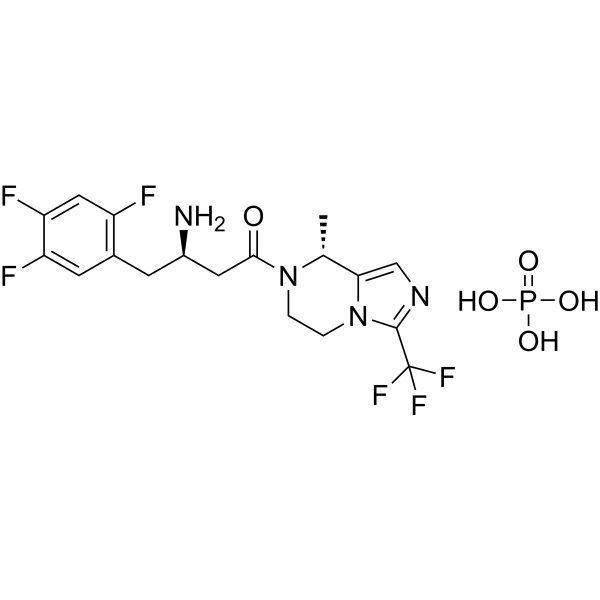
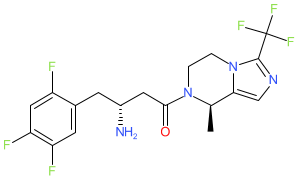
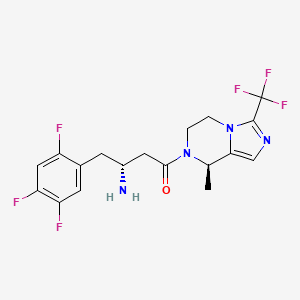
Cetagliptin
CAS No. FREE FORM : 2243737-33-7 C18H18F6N4O, 420.4 g/mol
[ Cetagliptin Phosphate 2243737-33-7 ]
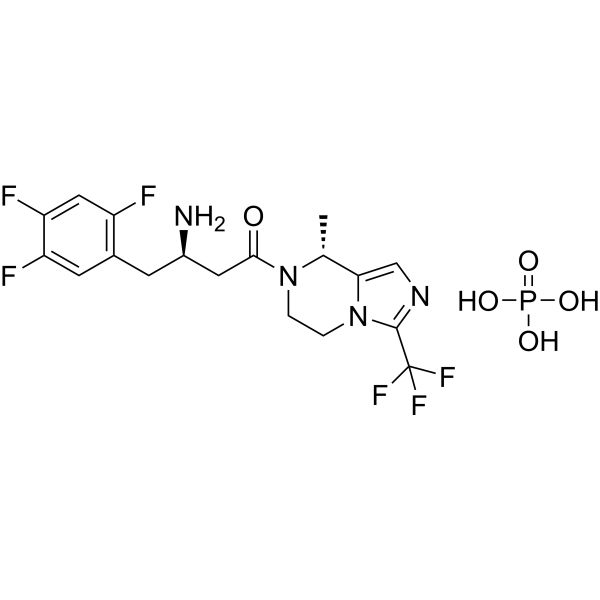
| 분자량 MW | 518.35 |
|---|---|
| 화학식 MF | C18H21F6N4O5P |
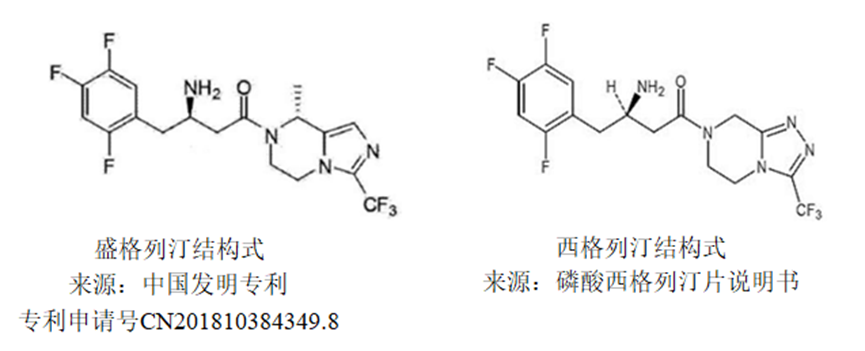
(3R)-3-amino-1-[(8R)-8-methyl-3-(trifluoromethyl)-6,8-dihydro-5H-imidazo[1,5-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one
(3R)-3-amino-1-[(8R)-8-methyl-3-(trifluoromethyl)-6,8-dihydro-5H-imidazo[1,5-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one
CHINA 2024, APPROVALS 2024, CGeneTec, DIABETES,
- GTPL13952
- CGT8012
- OriginatorCGeneTech
- Class2 ring heterocyclic compounds; Amines; Antihyperglycaemics; Fluorobenzenes; Imidazoles; Ketones; Pyrazines; Small molecules
- Mechanism of ActionDipeptidyl peptidase 4 inhibitors
RegisteredType 2 diabetes mellitus CHINA 2024
- 01 Dec 2024Registered for Type 2 diabetes mellitus in China (PO) – First global approval
- 20 Mar 2024Chemical structure information added
- 28 Jun 2023No recent reports of development identified for phase-I development in Type-2-diabetes-mellitus(In volunteers) in China (PO, Tablet)
- Cetagliptin is an orally active inhibitor for dipeptidyl peptidase 4 (DPP-4) and CYP2D6 (IC50 of 6 µM). Cetagliptin is a substrate for P-glycoprotein. Cetagliptin reduces the GLP-1 degradation, maintains the level of postprandial blood sugar, and can be used in type 2 diabetes mellitus research.
Cetagliptin (CGT-8012) is an orally bioavailable, dipeptidyl peptidase 4 enzyme (DPP-4) inhibitor (‘gliptin’) class drug. It was designed as an antihyperglycemic agent to treat type 2 diabetes mellitus (T2DM) via inhibition of DPP-4-mediated catbolism of incretin hormones including glucagon-like peptide-1 (GLP-1) [2].
- A DPP-4 inhibitor pharmaceutical composition and its preparation method and usePublication Number: CN-118557538-APriority Date: 2024-08-01
