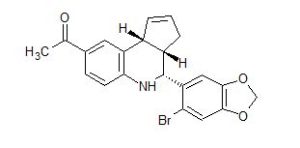
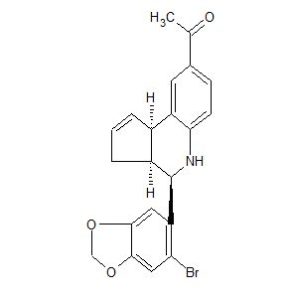
LNS-8801 (SRR G-1)
CAS 925419-53-0
Chemokine receptor-like 2 agonist
1-((3aS,4R,9bR)-4-(6-bromobenzo[d][1,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-1-one
- 1-[(3aS,4R,9bR)-4-(6-Bromo-1,3-benzodioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]ethanone
- G 1
- G 1 (estrogen receptor agonist)
Linnaeus Therapeutics Inc
NOTE 1-((3aR,4S,9bR)-4-(6-bromobenzo[d][1,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-1-one (RSS G-1 or LNS8812)
Novel crystalline forms of an enantiomerically purified LNS-8801 (SRR G-1), or a derivative useful for treating cancer, hearing disorder, depression and myocardial infarction.
Linnaeus Therapeutics is developing LNS-8801, a G-protein coupled estrogen receptor (GPER) agonist, an oral capsule formulation for the treatment of cancer including melanoma, pancreatic ductal adenocarcinoma, non-small cell lung cancer, solid tumor, hematological cancer, advanced cancer and colon carcinoma.
In October 2019, a phase I trial was initiated in patients with locally advanced or metastatic cancer.
PATENT
WO-2020023391
Novel crystalline and amorphous forms of 1-((3aS,4R,9bR)-4-(6-bromobenzo[d][1,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-1-one (here referred to as SRR-G-1 or LNS-8801) and 1-((3aS,4R,9bR)-4-(6-bromobenzo[d][1,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-1-one (RSS G-1 or LNS8812) and its derivative (having chiral purity of >90%; substantially free of its opposite enantiomer; designated as Form A-C) and their co-crystal (eg benzenesulfonic acid) and compositions and combinations comprising them are claimed. Also claimed is their use for treating cancers (disorder expresses GPER), depression, myocardial infarction, osteoporosis, rheumatoid arthritis, insomnia, inflammatory bowel disease, Crohn’s disease, thymic atrophy and hearing disorder and further type-2 diabetes. Further claimed are methods for reducing the likelihood of pregnancy after intercourse, increasing skin pigmentation and skin protection and preventing cancer, reoccurrence of cancer and inhibiting the progression of cancer
Embodiments of the present invention relate to an enantiomerically purified agonist of the G-protein coupled estrogen receptor (GPER), pharmaceutical compositions comprising an enantiomerically purified SRR G-l, or a derivative thereof, and methods of treating disease states and conditions in subjects in need thereof, and methods of treating disease states and conditions mediated through GPER receptors.
[0003] Estrogens mediate multiple complex physiological responses throughout the body. The responses are in turn mediated through the binding of estrogen to receptors. The classical receptors bind steroids, such as estrogen, and are soluble cytoplasmic/nuclear proteins that function as transcription factors. These receptors are known as estrogen receptor alpha and beta (two closely related proteins) that mediate transcriptional activity. GPER is a 7-transmembrane G protein-coupled receptor that also binds to estrogen with high affinity (Kd~6 nM) and mediates rapid cellular responses including cyclic adenosine monophosphate signaling, calcium mobilization and phosphatidylinositol 3,4,5 trisphosphate production.
[0004] Diseases whose development, progression, and or response to therapy, may be influenced by endogenous, and/or pharmacologic activation of GPER signaling include cancer (including the prevention of cancer, prevention of the reoccurrence of cancer, and the inhibition of the progression of cancer; and particularly melanoma, pancreatic, lymphomas, uveal melanoma, non-small cell lung cancer, breast, reproductive and other hormone-dependent cancers, leukemia, colon cancer, prostate, bladder cancer), reproductive (genito-urological) including endometritis, prostatitis, polycystic ovarian syndrome, bladder control, hormone-related disorders, hearing disorders, cardiovascular conditions including hot flashes and profuse sweating, hypertension, stroke, obesity, diabetes, osteoporosis, hematologic diseases, vascular diseases or conditions such as venous thrombosis, atherosclerosis, among numerous others and disorders of the central and peripheral nervous system, including depression, insomnia, anxiety, neuropathy, multiple sclerosis, neurodegenerative disorders such as Parkinson’s disease and Alzheimer’s disease, as well as inflammatory bowel disease, Crohn’s disease, coeliac (celiac) disease and related disorders of the intestine.
