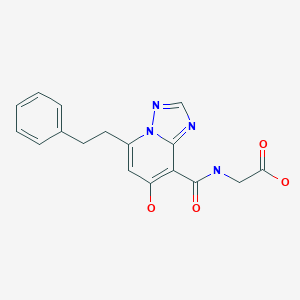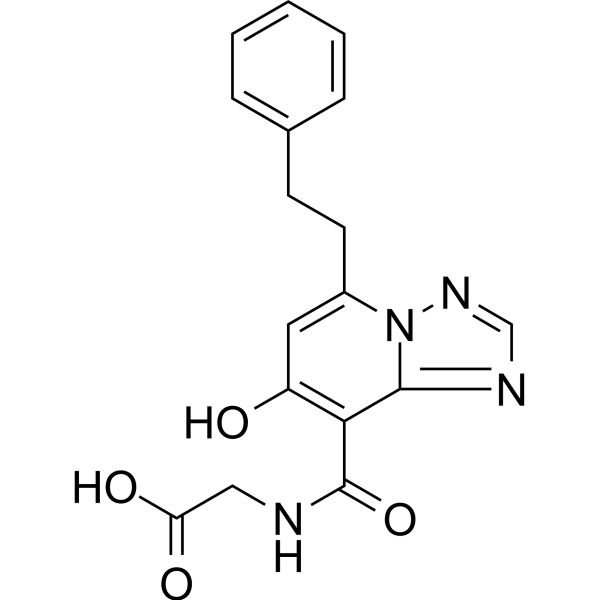
Enarodustat
エナロデュスタット
JTZ 951
| Formula |
C17H16N4O4
|
|---|---|
| CAS |
1262132-81-9
|
| Mol weight |
340.3333
|
PMDA 2020/9/25 APPROVED ENAROY
Anti-anemic, Hypoxia inducible factor-prolyl hydroxylase (HIF-PH) inhibitor
Originator Japan Tobacco
Developer Japan Tobacco; JW Pharmaceutical
Class Acetic acids; Amides; Antianaemics; Pyridones; Small molecules; Triazoles
Mechanism of Action Hypoxia-inducible factor-proline dioxygenase inhibitors
Preregistration Anaemia
27 Dec 2019 Japan Tobacco and SalubrisBio enter into a development and marketing agreement for enarodustat (JTZ 951) in China, Hong Kong, Macau and Taiwan for Anaemia
29 Nov 2019 Preregistration for Anaemia in Japan (PO)
31 Oct 2019 Phase I development in Anaemia is ongoing in USA
Enarodustat is a potent and orally active factor prolyl hydroxylase inhibitor, with an EC50 of 0.22 μM. Enarodustat has the potential for renal anemia treatment
PATENT
WO 2011007856
PAPER
ACS Medicinal Chemistry Letters (2017), 8(12), 1320-1325
https://pubs.acs.org/doi/10.1021/acsmedchemlett.7b00404
Abstract

Inhibition of hypoxia inducible factor prolyl hydroxylase (PHD) represents a promising strategy for the discovery of a next generation treatment for renal anemia. We identified several 5,6-fused ring systems as novel scaffolds of the PHD inhibitor on the basis of pharmacophore analysis. In particular, triazolopyridine derivatives showed potent PHD2 inhibitory activities. Examination of the predominance of the triazolopyridines in potency by electrostatic calculations suggested favorable π–π stacking interactions with Tyr310. Lead optimization to improve the efficacy of erythropoietin release in cells and in vivo by improving cell permeability led to the discovery of JTZ-951 (compound 14), with a 5-phenethyl substituent on the triazolopyridine group, which increased hemoglobin levels with daily oral dosing in rats. Compound 14 was rapidly absorbed after oral administration and disappeared shortly thereafter, which could be advantageous in terms of safety. Compound 14 was selected as a clinical candidate.
https://pubs.acs.org/doi/suppl/10.1021/acsmedchemlett.7b00404/suppl_file/ml7b00404_si_001.pdf
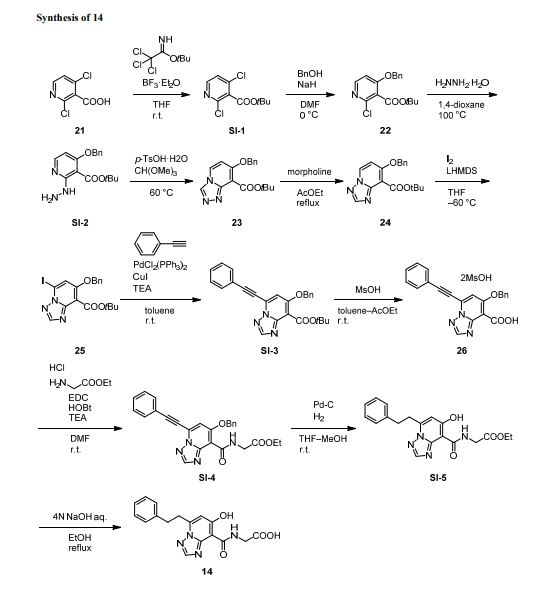
(7-Hydroxy-5-phenethyl-[1,2,4]triazolo[1,5-a]pyridine-8-carbonyl)glycine (14)
To a solution of SI-5 (2.28 g, 6.19 mmol) in EtOH (9.1 mL) was added 2N NaOH aq. (12.4 mL, 24.8 mmol) at room temperature. After stirring at 90 °C for 2 h, 6N HCl aq. (4.1 mL, 24.6 mmol). This was allowed to gradually cool with stirring and crystals were precipitated. The crystals were collected by filtration to give the title compound 14 (2.16 g, 103% yield). 1H NMR (400 MHz, DMSO-D6) δ: 14.22 (s, 1H), 12.98 (br s, 1H), 9.84 (t, J = 5.6 Hz, 1H), 8.58 (s, 1H), 7.33– 7.18 (m, 5H), 6.80 (s, 1H), 4.22 (d, J = 5.6 Hz, 2H), 3.40 (t, J = 7.7 Hz, 2H), 3.12 (t, J = 7.7 Hz, 2H). 13C NMR (126 MHz, CDCl3) δ: 170.28, 167.70, 165.32, 152.95, 148.53, 146.49, 140.05, 128.33, 128.20, 126.17, 106.72, 95.56, 41.00, 31.95, 31.72. HRMS m/z: [M+H]+ calcd for C17H17N4O4, 341.1244; found, 341.1243. Anal. (C17H16N4O4) calcd C 59.99%, H 4.74%, N 16.46%; found C 60.02%, H, 4.78%, N, 16.42%. Melting point: 186 °C Purity: 100.0%.
PATENT
WO 2018097254
PATENT
US 20200017492
/////////////Enarodustat, 2020 APPROVALS, JAPAN 2020, エナロデュスタット , JTZ 951, ENAROY, 2020 APPROVALS,