- A kind of preparation method of DPP-IV inhibitor and its key intermediatePublication Number: CN-114057751-APriority Date: 2022-01-17
- A kind of preparation method of DPP-IV inhibitor and its key intermediatePublication Number: CN-114057751-BPriority Date: 2022-01-17Grant Date: 2022-04-12
- A kind of preparation method of DPP-IV inhibitor and key intermediate thereofPublication Number: TW-202330535-APriority Date: 2022-01-17
- A preparation method of a DPP-IV inhibitor and its key intermediatePublication Number: TW-I842342-BPriority Date: 2022-01-17Grant Date: 2024-05-11
- Salt of cetagliptin, preparation method thereof, pharmaceutical composition, and use thereofPublication Number: US-2020123164-A1Priority Date: 2018-04-26
- Salt of cetagliptin, preparation method therefor, pharmaceutical composition, and use thereofPublication Number: EP-3785713-A1Priority Date: 2018-04-26
- Salt of cetagliptin, preparation method thereof, pharmaceutical composition, and use thereofPublication Number: US-11046701-B2Priority Date: 2018-04-26Grant Date: 2021-06-29
- Tetrahydro-imidaz0[1,5-a]pyrazine derivatives, preparation process and medicinal use thereofPublication Number: US-2010273786-A1Priority Date: 2007-12-26
- Tetrahydro-imidazo[1,5-α]pyrazine derivatives, preparation process and medicinal use thereofPublication Number: US-8207161-B2Priority Date: 2007-12-26Grant Date: 2012-06-26
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=CN84092509&_cid=P20-MERZ31-36806-1
SYN
CN103351391
https://patents.google.com/patent/CN103351391A/en
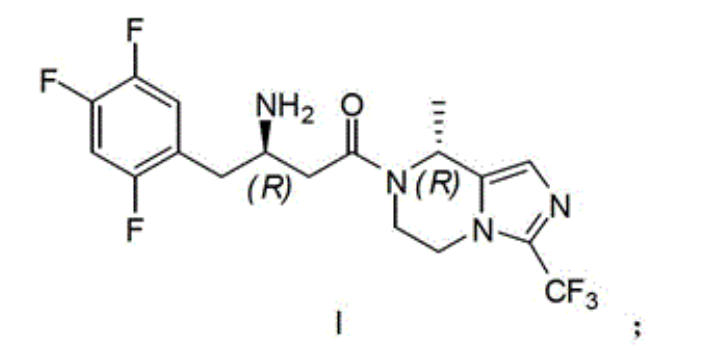
Synthetic route and the concrete steps of compound (I) are as follows:
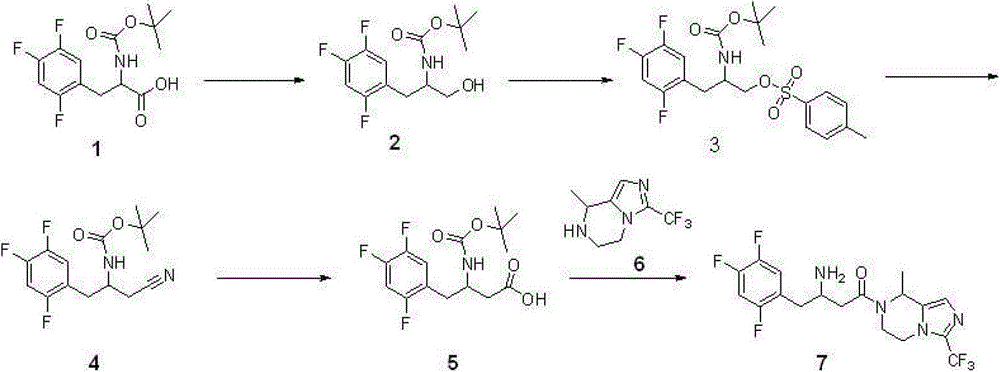
Step 1: synthetic compound 2
