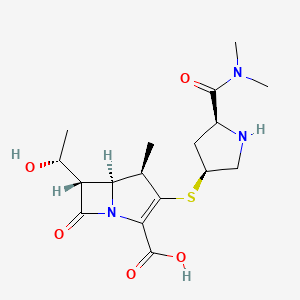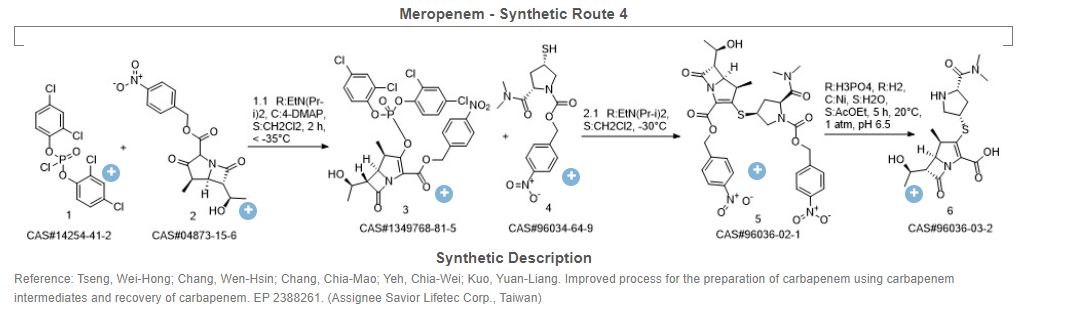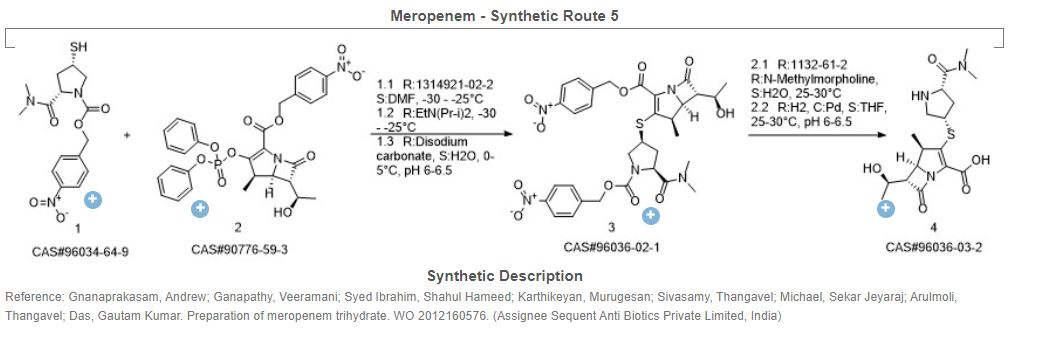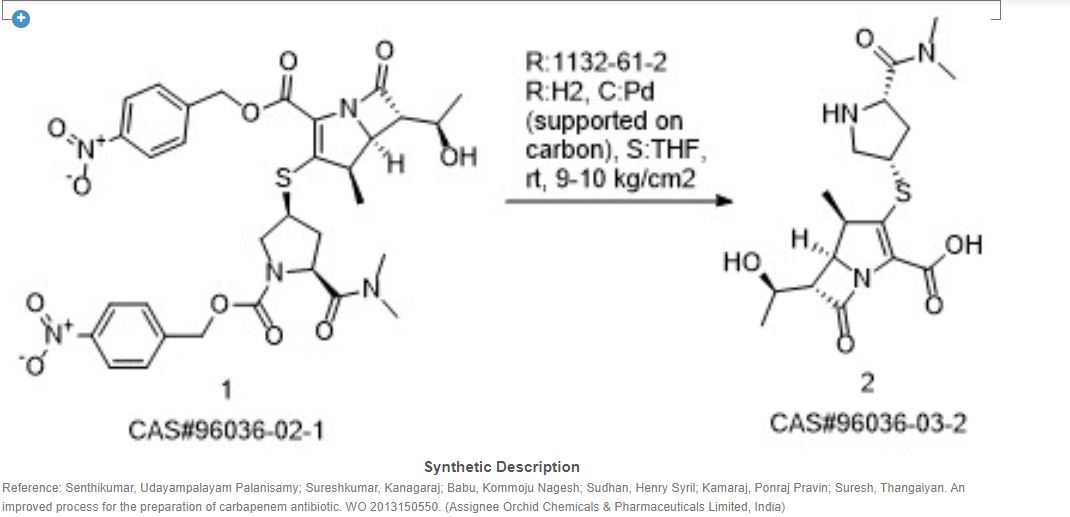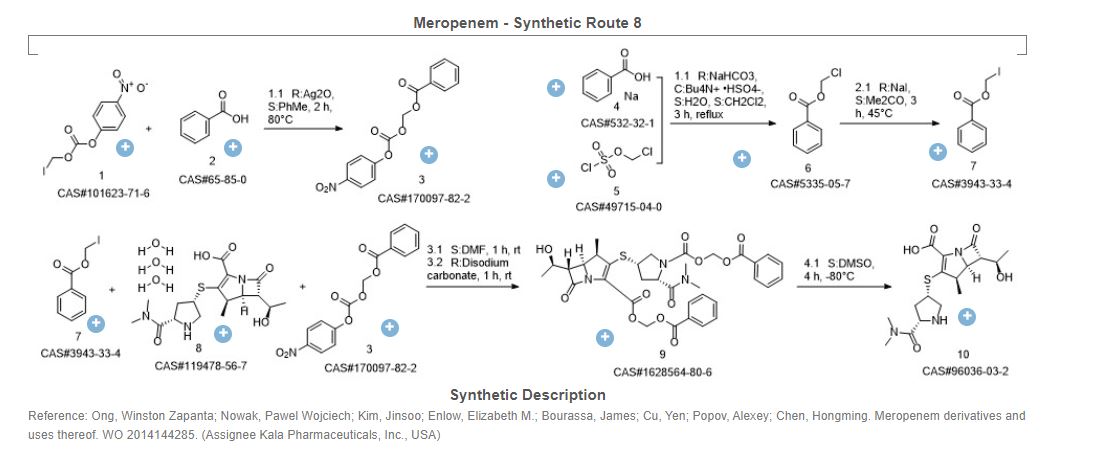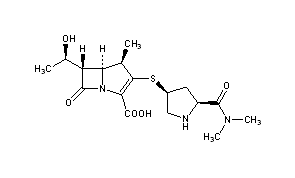
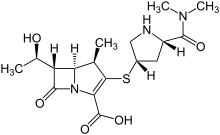
Meropenem
CAS number96036-03-2
IUPAC Name
WeightAverage: 383.463
Monoisotopic: 383.151491615
Chemical FormulaC17H25N3O5S
- Antibiotic SM 7338
- ICI 194660
- SM 7338
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Meropenem sodium | Not Available | 211238-34-5 | UBQRNADYCUXRBD-NACOAMSHSA-N |
