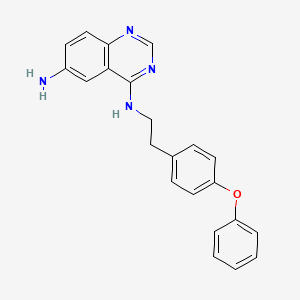
EVP4593; EVP 4593; EVP-4593
| M.Wt | 356.42 | 545380-34-5; QNZ (EVP4593); QNZ; 6-Amino-4-(4-phenoxyphenylethylamino)quinazoline; N4-(4-phenoxyphenethyl)quinazoline-4,6-diamine; | |
|---|---|---|---|
| Formula | C₂₂H₂₀N₄O | ||
| CAS No | 545380-34-5 |
QNZ(EVP4593) is a derivative of 6-aminoquinazoline class that has been previously isolated as an inhibitor of PMA/PHA-induced NF-κB pathway activation in Jurkat cells (IC50= 9 nM).
QNZ(EVP4593) is a derivative of 6-aminoquinazoline class that has been previously isolated as an inhibitor of PMA/PHA-induced NF-κB pathway activation in Jurkat cells (IC50= 9 nM).
IC50 Value: 9 nM [1]
Target: NF-kB signaling
in vitro: The efficacy of EVP4593 was dose-dependent in the range between 100 uM and 400 uM in the fly food. The EVP4593 had no significant effect on climbing performance of HD flies at 50 ?M. The EVP4593 had no toxic effects on Drosophila in the range of concentrations tested in our assays (50 – 400 ?M) [1]. Addition of 300 nM of EVP4593 resulted in strong attenuation of SOC Ca2+ influx in YAC128 MSN neurons. On average the amplitude of SOC Ca2+ entry in YAC128 MSN was reduced from 0.30 ± 0.02 (n = 29) in the presence of DMSO control to 0.11 ± 0.02 (n = 54) in the presence of 300 nM of EVP4593 (p < 0.001).
in vivo:
Paper
Identification of 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine as a novel, highly potent and specific inhibitor of mitochondrial complex I
Abstract
By probing the quinone substrate binding site of mitochondrial complex I with a focused set of quinazoline-based compounds, we identified substitution patterns as being critical for the observed inhibition. The structure activity relationship study also resulted in the discovery of the quinazoline 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine (EVP4593) as a highly potent inhibitor of the multisubunit membrane protein. EVP4593 specifically and effectively reduces the mitochondrial complex I-dependent respiration with no effect on the respiratory chain complexes II–IV. Similar to established Q-site inhibitors, EVP4593 elicits the release of reactive oxygen species at the flavin site of mitochondrial complex I. Recently, EVP4593 was nominated as a lead compound for the treatment of Huntingtons disease. Our results challenge the postulated primary mode-of-action of EVP4593 as an inhibitor of NF-κB pathway activation and/or store-operated calcium influx.
![Graphical abstract Graphical abstract: Identification of 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine as a novel, highly potent and specific inhibitor of mitochondrial complex I](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C6MD00655H)
Abstract
We disclose here a new structural class of low-molecular-weight inhibitors of NF-κB activation that were designed and synthesized by starting from quinazoline derivative 6a. Structure–activity relationship (SAR) studies based on 6a elucidated the structural requirements essential for the inhibitory activity toward NF-κB transcriptional activation, and led to the identification of the 6-amino-4-phenethylaminoquinazoline skeleton as the basic framework. In this series of compounds, 11q, containing the 4-phenoxyphenethyl moiety at the C(4)-position, showed strong inhibitory effects on both NF-κB transcriptional activation and TNF-α production. Furthermore, 11q exhibited an anti-inflammatory effect on carrageenin-induced paw edema in rats.
Compound 11q exhibited a highly inhibitory activity toward NF-κB activation and also showed an anti-inflammatory effect.

- 11q (72 mg, 77% yield):
- mp 168–170 C;
- 1 H NMR (DMSO-d6) d 8.33 (br s, 2H), 7.45 (d, J=8.9 Hz, 1H), 7.40–7.34 (m, 2H), 7.28 (d, J=8.6 Hz, 2H), 7.20–7.07 (m, 3H), 6.98–6.92 (m, 4H), 5.59 (br s, 2H), 3.79–3.72 (m, 2H), 2.95 (t, J=7.3 Hz, 2H);
- MS (TOF) m/z 357 (M + H)+; anal. calcd for C22H20N4O 1.0H2O: C, 70.57; H, 5.65; N, 14.96. Found: C, 70.48; H, 5.60; N, 14.87.
- REF
- Bioorganic & Medicinal Chemistry (2003), 11(18), 3869-3878.
- JP 2004059454
- CN 1709259
- Bioorganic & Medicinal Chemistry Letters (2009), 19(19), 5665-5669
- Journal of Medicinal Chemistry (2014), 57(6), 2247-2257
//////////
C1=CC=C(C=C1)OC2=CC=C(C=C2)CCNC3=NC=NC4=C3C=C(C=C4)N