With 11.8 gram (0.037 mole) compound 1{DL N-[(1,1-dimethyl oxyethyl group) carbonyl]-2,4; 5-trifluorophenyl-L-Ala, DL N-[(1,1-dimethylethoxy) carbonyl]-2; 4; 5-trifluorophen yl-alanine, CAS:1367740-01-9, reference: synthetic chemistry; 2011; 19 (4), 557-560} is dissolved among 40 milliliters of THF, adds 5.8 milliliters of triethylamines (0.042 mole) again; reaction is cooled to 0 ℃; add 4.0 milliliters of Vinyl chloroformates (0.041 mole), 0 ℃ was reacted 1 hour under nitrogen protection, after the filtration filtered liquid was cooled to 0 ℃; slowly add sodium borohydride (1.4 grams; 0.057 the mole) mixed solution in 15 ml waters, stirring is spent the night, and adds 1N HCl acidifying; ethyl acetate extraction three times; merge organic phase, sodium hydrogen carbonate solution is washed, the saturated salt washing; anhydrous sodium sulfate drying; the concentrated 7.6 gram products that obtain, namely compound 2, yield 67%.Repeat this step, make more compound 2, use for subsequent step.
Step 2: synthetic compound 3
8.2 gram (0.027 mole) compounds 2 are dissolved in 40 milliliters of methylene dichloride; add again 4.2 milliliters of triethylamines (0.030 mole); the catalytic amount DMAP; reaction is cooled to 0 ℃; add Tosyl chloride (6.8 grams; 0.035 mole); 0 ℃ is arrived room temperature reaction 2 hours under nitrogen protection, adds 1N HCl acidifying, dichloromethane extraction three times; merge organic phase; sodium hydrogen carbonate solution is washed, saturated salt washing, anhydrous sodium sulfate drying; concentrate and obtain crude product, namely compound 3.Repeat this step, make more compound 3, use for subsequent step.
Step 3: synthetic compound 4
12.4 gram (0.027 mole) compounds 3 are dissolved in 40 milliliters of dimethyl formamides, slowly add the mixed solution of sodium cyanide (4.5 grams, 0.092 mole) in 30 milliliters of dimethyl formamides, room temperature reaction 48 hours, pour in 100 milliliters of frozen water, ethyl acetate extraction three times merges organic phase, the saturated salt washing, anhydrous sodium sulfate drying, concentrated rear column chromatography purification obtains 7.8 gram products, be compound 4, yield 92%.