| Meropenem trihydrate | FV9J3JU8B1 | 119478-56-7 | CTUAQTBUVLKNDJ-OBZXMJSBSA-N |
International/Other BrandsAronem (ACI) / Aropen (Aristopharma) / Carbanem (Sanofi-Aventis) / Erope (Lincoln) / Fulspec (Acme) / I-penam (Incepta) / Merenz (Admac) / Merofit (FHC) / Meronem (AstraZeneca) / Meronis (Neiss) / Meropen (Swiss Parenterals) / Merotec (Zuventus) / Merrem I.V. (AstraZeneca) / Monan (AstraZeneca) / Ropenem (Drug International) / Zeropenem (Sanofi-Aventis)
Synthesis Reference
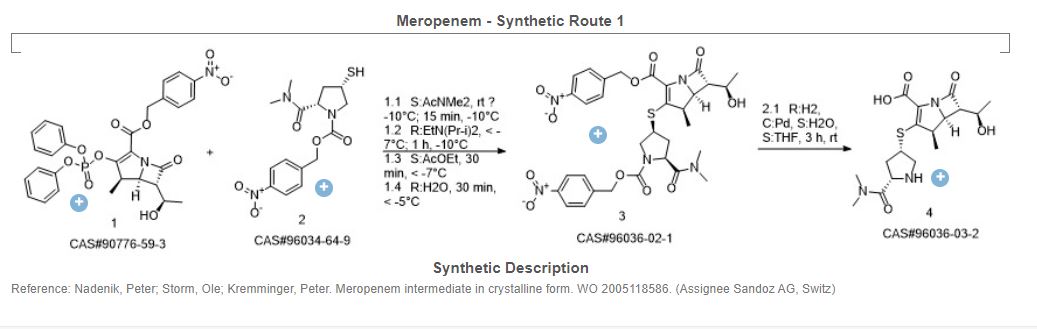
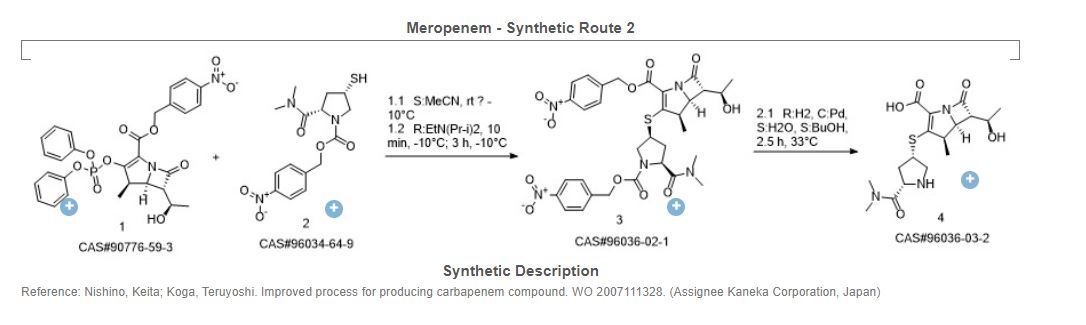
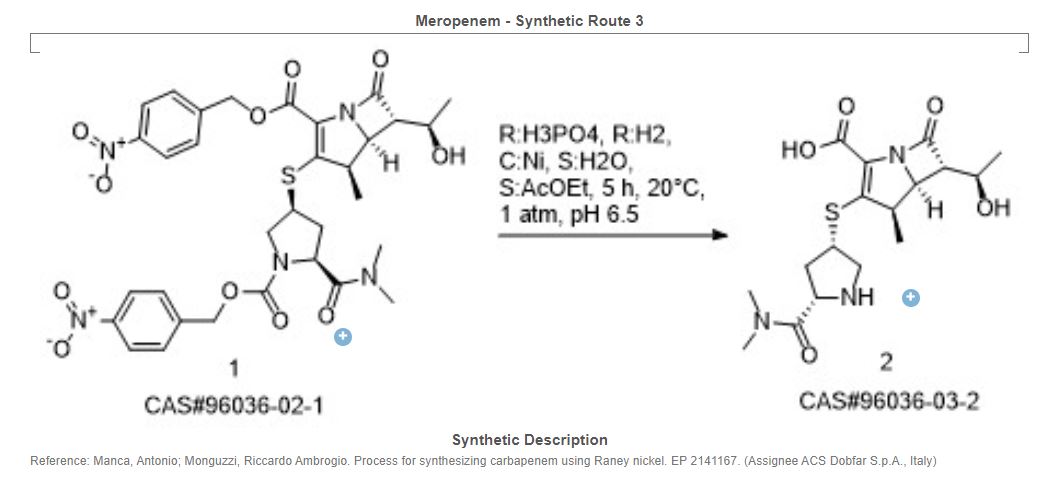
Yoon Seok Song, Sung Woo Park, Yeon Jung Yoon, Hee Kyoon Yoon, Seong Cheol Moon, Byung Goo Lee, Soo Jin Choi, Sun Ah Jun, “METHOD FOR PREPARING MEROPENEM USING ZINC POWDER.” U.S. Patent US20120065392, issued March 15, 2012.
SYN
Carbapenem antibiotic. Prepn: M. Sunagawa et al., EP 126587; M. Sunagawa, US 4943569 (1984, 1990 both to Sumitomo). Structure-activity study: M. Sunagawa et al., J. Antibiot. 43, 519 (1990).

