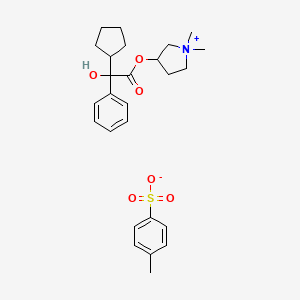
Glycopyrrolate Tosylate
| Molecular Formula: | C26H35NO6S |
|---|---|
| Molecular Weight: | 489.627 g/mol |
(1,1-dimethylpyrrolidin-1-ium-3-yl) 2-cyclopentyl-2-hydroxy-2-phenylacetate;4-methylbenzenesulfonate
CAS 873295-46-6 , C19 H28 N O3 . C7 H7 O3 S, Pyrrolidinium, 3-[(2-cyclopentyl-2-hydroxy-2-phenylacetyl)oxy]-1,1-dimethyl-, 4-methylbenzenesulfonate (1:1)
Glycopyrronium tosylate monohydrate
Molecular Formula, C19-H28-N-O3.C7-H8-O3-S.H2-O, Molecular Weight, 508.6522
https://chem.nlm.nih.gov/chemidplus/structure/1624259-25-1?maxscale=30&width=300&height=300
CAS 1624259-25-1, C19 H28 N O3 . C7 H7 O3 S . H2 O, Pyrrolidinium, 3-[(2-cyclopentyl-2-hydroxy-2-phenylacetyl)oxy]-1,1-dimethyl-, 4-methylbenzenesulfonate, hydrate (1:1:1)
Dermira (Originator)
DRM-04
DRM-04B
- DRM-04 tosylate monohydrate
- DRM04
- DRM04 tosylate
- Glycopyrronium tosylate
- Glycopyrronium tosylate monohydrate
- Glycopyrronium tosylate [USAN]
- UNII-1PVF6JLU7B
- UNII-X2N5209428
In 2018, the product was approved in the U.S. for the treatment of primary axillary hyperhidrosis in adult and pediatric patients 9 years of age and older.
