was evaporated off under reduced pressure and the residue was dissolved in EtOAc (1.5 L),washed with a sat. solution of NaHC03 (3 x 500 mL) and brine (500 mL), dried over Na2S04 and the solvent was evaporated off under reduced pressure to give a residue which was suspended in DCM (300 mL), stirred at RT for 15 min, filtered, washed with DCM (200 mL), dried and purified by chromatography (Silica gel, 1 kg, DCM / MeOH 95:5). The residue was dissolved in MeOH (500 mL) and treated with Si-Thiol (Biotage, 5.0 g , 6.5 mmol) for 16 h at 25°C. The resin was filtered off, the solvent was evaporated off under reduced pressure and the residue was crystallized from MeCN to afford the title compound as a white crystalline solid.
[00367] Alternatively, Example 9 was prepared by the dropwise addition of aqueous HC1
(7.7 mL of 6M) to a solution of N-(4-(chlorodifluoromethoxy)phenyl)-6-((R)-3-hydroxypyrrolidin- 1 -yl)-5-( 1 -(tetrahydro-2H-pyran-2-yl)- 1 H-pyrazol-5-yl)nicotinamide (Stage 9.1, 3.8 g, 7.12 mmol) in MeOH (20 mL) and THF (10 mL) with cooling (below 35°C). The mixture was stirred at 22°C for 2 h and then added to cooled (10°C) 1.2 M NaOH (22 mL).
Throughout the addition the temperature was kept below 30°C and pH was kept in the range of 9-10. The RM was then stirred for 30 min at 30°C. The solvent was evaporated off under reduced pressure, until the desired compound precipitated. The precipitate was filtered and dried to give the title compound as a yellow solid.
[00368] Analytical data for Example 9: HPLC (Condition 5) tR = 5.54 min, HPLC Chiral
(CHIRALCEL® OD-H, 250 x 4.6 mm, eluent : n-heptane/EtOH/MeOH (85: 10:5), 1 mL/min, UV 210 nm) tR = 10.17 min, UPLC-MS (condition 3) tR = 0.93 min, m/z = 450.3 [M+H]+, m/z = 494.1 [M+formic acid-H]“; XH-NMR (400 MHz, DMSO-d6) δ ppm 1.65 – 1.76 (m, 1 H) 1.76 – 1.87 (m, 1 H) 2.93 (d, J=l 1.73 Hz, 1 H) 3.19 – 3.29 (m, 2 H) 3.35 – 3.51 (m, 1 H) 4.10 – 4.25 (m, 1 H) 4.89 (br. s, 1 H) 6.41 (br. s, 1 H) 7.33 (d, J=8.50 Hz, 2 H) 7.57/7.83 (br. s, 1 H) 7.90 (d, J=8.50 Hz, 2 H) 8.07 (br. s, 1 H) 8.77 (br. s, 1 H) 10.23 (s, 1 H) 12.97/13.15 (br. s, 1 H).
[00369] Stage 9.1 : N-(4-(Chlorodifluoromethoxy)phenyl)-6-((R)-3-hydroxypyrrolidin- 1 -yl)-5-( 1 -(tetrahydro-2H-pyran-2- l)- 1 H-pyrazol-5-yl)nicotinamide