SYN
https://patents.google.com/patent/WO2012062035A1/en

Meropenem, sold under the brandname Merrem among others, is an intravenous β-lactam antibiotic used to treat a variety of bacterial infections.[1] Some of these include meningitis, intra-abdominal infection, pneumonia, sepsis, and anthrax.[1]
Common side effects include nausea, diarrhea, constipation, headache, rash, and pain at the site of injection.[1] Serious side effects include Clostridium difficile infection, seizures, and allergic reactions including anaphylaxis.[1] Those who are allergic to other β-lactam antibiotics are more likely to be allergic to meropenem as well.[1] Use in pregnancy appears to be safe.[1] It is in the carbapenem family of medications.[1] Meropenem usually results in bacterial death through blocking their ability to make a cell wall.[1] It is more resistant to breakdown by β-lactamase producing bacteria.[1]
Meropenem was patented in 1983.[2] It was approved for medical use in the United States in 1996.[1] It is on the World Health Organization’s List of Essential Medicines.[3] The World Health Organization classifies meropenem as critically important for human medicine.[4]
Medical uses
The spectrum of action includes many Gram-positive and Gram-negative bacteria (including Pseudomonas) and anaerobic bacteria. The overall spectrum is similar to that of imipenem, although meropenem is more active against Enterobacteriaceae and less active against Gram-positive bacteria. It works against extended-spectrum β-lactamases, but may be more susceptible to metallo-β-lactamases.[5] Meropenem is frequently given in the treatment of febrile neutropenia. This condition frequently occurs in patients with hematological malignancies and cancer patients receiving anticancer drugs that suppress bone marrow formation. It is approved for complicated skin and skin structure infections, complicated intra-abdominal infections and bacterial meningitis.
In 2017 the FDA granted approval for the combination of meropenem and vaborbactam to treat adults with complicated urinary tract infections.[6]
Administration
Meropenem is administered intravenously as a white crystalline powder to be dissolved in 5% monobasic potassium phosphate solution. Dosing must be adjusted for altered kidney function and for haemofiltration.[7]
As with other ß-lactams antibiotics, the effectiveness of treatment depends on the amount of time during the dosing interval that the meropenem concentration is above the minimum inhibitory concentration for the bacteria causing the infection.[8] For ß-lactams, including meropenem, prolonged intravenous administration is associated with lower mortality than bolus intravenous infusion in persons with whose infections are severe, or caused by bacteria that are less sensitive to meropenem, such as Pseudomonas aeruginosa.[8][9]
Side effects
The most common adverse effects are diarrhea (4.8%), nausea and vomiting (3.6%), injection-site inflammation (2.4%), headache (2.3%), rash (1.9%) and thrombophlebitis (0.9%).[10] Many of these adverse effects were observed in severely ill individuals already taking many medications including vancomycin.[11][12] Meropenem has a reduced potential for seizures in comparison with imipenem. Several cases of severe hypokalemia have been reported.[13][14] Meropenem, like other carbapenems, is a potent inducer of multidrug resistance in bacteria.
Pharmacology
Mechanism of action
Meropenem is bactericidal except against Listeria monocytogenes, where it is bacteriostatic. It inhibits bacterial cell wall synthesis like other β-lactam antibiotics. In contrast to other beta-lactams, it is highly resistant to degradation by β-lactamases or cephalosporinases. In general, resistance arises due to mutations in penicillin-binding proteins, production of metallo-β-lactamases, or resistance to diffusion across the bacterial outer membrane.[10] Unlike imipenem, it is stable to dehydropeptidase-1, so can be given without cilastatin.
In 2016, a synthetic peptide-conjugated PMO (PPMO) was found to inhibit the expression of New Delhi metallo-beta-lactamase, an enzyme that many drug-resistant bacteria use to destroy carbapenems.[15][16]
Society and culture
Trade names
| Country | Name | Maker |
|---|---|---|
| India | Inzapenum | Dream India |
| Aurobindo Pharma | ||
| Penmer | Biocon | |
| Meronir | Nirlife | |
| Merowin | Strides Acrolab | |
| Aktimer | Aktimas Biopharmaceuticals | |
| Neopenem | Neomed | |
| Mexopen | Samarth life sciences | |
| Meropenia | SYZA Health Sciences LLP | |
| Ivpenem | Medicorp Pharmaceuticals | |
| Merofit | ||
| Lykapiper | Lyka Labs | |
| Winmero | Parabolic Drugs | |
| Bangladesh | ||
| Meroject | Eskayef Pharmaceuticals Ltd. | |
| Merocon | Beacon Pharmaceuticals | |
| Indonesia | Merofen | Kalbe |
| Brazil | Zylpen | Aspen Pharma |
| Japan, Korea | Meropen | |
| Australia | Merem | |
| Taiwan | Mepem | |
| Germany | Meronem | |
| Nigeria | Zironem | Lyn-Edge Pharmaceuticals |
| US | Meronem | AstraZeneca |
| … | Merosan | Sanbe Farma |
| Merobat | Interbat | |
| Zwipen | ||
| Carbonem | ||
| Ronem | Opsonin Pharma, BD | |
| Neopenem | ||
| Merocon | Continental | |
| Carnem | Laderly Biotech | |
| Penro | Bosch | |
| Meroza | German Remedies | |
| Merotrol | Lupin) | |
| Meromer | Orchid Chemicals | |
| Mepenox | BioChimico | |
| Meromax | Eurofarma | |
| Ropen | Macter | |
| mirage | adwic | |
| Meropex | Apex Pharma Ltd. | |
| Merostarkyl | Hefny Pharma Group[17] |
References
- ^ Jump up to:a b c d e f g h i j “Meropenem”. The American Society of Health-System Pharmacists. Retrieved 8 December 2017.