In 2016, Maruho signed an exclusive license agreement with Dermina for product development and marketing in Japan for the treatment of axillary hyperhidrosis.
PATENT
https://patents.google.com/patent/US8558008B2/en
PATENT
https://patents.google.com/patent/US20130211101A1/en
-
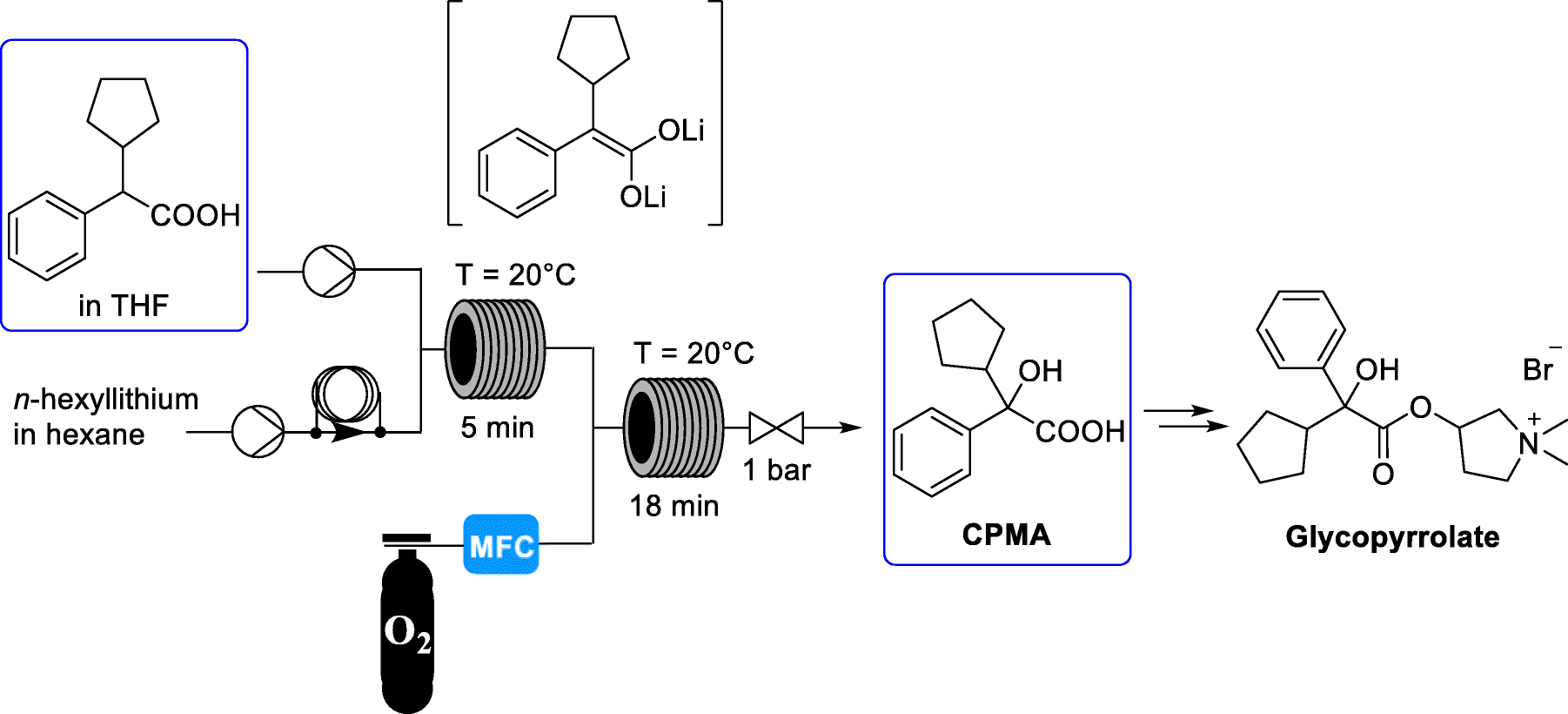
Glycopyrrolate is a quaternary ammonium cation of the muscarinic anticholinergic group. Glycopyrrolate, typically as a bromide salt, has been used in the treatment of a variety of conditions including diarrhea (U.S. Pat. Nos. 6,214,792 and 5,919,760), urinary incontinence (U.S. Pat. Nos. 6,204,285 and 6,063,808), and anxiety (U.S. Pat. No. 5,525,347). Additionally, U.S. Pat. No. 5,976,499 discloses a method for diagnosing cystic fibrosis in a patient by, in part, stimulating sweat production through the injection of a glycopyrrolate solution into a patient. Glycopyrrolate has also been used for the treatment of hyperhidrosis in US 20100276329.
- [0002]
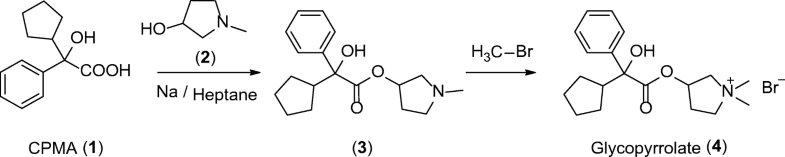
Glycopyrrolate has previously been made available as a bromide salt or an acetate salt. The bromide salt of glycopyrrolate is sold as Rubinol®. The term “glycopyrrolate” as used in the label for Rubinol® refers to the bromide salt which is more formally referred to as glycopyrronium bromide.