Step 4: synthetic compound
5
3.1 gram (0.010 mole) compounds 4 are dissolved in 15 milliliters of 6N hydrochloric acid, and reflux is spent the night, and adds the neutralization of 2N sodium hydroxide solution, cooling drying.The gained solid is dissolved among 30 milliliters of THF, adds 20 milliliters of 0.5N sodium hydroxide solutions, adds tert-Butyl dicarbonate (2.4 grams again, 0.011 mole), room temperature reaction
16 hours, concentrated, add the neutralization of 10% sodium bisulfate, ethyl acetate extraction three times merges organic phase, the saturated salt washing, anhydrous sodium sulfate drying, the concentrated 3.3 gram products that obtain, namely, compound
5, yield 99%.
Step 5: synthetic compound 7
Compound 6{5; 6; 7; 8-tetrahydrochysene-8-methyl-3-(trifluoromethyl)-imidazo [1,5-a] pyrazine, 5; 6; 7,8-tetrahydro-8-methyl-3-(trifluoromethyl)-imidazo[1,5-a] pyrazine; synthesize and see CN103087067; 2.1 gram, 0.010 mole } be dissolved in 8 milliliters of methylene dichloride, add triethylamine 1.2 grams (0.012 mole); compound 5 (3.3 grams; 0.010 mole), EDCI2.3 restrains (0.012 mole), room temperature reaction is 24 hours under nitrogen protection; pour in 100 milliliters of frozen water; organic phase is washed saturated salt washing, anhydrous sodium sulfate drying; the concentrated crude product that obtains; be dissolved in 100 milliliters of the 2N HCl/ methanol solutions (anhydrous HCl gas is dissolved in the solution of methyl alcohol), room temperature reaction 4 hours is spin-dried for; cooling; pour in 100 milliliters of frozen water, transfer PH to 9, ethyl acetate extraction three times; merge organic phase; and wash saturated salt washing, anhydrous sodium sulfate drying; concentrated; column chromatography purification obtains 2.8 gram products, and namely compound 7, yield 66%.
Compound 7 comprises four optical isomers, and route and the concrete steps of their separation and purification are as follows:
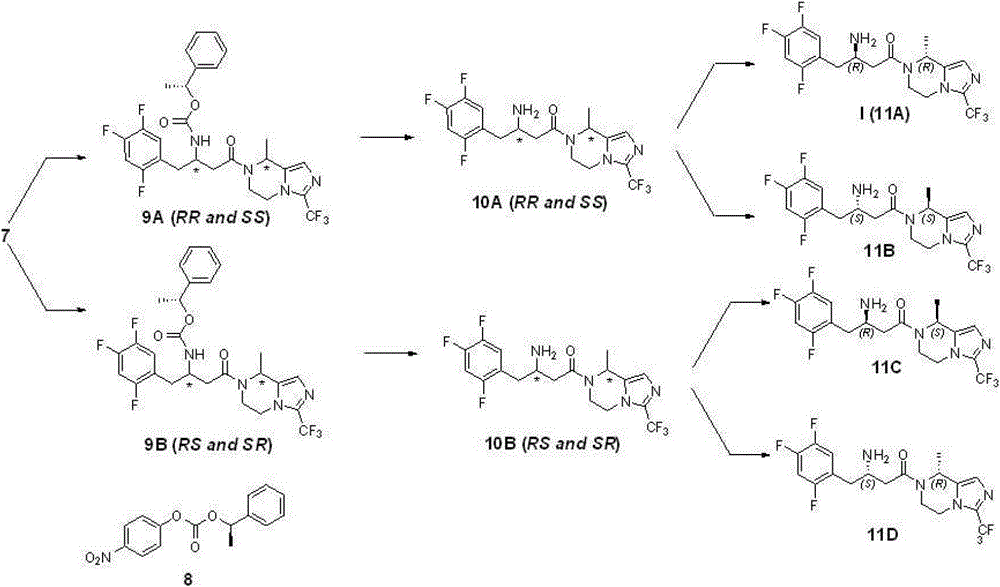
Step 6: preparation compound 9A and 9B
2.8 gram (6.67 mmole) compounds 7 are dissolved in 50 milliliters of acetonitriles; add triethylamine 1.2 grams (8.0 mmole); add again compound 8 (1.9 grams; 6.67 mmole; reference: J.Org.Chem.1995; 60 (3); 730), reflux is spent the night under nitrogen protection, and is concentrated; add ethyl acetate; the 1N sodium hydroxide solution is washed, and ethyl acetate milliliter extraction three times merges organic phase; the saturated salt washing; anhydrous sodium sulfate drying, the evaporating column chromatography purification obtains 1.6 gram 9A (43%) and 1.4 gram 9B (39%) products (de>98%); structural analysis determines that tentatively 9A is RR and SS mixture of enantiomers, and 9B is RS and SR mixture of enantiomers.Gained compound 9A and 9B give over to respectively next step and use.