[00370] l-(Tetrahydro-2H-pyran-2-yl)-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH-pyrazole (29.6 g, 102 mmol), K3P04 (51.6 g, 236 mmol) and Pd(PPh3)4 (4.55 g, 3.93 mmol) were added to a suspension of (R)-5-bromo-N-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-l-yl)nicotinamide (Stage 9.2, 36.4 g, 79 mmol) in toluene (360 mL) under an argon atmosphere and the mixture was stirred at 110°C for 4 h. The RM was poured into brine (500 mL) and extracted with EtOAc (2 x 1 L). The combined extracts were washed with brine (500 mL), dried over Na2S04, and the solvent was evaporated off under reduced pressure to give a residue which was purified by chromatography (Silica gel column, 1.5 kg, DCM / MeOH 95:5) to afford a dark yellow foam, that was dissolved in MeOH / DCM (1 L of 3: l) and treated with Si-Thiol (Biotage, 35 g , 45.5 mmol) for 17 h at 30°C. The resin was filtered off, and solvent was evaporated off under reduced pressure, until the desired compound crystallized. The product was filtered washed with MeOH and dried to afford the title compound.
[00371] Alternatively, Stage 9.1 was prepared by adding 4-(chlorodifluoromethoxy)aniline
(16.6 g, 84.9 mmol), NMM (21.7 g, 212.1 mmol), hydroxybenzotriazole hydrate (HOBt H20, 11.9 g, 77.77 mmol) and l-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCIHCl, 20.9 g, 109.0 mmol) to a solution of 6-((R)-3-hydroxypyrrolidin-l-yl)-5-(l-(tetrahydro-2H-pyran-2-yl)-lH-pyrazol-5-yl)nicotinic acid (Stage 9.4, 29.83 g, 70.7 mmol) in THF (271 mL). The mixture was stirred for 1.5 h at 25°C and then at 65°C for 16 h. After cooling the RM to 35 °C, further EDCIHCl (13.3 g, 69.4 mmol) was added and the RM was stirred for 1.5 h at 35°C then again at 65°C for 16 h. After cooling the RM to 35°C, water (150 mL) was added, the THF was removed under reduced pressure, EtOAc (180 mL) was added and the mixture was stirred for at 35 °C fori h. The two layers were separated and the aq. phase was then extracted with EtOAc (60 mL). The combined organic layers were washed with water (90 mL), brine (90 mL). The solvent was evaporated off under reduced pressure to give a brown solid which was purified by column chromatography (Silica gel, DCM / MeOH 40: 1 to 20: 1) to afford the title compound as a yellow solid.
[00372] Analytical data for Stage 9.1: HPLC (Condition 5) tR = 6.12 min, UPLC-MS
(Condition 3) tR = 1.06 min, m/z = 533.2 [M+H]+; XH-NMR (400 MHz, DMSO-d6) δ ppm 1.36 -2.02 (m, 7 H) 2.23 – 2.38 (m, 1 H) 3.08 – 3.29 (m, 2 H) 3.32 – 3.52 (m, 2 H) 3.73 – 3.93 (m, 1 H) 4.13 – 4.25 (m, 1 H) 4.80 – 4.90 (m, 1 H) 4.95 – 5.17 (m, 1 H) 6.33 – 6.50 (m, 1 H) 7.33 (d, J=8.99 Hz, 2 H) 7.61 (d, J=1.56 Hz, 1 H) 7.86 (d, J=8.99 Hz, 2 H) 7.97 – 8.11 (m, 1 H) 8.82 (s, 1 H) 10.13 – 10.25 (m, 1 H).
[00373] Stage 9.2: (R)-5-Bromo-N-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin- 1 -yl)nicotinamide

[00374] (R)-Pyrrolidin-3-ol (9.55 g, 109.6 mmol) and DIPEA (35.1 ml, 201.3 mmol) were added to a suspension of 5-bromo-6-chloro-N-(4-(chlorodifluoromethoxy)phenyl)nicotinamide (Stage 9.3, 37.7 g, 91.5 mmol) in iPrOH (65 mL) and stirred at 140°C for 1 h. EtOAc (700 mL) was added and the solution was washed IN HC1 (2 x 200 mL), sat. NaHCC (200 mL) and brine (2 x 200 mL), dried over Na2S04, and the solution was concentrated under reduced pressure until crystallization commenced. n-Heptane (1 L) were added and the mixture was stirred at RT for 30 min, filtered and washed with ΪΡΓ20 (500 mL) to afford the title compound as a white crystalline solid. HPLC (Condition 5) tR = 6.68 min, UPLC-MS (Condition 3) tR = 1.10 min, m/z =
462.2/464.2 [M+H]+; XH-NMR (400 MHz, DMSO-d6) δ ppm 1.78 – 2.01 (m, 2 H) 3.55 (d, J=l 1.34 Hz, 1 H) 3.66 – 3.75 (m, 1 H) 3.79 – 3.93 (m, 2 H) 4.34 (br. s, 1 H) 4.98 (d, =3.13 Hz, 1 H) 7.32 (d, J=8.99 Hz, 2 H) 7.84 (d, J=8.99 Hz, 2 H) 8.33 (d, J=1.96 Hz, 1 H) 8.66 (d, J=1.96 Hz, 1 H) 10.21 (s, 1 H).
[00375] Stage 9.3: 5-Bromo-6-chloro-N- 4-(chlorodifluoromethoxy)phenyl)nicotinamide

[00376] DMF (2.55 mL, 33.0 mmol) and SOCl2 (24.08 ml, 330 mmol) were added to a suspension of 5-bromo-6-chloro-nicotinic acid (26 g, 110 mmol) in toluene (220 mL) and the RM was stirred at 80°C for 1 h. The solvent was evaporated off under reduced pressure and the residue was dissolved in THF (220 mL) and cooled to -16°C. DIPEA (38.4 mL, 220 mmol) was added, followed by dropwise addition of a solution of 4-(chlorodifluoromethoxy)aniline (22.35 g, 115 mmol) in THF (220 mL) over 15 min. The suspension was stirred for 1 h at RT. The solvent was evaporated off under reduced pressure and the residue was dissolved in TBME (700 mL), washed with IN HC1 (2 x 200 mL), sat. NaHC03 (200 mL) and brine (2 x 200 mL), dried over Na2S04, and the solvent was evaporated off under reduced pressure to give the product which was crystallized from EtOAc – n-heptane to afford the title compound as a white crystalline solid. HPLC (Condition 5) tR = 7.77 min, UPLC-MS (Condition 3) tR = 1.24 min, m/z =
409.1/411.1/413.1 [M+H]+; XH-NMR (400 MHz, DMSO-d6) δ ppm 7.38 (d, =8.99 Hz, 2 H) 7.85 (d, =8.99 Hz, 2 H) 8.72 (br. s, 1 H) 8.92 (br. s, 1 H) 10.68 (s, 1 H).
[00377] Stage 9.4: 6-((R)-3-Hydroxypyrrolidin-l-yl)-5-(l-(tetrahydro-2H-pyran-2-yl)-lH-pyrazol-5-yl)nicotinic acid