- [0124]
In a dark room, silver tosylate (3.5 g) was dissolved in water (˜100 mL) by sonication. The solution was heated to approximately 40° C. and additional water was added (˜15 mL). An equimolar amount of glycopyrrolate bromide (5 g) (mixture of R,S and S,R diastereomers) was added and immediately resulted in a yellow precipitate. The slurry was stirred at approximately 40° C. overnight, and then slowly cooled while stirring to ambient temperature. At ambient temperature, the solids were vacuum filtered and the wet cake was washed three times with approximately 10 mL of water. The mother liquor was collected and filtered two times through a 0.2 μm nylon filter with glass microfiber (GMF). A clear solution was observed after filtration and was lyophilized at approximately −50° C. After 6 days, a mixture of white, needle-like and slightly sticky, glassy solids was observed. Toluene (˜20 mL) was added, and the slurry was briefly sonicated and then stirred at ambient temperature. Additional toluene (˜80 mL) was added for easier stirring, and the mixture was allowed to stand at ambient conditions for 1 day. Solids of glycopyrrolate tosylate were collected by vacuum filtration and vacuum drying at ambient temperature for 1 day.
- Example 6 Glycopyrrolate Tosylate
Example 7 Preparation of Glycopyrrolate Tosylate
- [0125]
A slurry of equimolar amounts of glycopyrrolate acetate and p-toluenesulfonic acid was prepared in isopropanol (1 mL). The mixture was stirred at ambient temperature. Additional isopropanol (0.5 mL) was added to improve stirring, and the mixture was stirred overnight. Solids of glycopyrrolate tosylate were isolated by vacuum filtration and analyzed.
Example 8 Preparation of Glycopyrrolate Tosylate Form D
- [0126]
Glycopyrrolate tosylate (1.0569 g) made from Example 6 was dissolved in 4 mL ACN/H2O (50/50 vol/vol) by sonication. The solution was filtered through 0.2 μm nylon filter into a clean vial. The solvent was allowed to partially evaporate from an open vial under ambient conditions. Further evaporation was subsequently performed under nitrogen gas flow. A gel resulted which was vacuum dried at 40° C. for 1 day. Toluene (5 mL) was added and the mixture was sonicated for approximately 10 minutes causing white solids to precipitate. The mixture was stirred at ambient temperature for 1 day. The solids were isolated by vacuum filtration and the wet cake was washed with approximately 10 mL of toluene. The solids were vacuum dried at ambient temperature for 1 day. After vacuum drying the solids were placed in a vial which remained uncapped and placed inside a relative humidity chamber (˜97%). The chamber was placed inside an oven at 41° C. After 6 days, the solids were analyzed by XRPD showing Form D.
Example 9 Single Crystal Preparation of Form D
- [0127]
Glycopyrrolate tosylate (54.9 mg) made from Example 6 was dissolved in EtOAc/DMF (87/13 vol/vol) at approximately 55° C. at 24 mg/ml. The solution was hot filtered through a 0.2 μm nylon filter into a pre-warmed vial. The vial containing the solution was first placed in a dry ice/acetone bath and then in a freezer (approximately −25 to −10° C.). After 3 days, the solution was re-heated to approximately 50° C. and additional EtOAc was added for 96/4 EtOAc/DMF (vol/vol) at 7 mg/ml. The solution was quickly removed from elevated temperature and placed in the freezer. Solids were isolated by decanting the solvent and drying the solids under ambient conditions.
- [0128]
Single Crystal Data Collection
- [0129]
A colorless chunk of C26H37NO7S [C7H7O3S, C19H28NO3, H2O] having approximate dimensions of 0.23×0.20×0.18 mm, was mounted on a fiber in random orientation. Preliminary examination and data collection were performed with Cu Kα radiation (λ=1.54184 Å) on a Rigaku Rapid II diffractometer equipped with confocal optics. Refinements were performed using SHELX97.
Example 10 Preparation of Dehydrated Form D
- [0130]
A mixture of glycopyrrolate tosylate solids, including Form C and Form D, and a trace amount of silver tosylate was kept over P2O5 at ambient temperature for 18 days. The resulting solids were composed of a mixture of dehydrated Form D with a trace of silver tosylate as shown by XRPD analysis.