- ^ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 497. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2019). Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. hdl:10665/312266. ISBN 9789241515528.
- ^ AHFS Drug Information (2006 ed.). American Society of Health-System Pharmacists. 2006.
- ^ Commissioner, Office of the (24 March 2020). “Press Announcements – FDA approves new antibacterial drug”. www.fda.gov.
- ^ Bilgrami, I; Roberts, JA; Wallis, SC; Thomas, J; Davis, J; Fowler, S; Goldrick, PB; Lipman, J (July 2010). “Meropenem dosing in critically ill patients with sepsis receiving high-volume continuous venovenous hemofiltration” (PDF). Antimicrobial Agents and Chemotherapy. 54 (7): 2974–8. doi:10.1128/AAC.01582-09. PMC 2897321. PMID 20479205.
- ^ Jump up to:a b Yu Z, Pang X, Wu X, Shan C, Jiang S (2018). “Clinical outcomes of prolonged infusion (extended infusion or continuous infusion) versus intermittent bolus of meropenem in severe infection: A meta-analysis”. PLOS ONE. 13 (7): e0201667. Bibcode:2018PLoSO..1301667Y. doi:10.1371/journal.pone.0201667. PMC 6066326. PMID 30059536.
- ^ Vardakas KZ, Voulgaris GL, Maliaros A, Samonis G, Falagas ME (January 2018). “Prolonged versus short-term intravenous infusion of antipseudomonal β-lactams for patients with sepsis: a systematic review and meta-analysis of randomised trials”. Lancet Infect Dis. 18 (1): 108–120. doi:10.1016/S1473-3099(17)30615-1. PMID 29102324.
- ^ Jump up to:a b Mosby’s Drug Consult 2006 (16 ed.). Mosby, Inc. 2006.
- ^ Erden, M; Gulcan, E; Bilen, A; Bilen, Y; Uyanik, A; Keles, M (7 March 2013). “Pancytopenýa and Sepsýs due to Meropenem: A Case Report” (PDF). Tropical Journal of Pharmaceutical Research. 12 (1). doi:10.4314/tjpr.v12i1.21.
- ^ “Meropenem side effects – from FDA reports”. eHealthMe.
- ^ Margolin, L (2004). “Impaired rehabilitation secondary to muscle weakness induced by meropenem”. Clinical Drug Investigation. 24(1): 61–2. doi:10.2165/00044011-200424010-00008. PMID 17516692. S2CID 44484294.
- ^ Bharti, R; Gombar, S; Khanna, AK (2010). “Meropenem in critical care – uncovering the truths behind weaning failure”. Journal of Anaesthesiology Clinical Pharmacology. 26 (1): 99–101.
- ^ “New molecule knocks out superbugs’ immunity to antibiotics”. newatlas.com. 20 January 2017. Retrieved 2017-01-25.
- ^ K., Sully, Erin; L., Geller, Bruce; Lixin, Li; M., Moody, Christina; M., Bailey, Stacey; L., Moore, Amy; Michael, Wong; Patrice, Nordmann; M., Daly, Seth (2016). “Peptide-conjugated phosphorodiamidate morpholino oligomer (PPMO) restores carbapenem susceptibility to NDM-1-positive pathogens in vitro and in vivo”. Journal of Antimicrobial Chemotherapy. 72 (3): 782–790. doi:10.1093/jac/dkw476. PMC 5890718. PMID 27999041.
- ^ “Hefny Pharma Group”. hefnypharmagroup.info. Retrieved 2018-05-22.