[00378] Aq. NaOH (180 niL of 2.6 M) was added to a solution of methyl 6-((R)-3-hydroxypyrrolidin- 1 -yl)-5-(l -(tetrahydro-2H-pyran-2-yl)- 1 H-pyrazol-5-yl)nicotinate (Stage 9.5, 11 lg, 299 mmol) in MeOH (270 mL) and the RM was stirred at RT for 14 h. The MeOH was evaporated off under reduced pressure and the aq. residue was treated with brine (90 mL), extracted with MeTHF twice (540 mL + 360 mL) and the combined organic layers were washed with water (90 mL). MeTHF was added to the combined aq. layers, the biphasic mixture was cooled to 0 °C and acidified (pH = 4-4.5) with aq. HC1 solution (18%) and extracted with
MeTHF. The combined organic extracts were washed with brine and the solvent was evaporated off under reduced pressure to give a residue which was recrystallized from a EtOAc / TBME (1 : 1) to afford the title compound as a white solid. HPLC (Condition 7) tR = 4.74 min, LC-MS
(Condition 8) tR = 3.37 min, m/z = 359.0 [M+H]+; XH-NMR (400 MHz, DMSO-d6) δ ppm 1.44 (br. s, 2 H), 1.51 (d, J=11.54 Hz, 2 H), 1.64 – 1.86 (m, 4 H), 1.90 (br. s, 1 H), 2.31 (d, J=9.29 Hz, 1 H), 2.77 (br. s, 1 H), 3.10 (br. s, 1 H), 3.21 (d, J=8.78 Hz, 2 H), 3.27 – 3.51 (m, 4 H), 3.87 (d, J=11.54 Hz, 1 H), 4.16 (br. s, 1 H), 4.75 – 4.93 (m, 1 H), 5.04 (br. s, 1 H), 6.35 (d, J=17.32 Hz, 1 H), 7.51 – 7.64 (m, 1 H), 7.64 – 7.82 (m, 1 H), 8.67 (d, J=2.26 Hz, 1 H), 12.58 (br. s, 1 H).
[00379] Stage 9.5: Methyl 6-((R)-3-hydroxypyrrolidin-l-yl)-5-(l-(tetrahydro-2H-pyran-2-yl)- 1 H-pyrazol-5-yl)nicotinate

[00380] A mixture of (R)-methyl 5-bromo-6-(3-hydroxypyrrolidin-l-yl)nicotinate (Stage
9.6, 90 g, 299 mmol), l-(tetrahydro-2H-pyran-2-yl)-lH-pyrazole-5-boronic acid pinacol ester (103.9 g, 373.6 mmol), K3P04 (126.9 g, 597.7 mmol), Pd(PPh3)2Cl2 (6.29 g, 8.97 mmol) in toluene (900 mL) was stirred at 92°C and for 16 h. After cooling the mixture to RT, the solution was washed with water (450 mL), 5% NaHCC solution (430 mL) and the solvent was evaporated off under reduced pressure to give a residue which was used without further purifications in the next step. HPLC (Condition 7) tR = 6.929 min, LC-MS (Condition 8) tR = 4.30 min, m/z = 373.0 [M+H ; XH-NMR (400 MHz, DMSO-d6) δ ppm 1.19 – 1.28 (m, 1 H), 1.35 – 1.63 (m, 4 H), 1.63 -1.86 (m, 3 H), 1.89 (br. s, 1 H), 2.12 – 2.39 (m, 1 H), 3.11 (br. s, 1 H), 3.18 – 3.48 (m, 4 H), 3.78 (s, 4 H), 3.88 (d, J=11.54 Hz, 1 H), 4.08 – 4.24 (m, 1 H), 4.86 (dd, J=18.20, 2.89 Hz, 1 H), 5.02 (d, J=8.28 Hz, 1 H), 6.39 (br. s, 1 H), 7.58 (d, J=1.25 Hz, 1 H), 7.78 (br. s, 1 H), 8.69 (t, J=2.01 Hz, 1 H).
[00381] Stage 9.6: (R)-methyl 5-bromo-6-(3-hydroxypyrrolidin-l-yl)nicotinate