Example 11 Preparation of Form C Glycopyrrolate Tosylate
- [0131]
Glycopyrrolate tosylate Form D, containing trace amounts of Form C and silver tosylate, was heated on an Anton Paar TTK 450 stage and XRPD patterns were collected in situ in the range 3.5-26° (2θ). All heating steps were at approximately 10° C./min. The stage was heated in incremental steps of 20° C. from 25 to 125° C. At each step, an XRPD pattern was collected over approximately 4 minutes. The stage was then heated to 135° C. and an XRPD pattern was collected over approximately 16 minutes and after heating further to 145° C., a pattern was collected in approximately 31 minutes. The sample was subsequently cooled to 25° C. at approximately 24° C./min, upon which a final XRPD pattern was collected over approximately 16 min. The XRPD pattern of this final pattern was indexed as Form C.
Example 12 Preparation of Form C Glycopyrrolate Tosylate
- [0132]
Glycopyrrolate tosylate Form D from Example 6 was heated to an approximate temperature in the range 143-149° C. under a continuous nitrogen purge for approximately 3.3 hours. The vial containing the solids was capped, placed on a lab bench and allowed to cool down to room temperature. At room temperature, the vial was placed in a jar containing P2O5. The sample was prepared for XRPD analysis under nitrogen which confirmed production of Form C.
Example 13 Preparation of Form C Glycopyrrolate Tosylate
- [0133]
Glycopyrrolate tosylate (59.5 mg) from Example 6 was dissolved in acetone at approximately 50° C. at 27 mg/ml. The solution was hot filtered through a 0.2 μm nylon filter into a pre-warmed vial. The vial was capped and left on the hot plate which was subsequently turned off to allow the sample to cool slowly to ambient temperature. At ambient temperature the solution was stirred causing white solids to precipitate. The solids were isolated by vacuum filtration and the wet cake was washed with approximately 2 ml of acetone. XRPD analysis resulted in Form C.
Example 14 Amorphous Glycopyrrolate Tosylate
- [0134]
Glycopyrrolate tosylate from Example 6 was melted and cooled repeatedly until the majority of the solids had the appearance of a glass by microscopy. XRPD analysis indicated that the “glassy” sample was observed to be amorphous. A 2.2% weight loss was observed by TGA from 25 to 250° C. of the amorphous glycopyrrolate tosylate. The onset of the glass transition temperature was measured at 11.6° C.
In a dark room, silver tosylate (3.5 g) was dissolved in water (~ 100 mL) by sonication. The solution was heated to approximately 40°C. and additional water was added (-15 mL). An equimolar amount of glycopyrrolate bromide (5 g) (mixture of R,S and S,R diastereomers) was added and imme diately resulted in a yellow precipitate. The slurry was stirred at approximately 40°C. overnight, and then slowly cooled while stirring to ambient temperature. At ambient tempera ture, the solids were vacuum filtered and the wet cake was washed three times with approximately 10 mL of water. The mother liquor was collected and filtered two times through a 0.2 pm nylon filter with glass microfiber (GMF). A clear solution was observed after filtration and was lyophilized at approximately -50°C. After 6 days, a mixture of white, needle-like and slightly sticky, glassy solids was observed. Toluene (-20 mL) was added, and the slurry was briefly sonicated and then stirred at ambient temperature. Additional toluene (-80 mL) was added for easier stirring, and the mix ture was allowed to stand at ambient conditions for 1 day. Solids of glycopyrrolate tosylate were collected by vacuum filtration and vacuum drying at ambient temperature for 1 day. Glycopyrrolate Tosylate.
///////////Glycopyrrolate Tosylate, DRM-04 , DRM-04B , FDA 2018, Qbrexza
CC1=CC=C(C=C1)S(=O)(=O)[O-].C[N+]1(CCC(C1)OC(=O)C(C2CCCC2)(C3=CC=CC=C3)O)C