Step 7: preparation compound 10A and 10B
1.5 gram (2.64 mmole) compound 9A are dissolved in 50 milliliters of methylene dichloride, reaction is cooled to 0 ℃, adds HBr solution (2M, 2.6 milliliters, 5.2 mmole), be dissolved in ethyl acetate after concentrated, sodium hydrogen carbonate solution is washed, the saturated salt washing, anhydrous sodium sulfate drying, the concentrated product that obtains, namely compound 10A (RR and SS mixture of enantiomers) gives over to next step and uses.
According to same reaction principle, condition and step, take compound 9B as starting raw material, obtain compound 10B (RS and SR mixture of enantiomers), give over to next step and use.
Step 8: preparation compound 11A, 11B and 11C, 11D
Resulting compound 10A in the step 7 (1.1 gram) is dissolved in 20 milliliters of ethanol, adds D-tartrate 0.4 gram (2.64 moles), reflux 0.5 hour, cooling, filter, obtain white solid, again with behind ten times of amount ethyl alcohol recrystallizations 2 times, obtain white solid, free with saturated sodium bicarbonate aqueous solution, obtain 0.29 and digest compound 11A, be i.e. compound (I), yield 26% is surveyed ee value>95%.
PAPER
https://www.tandfonline.com/doi/full/10.1080/00498254.2022.2091494
SYN
https://patents.google.com/patent/US11046701B2/en
SYN
European Journal of Medicinal Chemistry 291 (2025) 117643
Cetagliptin phosphate, developed by CGeneTec, is a DPP-4 inhibitor designed for the treatment of T2DM. In 2024, the NMPA approved cetagliptin phosphate for managing T2DM. As a member of the DPP-4inhibitor, Cetagliptin exerts its effect on glycemic regulation by impeding the breakdown of incretin hormones. This action leads to a glucose-dependent increase in insulin secretion and a concurrent decrease in glucagon levels. Multiple clinical investigations have attested to the effectiveness and safety profile of sitagliptin. In a particular instance, a randomized, double-blind, placebo-controlled Phase 3 study was carried out to assess the use of sitagliptin as a single-agent treatment in patients diagnosed with type 2 diabetes [67]. The study found that cetagliptin significantly reduced HbA1c levels compared to placebo, with a greater proportion of patients achieving target glycemic control.
The treatment was generally well tolerated, with a safety profile comparable to placebo [68,69]. Regarding toxicity, cetagliptin was well tolerated in clinical studies, with no significant increase in adverse effects compared to placebo. No drug-related hypoglycemia was reported,
indicating a favorable safety profile [70].
The synthesis of Cetagliptin, depicted in Scheme 16, initiates with Ceta-001 cyanidation affording Ceta-002, whose hydrogenative reduction yields Ceta-003 [71]. Subsequent amidation constructs Ceta-004,
followed by cyclization rearrangement producing Ceta-005. Hydrogenation delivers Ceta-006, which undergoes coupling with Ceta-007 assembling Ceta-008. Final TFA-mediated deprotection achieves
Cetagliptin. Concurrently, the side route involves Ceta-009 nucleophilic substitution forming Ceta-010. Sequential imine hydrolysis/protection converts Ceta-010 to Ceta-011, whose controlled hydrolysis ultimately delivers Ceta-007
67-70
[67] J. Lu, J. Zhao, D. Xie, J. Ding, Q. Yu, T. Wang, Use of a PK/PD model to select
Cetagliptin dosages for patients with type 2 diabetes in phase 3 trials, Clin.
Pharmacokinet. 63 (2024) 1463–1476.