[00382] DIPEA (105.3 g, 142.2 mL, 814.4 mmol) was added to a solution of methyl-5-bromo-6-chroronicotinate (85 g, 339.5 mmol) and (R)-pyrrolidin-3-ol (54.2 g, 441.2 mmol) in isopropyl acetate and the RM was stirred at 70°C for 14 h . The solvent was evaporated off under reduced pressure to give a the residue which was dissolved in toluene (850 mL), washed with water (127 mL) and brine (127 mL)and concentrated under reduced pressure until precipitation commenced. n-Heptane (340 mL) was slowly added to the stirred mixture at 22 °C, which was then cooled to 0 °C and the product was filtered, washed with a toluene / n-heptane mixture
(1 : 1.5) and dried to give the title compound as a yellow solid. HPLC (Condition 7) tR = 8.54 min, LC-MS (Condition 8) tR = 4.62 min, m/z = 300.9/302.9 [M+H]+; XH-NMR (400 MHz, DMSO-d6) δ ρριη 1.77 – 1.99 (m, 2 H), 3.57 (d, J=11.54 Hz, 1 H), 3.72 (ddd, J=l 1.11, 7.97, 3.26 Hz, 1 H), 3.78 (s, 3 H), 3.81 -3.90 (m, 2 H), 4.26 – 4.39 (m, 1 H), 4.99 (br. s, 1 H), 8.11 (d, J=2.01 Hz, 1 H), 8.56 (d, J=1.76 Hz, 1 H).
PAPER
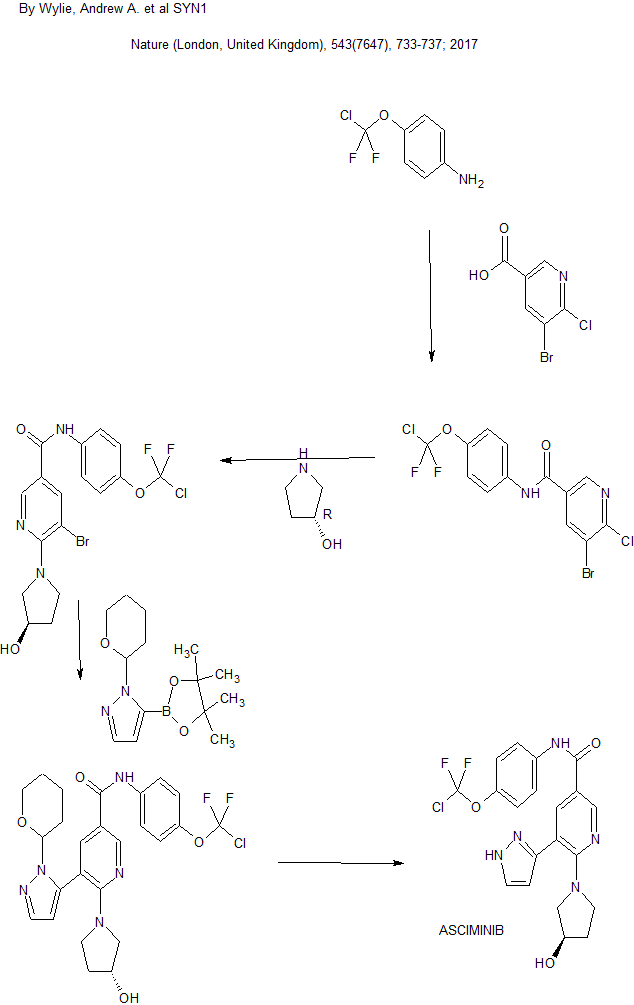
- By Wylie, Andrew A.; Schoepfer, Joseph; Jahnke, Wolfgang; Cowan-Jacob, Sandra W.; Loo, Alice; Furet, Pascal; Marzinzik, Andreas L.; Pelle, Xavier; Donovan, Jerry; Zhu, Wenjing; et al
- From Nature (London, United Kingdom) (2017), 543(7647), 733-737.
By Wylie, Andrew A. et alFrom Nature (London, United Kingdom), 543(7647), 733-737; 2017

PAPER
- By Molica, Matteo; Massaro, Fulvio; Breccia, Massimo
- From Expert Opinion on Pharmacotherapy (2017), 18(1), 57-65.
PATENT
US 20170216289
PAPER
- By El Rashedy, Ahmed A.; Olotu, Fisayo A.; Soliman, Mahmoud E. S.
- From Chemistry & Biodiversity (2018), 15(3), n/a.
////////////////ABL001, Asciminib, ABL 001, ABL-001, PHASE 3, Chronic Myeloid Leukemia, NOVARTIS
O=C(NC1=CC=C(OC(F)(Cl)F)C=C1)C2=CN=C(N3C[C@H](O)CC3)C(C4=CC=NN4)=C2