Embodiments of the present invention encompass compounds comprising enantiomerically purified G-l and methods of use in the treatment of diseases. G-l is a racemic mixture of the enantiomers l-((3aS,4R,9bR)-4-(6-bromobenzo[if][l,3]dioxol-5-yl)-3a.4.5.9b-tetrahydro-3H-cyclopenta|c |quinolin-8-yl)ethan- 1 -one (henceforth referred to as “SRR G-l” or “LNS8801”) and l-((3aR,4S,9bS)-4-(6-bromobenzo[if][l,3]dioxol-5-yl)-3a.4.5.9b-tetrahydro-3H-cyclopenta|c |quinolin-8-yl)ethan- 1 -one (henceforth referred to as “RSS G-l” or“LNS8812”).
Enantiomerically purified G-l has been purified in favor of its l-((3aS,4R,9bR)-4-(6-bromobenzo[ri][l,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-l-one enantiomer over the corresponding l-((3aR,4S,9bS)-4-(6-bromobenzo[ri][l,3]dioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-l-one enantiomer. Unless specifically described, SRR Gl, or a derivative thereof includes, any physical form, including an amorphous form or any crystalline solid forms such as A, B, C or combinations thereof.
Experimental Section
Scheme 1
[0166] A synthesis of G-l is described in Org. Biomol Client., 2010,8, 2252-2259, which is hereby incorporated by reference, and depicted in Scheme 1. A catalytic amount of Sc(OTf)3 (0.492 g, 1.0 mmol) in anhydrous acetonitrile (2.0 cm3) was added to the mixture of 6-bromopiperonal (2.30 g, 10.0 mmol). / aminoacetophenone (1.30 g, 10.0 mmol) and cyclopentadiene (3.30 g, 50.0 mmol) in acetonitrile (25 cm3). The reaction mixture was stirred at ambient temperature (~23 °C) for 2.0 h. The volatiles were removed in vacuo. The residue was purified by preparative silica gel column chromatography using ethyl acetate-hexanes (10 : 90) to provide G-l (4.03 g, 98%, dr. = 94 : 6) as a white solid. The minor diastereomer was substantially removed by recrystallization to yield a racemic mixure of SRR G-l and RSS G-l.
Example 1: Isolation of the SRR G-l and RSS G-l Enantiomers
[0167] Starting with a highly purified sample of G-l, (±)l-(4-(6-bromobenzo[ri][l,3]dioxol-5-yl)-(3aS*,4R*,9bR*)-tetrahydro-3H-cyclopenta[c]quinolin-8-yl)ethan-l-one, (99.4% purity) purchased from Tocris Bioscience, the material was dissolved in 90: l0:0. l(v/v/v) methyl tert-butyl ether / ethanol / diethyl amine and subjected to preparative chromatography using a column packed with Chrialpak 1A resin. Elution was conducted with 90: l0:0. l(v/v/v) methyl tert-butyl ether / ethanol / diethyl amine and the fractions corresponding to each enantiomer were collected and concentered to a solid. The early eluting enantiomer was determined to be the SRR G-l enantiomer by single crystal x-ray structural analysis.
Example 2: SRR G-l Polymorph Screen
[0168] Starting with SRR G-l prepared according to Example 1, a polymorph screening study was conducted analyzing the solids isolated from slurry of the solid, or from fast and slow evaporation and cooling of solutions (Table 1). Two crystal forms were identified, an anhydrous form designated Form A and mono dichloromethane solvate designated Form B. On exposure to elevated temperature the Form B crystal form desolvates to form the Form C crystal form. Amorphous material was generated from purified SRR G-l by two different methods; quick evaporating a diethyl ether solution of SRR G-l or rotary evaporating from a solution of a dichloromethane solution of SRR G-l.
Table 1
PAPER
Anti-Cancer Agents in Medicinal Chemistry (2019), 19(6), 760-771.
/////////RSS G-1, LNS8812, phase I, Locally advanced, metastatic cancer, LNS-8801, SRR G-1, Linnaeus Therapeutics
CC(=O)c1cc4c(cc1)N[C@@H](c2cc3OCOc3cc2Br)[C@H]5CC=C[C@@H]45