[68] L. Guo, F. Tian, L. Liu, M. Chen, C. Jiang, S. Li, C. Liu, Y. Zhang, J. Qin, D. Yu,
Y. Zong, W. Dai, Retagliptin as add-on therapy to metformin in Chinese patients
with type 2 diabetes inadequately controlled with metformin: a multicentre,
randomized, double-blind, placebo-controlled, phase 3 trial, Diabetes Obes Metab
26 (2024) 2830–2838.
[69] C. Hu, J. Zheng, J. Miao, F. Liu, T.T. Hu, J.K. Gu, S.Q. Shu, Y. Wang, X.H. Zhu, M.
Z. Liang, [Pharmacokinetics of Phosphate Retagliptin Tabletin in Patients with
Renal Dysfunction], Sichuan Da Xue Xue Bao Yi Xue Ban 49 (2018) 74–80.
[70] A. Cahn, S. Cernea, I. Raz, An update on DPP-4 inhibitors in the management of
type 2 diabetes, Expert Opin Emerg Drugs 21 (2016) 409–419.



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
https://en.cgenetech.com.cn/news/55.html
Since the listing application of a class of innovative drug Cetagliptin independently developed by CGeneTech was accepted by the State Food and Drug Administration, it has received great attention in the industry. Recently, the well-known industry media “Shell News Agency” also took this opportunity to comprehensively sort out the hot track and broad market prospects of domestic DPP-4 inhibitors. This article is shared with you. In the face of the high expectations given by the industry, CGeneTech will continue to run the last “one kilometer” of product launch with a scientific and rigorous attitude.
Diabetes (DM), as a chronic disease, has attracted much attention. Diabetes drugs have become the second largest drug market after tumor drugs, and it is also a place for pharmaceutical enterprises to compete.
With the development of medicine, some new drugs with different mechanism of action from traditional oral hypoglycemic drugs have emerged in recent years. Dipeptidyl peptidase-4 (DPP-4) inhibitor is one of them. At present, there are dozens of DPP-4 inhibitors, which are collectively known as “gliptin drugs”. In the future, the market size of gliptin drugs in China will exceed 30 billion yuan.
Cetagliptin seven-year long run
On February 2, CGeneTech submitted to the National Drug Administration (NMPA) the marketing application (NDA) of Cetagliptin, a DPP-4 inhibitor, which was mainly used to treat type 2 diabetes. This means that the domestic DPP-4 inhibitor market will usher in new members, and the official website of CGeneTech will also publicize the progress of Cetagliptin research and development in the product pipeline for the first time, and the listing has been confirmed.
Cetagliptin is a Class 1 innovative drug independently developed by CGeneTech, and once was its own fist product in its pipeline. It has also experienced a seven-year long run since its launch of research and development, and is about to hit the line successfully.
In 2006, the targeted hypoglycemic drug Sigliptin was approved by the FDA of the United States, which is undoubtedly a major event in the industry. Ten years later, CGeneTech completed the pre-clinical study of head-to-head comparison of Cetagliptin and Xigliptin.
At the beginning of 2018, CGeneTech launched the phase I clinical trial of head-to-head comparison of Cetagliptin and Xigliptin. Among nearly 200 patients in the Phase I clinical trial completed by Cetagliptin, the data showed that when the intake of Cetagliptin reached 50 mg, it was able to achieve the DPP-4 inhibition capacity equivalent to the intake of 100 mg of Xigliptin. Cetagliptin is administered once a day. It can reach the peak within 1 to 2 hours after administration, and has a longer half-life than Sigliptin, which can maintain stable glucose reduction for a longer time.
Diabetes requires long-term medication, and safety is the first factor to be considered when doctors choose drugs when prescribing. In the safety study, the adverse effects of the intake of Cetagliptin on the body of patients were almost undetectable, lower than that of the blank group and Sigliptin group. In addition, although Cetagliptin has a long half-life, there is no accumulation of residual drugs in the body in the phase I clinical trial, which reflects the high selectivity and strong inhibition of Cetagliptin. The beautiful phase I clinical trial data have provided the foundation for the later clinical trial research of Cetagliptin.
In 2019, Cetagliptin was officially approved by the National Drug Evaluation Center to “exempt Phase II clinical trials from Phase III trials”, becoming the first DPP-4 inhibitor in the world to pass the quantitative pharmacological model, exempt Phase II clinical trials, and directly carry out Phase III confirmatory trials, which attracted the attention of experts in the field of diabetes at home and abroad.