External links
- “Meropenem”. Drug Information Portal. U.S. National Library of Medicine.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Merrem, others |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration |
Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% |
| Protein binding | Approximately 2% |
| Elimination half-life | 1 hour |
| Excretion | Renal |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.169.299 |
| Chemical and physical data | |
| Formula | C17H25N3O5S |
| Molar mass | 383.46 g·mol−1 |
| 3D model (JSmol) | |
| (verify) | |
Patent


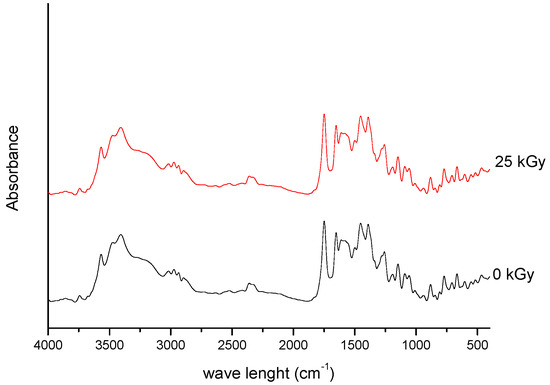
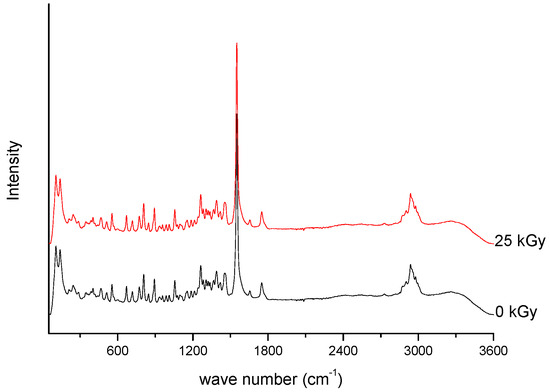
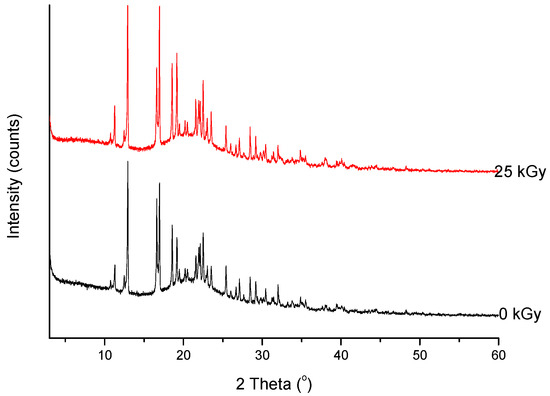
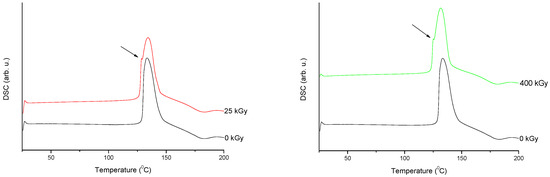
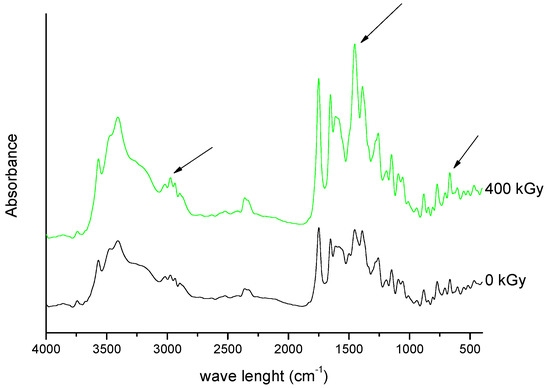
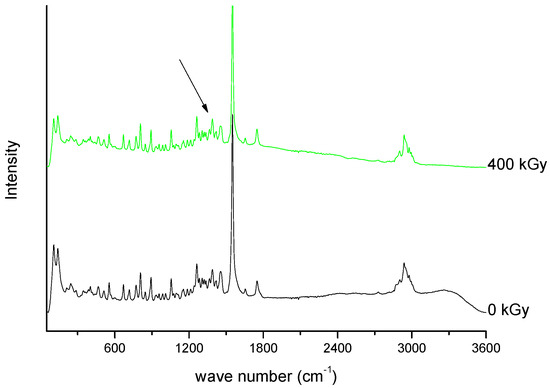
///////////////Meropenem, Merrem, intravenous β-lactam antibiotic, bacterial infections, meningitis, intra-abdominal infection, pneumonia, sepsis, anthrax, Antibiotic SM 7338, ICI 194660, SM 7338, ANTIBACTERIALS
[H][C@]1([C@@H](C)O)C(=O)N2C(C(O)=O)=C(S[C@@H]3CN[C@@H](C3)C(=O)N(C)C)[C@H](C)[C@]12[H]