In October 2022, the unblinding results of Cetagliptin phase III clinical trial showed that the reduction of glycosylated hemoglobin (HbA1c) in Cetagliptin tablet 50mg group reached the main clinical end point at the end of the 24th week, which was significantly superior to the control group. After 28 weeks, the Cetagliptin 100mg dose group also showed good drug safety, and the incidence of adverse reactions was similar to that of the placebo group. The clinical trial has shown the advantages of halving the dose but the same efficacy as similar products.
In February 2023, the marketing application (NDA) of Cetagliptin has been accepted by NMPA for the treatment of type 2 diabetes.
The approval of Cetagliptin has attracted much attention, which means that CGeneTech will officially participate in the domestic hot track of DPP-4 inhibitors, and the market of 10 billion statins will usher in new members.
DPP-4 inhibitor track is hot
DPP-4 inhibitors play a hypoglycemic role mainly by inhibiting the degradation of glucagon-like peptide-1 (GLP-1) by DPP-4 enzyme, promoting insulin and glucose dependent secretion, and inhibiting glucagon secretion, which can improve β Cell dysfunction does not increase the risk of hypoglycemia and body weight of patients. Moreover, DPP-4 inhibitor is a “mild and versatile”. It is mild, versatile and safe in reducing blood sugar. It is an oral drug that can be combined with various drugs in the whole process of management.
As the current mainstream hypoglycemic drug, DPP-4 inhibitor has become a hot spot in the eyes of major pharmaceutical enterprises. At present, there are five kinds of DPP-4 inhibitors that are taken daily on the market in China: Sigliptin, Viggliptin, Shagliptin, Aggliptin and Liggliptin, and these “five golden flowers” are included in the national health insurance list.
After entering medical insurance, the sales of several products have increased significantly. It is understood that from 2016 to 2022, the annual sales of DPP-4 inhibitors showed a continuous growth trend, with the highest year-on-year growth rate in 2018. In 2021 alone, the domestic sales of DPP-4 inhibitors reached nearly 7 billion yuan.
Sigliptin
Sigliptin is the first oral DPP-4 inhibitor on the market in the world, developed by MSD. It was approved by FDA for listing in October 2006; Sigliptin was approved for listing in China in September 2009; In July 2012, its compound preparation was approved for registration in China.
According to MSD’s annual report, the global market share of Sigliptin has been stable at more than US $3 billion in the past four years, ranking first in the global sales of DPP-4 inhibitors. At present, there are 14 pharmaceutical enterprises in China, including Zhengda Tianqing, Qilu Pharmaceutical, Kelun Pharmaceutical and Zhejiang Pharmaceutical, which have been copied and approved for production.
Viggliptin is the second DPP-4 inhibitor in the world developed by Novartis. In September 2007, Viggliptin was first approved for listing by the European Commission; In August 2011, it was officially approved for listing in China.
According to Novartis annual report, the global sales volume of Vigiletin has fluctuated steadily in recent years, basically maintaining at about 1.1 billion US dollars. The imitative production of Viggliptin in the domestic market is also hot. At present, 18 pharmaceutical enterprises such as Qilu Pharmaceutical, Yangzijiang Pharmaceutical, Jiangsu Haosen Pharmaceutical, Shandong Langnuo Pharmaceutical and Nanjing Shenghe Pharmaceutical have been approved for production. They are worthy of the title of the king of domestic imitative drugs for DPP-4 inhibitors.
Shagliptin was jointly developed by Bristol-Myers Squibb and AstraZeneca. It was approved by FDA for listing in July 2009; In May 2011, Shagliptin was approved for listing in China. Shagliptin’s overseas market share exceeded 20%. At present, there are five pharmaceutical enterprises in China, including Zhengda Tianqing, Qilu Pharmaceutical and Jiangsu Aosaikang Pharmaceutical, whose generic drugs have been approved for production.
Liggliptin was developed by BI. In May 2011, it was approved for listing by the FDA of the United States, and was jointly sold by Berger Ingelheim and Lilly. In March 2013, China approved the import registration of liggliptin. Liggliptin’s overseas market share exceeds 15%. At present, there are 6 pharmaceutical enterprises in China, including Guangdong East Sunshine Pharmaceutical, Yangzijiang Pharmaceutical and Kelun Pharmaceutical, which have been approved for production.
Agiletin
Agiletin was developed by Takeda Pharmaceutical of Japan. Approved for listing in Japan in April 2010; In January 2013, it was approved by the US FDA for listing; In July of the same year, Agiletin obtained the import registration certificate of China. According to the statistics of IQVIA, the sales amount of Agiletin in the Chinese market in 2022 was 52.36 million yuan. At present, 11 pharmaceutical enterprises such as Yabao Pharmaceutical, Ruiyang Pharmaceutical and Guorui Pharmaceutical of the National Pharmaceutical Group have been approved for production.
Throughout the domestic market of DPP-4 inhibitors, the original drugs and generic drugs of the “five golden flowers” are all in the Jianghu. In order to break the competition pattern, pharmaceutical enterprises have also invested in innovative self-research teams.
At present, the research and development of innovative DPP-4 inhibitors is also advancing rapidly. According to the data, in addition to the approval of CGeneTech’s Cetagliptin, many innovative DPP-4 inhibitors (excluding compound preparations) have entered the clinical research stage in China.
TQ-F3083 of Nanjing Shunxin, Shingliptin of Chenxin Pharmaceutical, and Boggliptin of Shandong Baiji Dichang Pharmaceutical are in clinical phase II; Fugliptin of Xinritai, DBPR108 of Shiyao Group, HSK7653 of Hisco and Unigliptin of Yuandong Biological are all in clinical phase III; Hengrui Pharmaceutical’s Retagliptin has submitted its listing application.
Although there are only a few “Ting” who have been approved to market independently developed DPP-4 inhibitors in China, the approval of Cetagliptin will take the lead in ushering in the harvest period of domestic innovative DPP-4 inhibitors, break the monopoly of non-self-developed DPP-4 inhibitors again, and give great confidence to pharmaceutical enterprises engaged in the research and development of DPP-4 inhibitors.
epilogue
The huge market potential of diabetes is like a magnet, attracting pharmaceutical enterprises to participate in the hot domestic track of DPP-4 inhibitors.
As the first oral DPP-4 inhibitor launched in the world and China, Sigliptin has been in the Chinese market for more than ten years, and still dominates the market. According to the Phase I clinical trial study, Cetagliptin has obtained significantly better data than Sigliptin in terms of efficacy, safety, half-life, toxicology and pathology, which will have considerable market persuasion and is expected to help it become a similar Best-in-class product, or change the curve overtaking into a competitive pattern.
Cetagliptin is only one step away from its listing. Not only is CGeneTech full of expectations for it, but also the industry has high expectations. Cetagliptin can be expected in the future, and we also expect more home-made original new “Ting” to come out.
reference material:
1. CGeneTech official website, official account
2. New weapon for treating diabetes (I) – DPP-4 inhibitor, Department of General Medicine, Shenzhen Hospital, University of Hong Kong, December 9, 2020
3. Unique Mechanism, Multi-dimensional Benefits – Mechanism and Clinical Application of DPP-4 Inhibitor, China Medical Forum Endocrinology Today, April 9, 2020
4. DPP-4 inhibitor market may add new force. Can CGeneTech break the “five giants” pattern
5. Market | DPP-4 inhibitor market pattern seen from the withdrawal of the first generic antidiabetic drug from the network of East Sunshine, CPHI Pharmaceutical Online, November 17, 2022
- [1]. Zhou C, et al., Safety, tolerability, pharmacokinetics and pharmacokinetic-pharmacodynamic modeling of cetagliptin in patients with type 2 diabetes mellitus. Front Endocrinol (Lausanne). 2024 Mar 11;15:1359407. [Content Brief][2]. Lu J, et al., In vitro study of the drug-drug interaction potential of cetagliptin and clinical study of pharmacokinetic interaction of cetagliptin and metformin in healthy volunteers. Xenobiotica. 2021 Oct;51(10):1122-1131. [Content Brief]
/////////Cetagliptin, CHINA 2024, APPROVALS 2024, CGeneTec, DIABETES, GTPL13952, CGT 8012,














