
MK 0633, SETILEUTON
(-)-enantiomer
910656-27-8 CAS free form
MW 463.3817, C22 H17 F4 N3 O4 FREE FORM
Tosylate cas 1137737-87-1
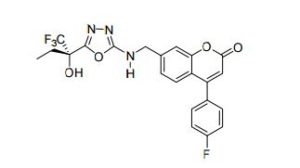
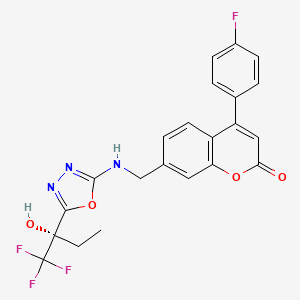
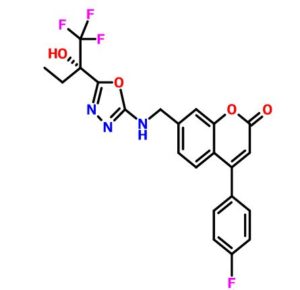
2H-1-Benzopyran-2-one, 4-(4-fluorophenyl)-7-[[[5-[(1S)-1-hydroxy-1-(trifluoromethyl)propyl]-1,3,4-oxadiazol-2-yl]amino]methyl]-
4-(4-Fluorophenyl)-7-[[[5-[(1S)-1-hydroxy-1-(trifluoromethyl)propyl]-1,3,4-oxadiazol-2-yl]amino]methyl]-2H-1-benzopyran-2-one

WO2006099735A1

MK-0633 had been in early clinical development for several indications, including the treatment of chronic obstructive pulmonary disease (COPD), asthma and atherosclerosis
Leukotriene metabolism plays a central role in inflammatory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and atherosclerosis. In particular, the activation of the enzyme 5-lipoxygenase (5-LO) and its associated protein, 5-LO activating protein (FLAP), initiates a cascade that transforms arachidonic acid into inflammatory leukotrienes
Inhibition of leukotriene biosynthesis has been an active area of pharmaceutical research for many years. The leukotrienes constitute a group of locally acting hormones, produced in living systems from arachidonic acid. Leukotrienes are potent contractile and inflammatory mediators deπved by enzymatic oxygenation of arachidonic acid by 5-hρoxygenase. One class of leukotriene biosynthesis inhibitors are those known to act through inhibition of 5 -lipoxygenase (5-LO).
The major leukotrienes are Leukotriene B4 (abbreviated as LTB4), LTC4, LTD4 and LTE4. The biosynthesis of these leukotrienes begins with the action of the enzyme 5-lipoxygenases on arachidonic acid to produce the epoxide known as Leukotriene A4 (LT A4), which is converted to the other leukotπenes by subsequent enzymatic steps. Further details of the biosynthesis as well as the metabolism of the leukotπenes are to be found in the book Leukotrienes and Lipoxygenases, ed. J. Rokach, Elsevier, Amsterdam (1989). The actions of the leukotπenes in living systems and their contπbution to various diseases states are also discussed in the book by Rokach.
In general, 5 -LO inhibitors have been sought for the treatment of allergic rhinitis, asthma and inflammatory conditions including arthπtis. One example of a 5-LO inhibitor is the marketed drug zileuton (ZYLOFT®) which is indicated for the treatment of asthma. More recently, it has been reported that 5-LO may be an important contributor to the atherogenic process; see Mehrabian, M. et al., Circulation Research, 2002 JuI 26, 91(2): 120-126.
Despite significant therapeutic advances in the treatment and prevention of conditions affected by 5-LO inhibition, further treatment options are needed. The instant invention addresses that need by providing novel 5-LO inhibitors which are useful for inhibiting leukotriene biosynthesis.

Synthesis of coumarin intermediate in MK-0633. Reagents and conditions: a) 2.7 M H2SO4 (1 mL/1 mmol), 1.1 equiv. NaNO2, –5 °C, 15 min, 1.5 equiv. KI (1 M H2SO4, 1 mL/0.5 mmol), 0–70 °C, 20 min; b) 1.5 equiv. CuCN, DMF, 110 °C, 24 h, 72 % (over two steps); c) 0.05 equiv. H2SO4, MeOH, 60 °C, 12 h, 81 %; d) 2.5 equiv. 2 M AlMe3, 1.5 equiv. NH(OMe)Me·HCl, THF, room temp., 24 h, 86 %; e) 4.0 equiv. C6H4FMgBr, THF, 0 °C to room temp., 3 h, 74 %; f) toluene, reflux, 24 h, 83 %.
Study of the Chemoselectivity of Grignard Reagent Addition to Substrates Containing Both Nitrile and Weinreb Amide Funct…
Article · Aug 2013 · European Journal of Organic Chemistry
Paper
Synthesis of 4-arylcoumarins via palladium-catalyzed arylation/cyclization of ortho-hydroxylcinnamates with diaryliodonium salts
Tetrahedron Letters (2015), 56, (24), 3809-3812
PAPER
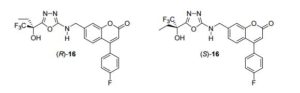
The discovery of novel and selective inhibitors of human 5-lipoxygenase (5-LO) is described. These compounds are potent, orally bioavailable, and active at inhibiting leukotriene biosynthesis in vivo in a dog PK/PD model. A major focus of the optimization process was to reduce affinity for the human ether-a-go-go gene potassium channel while preserving inhibitory potency on 5-LO. These efforts led to the identification of inhibitor (S)-16 (MK-0633, setileuton), a compound selected for clinical development for the treatment of respiratory diseases.
4-(4-fluorophenyl)-7-[({5-[(2R)-1,1,1-trifluoro-2-hydroxybutan-2-yl]- 1,3,4-oxadiazol-2-yl}amino)methyl]-2H-chromen-2-one ((R)-16) and 4-(4- fluorophenyl)-7-[({5-[(2S)-1,1,1-trifluoro-2-hydroxybutan-2-yl]-1,3,4-oxadiazol-2- yl}amino)methyl]-2H-chromen-2-one ((S)-16)
A solution of (±)-4-(4-fluorophenyl)-7-[({5-[1-hydroxy-1-(trifluoromethyl)propyl]-1,3,4- oxadiazol-2-yl}amino)methyl]-2H-chromen-2-one (16) (0.5-0.6 g) in EtOH-Hexane (30:70, ca. 40 mL) was injected on a CHIRALPAK AD preparative (5 cm x 50 cm) HPLC column (eluting with EtOH/Hexane, 30/70 with UV detection at 280 nm). The enantiomers were separated with the fast-eluting enantiomer having a retention time of ~ 34 min for the (-) and the slow-eluting enantiomer having a retention time of ~ 49 min for the (+)-enantiomer.
4-(4-fluorophenyl)-7-[({5-[(2S)-1,1,1-trifluoro-2-hydroxybutan-2-yl]-1,3,4-oxadiazol- 2-yl}amino)methyl]-2H-chromen-2-one ((S)-16, MK-0633, setileuton):
A mixture of oxadiazole (S)-35 (41.9 g, 156 mmol) and aldehyde 25 (39.2 g, 186 mmol) in toluene (2 L) with 10% of pyridinium p-toluenesulfonate was refluxed overnight. The system was equipped with a Dean-Stark apparatus to collect water. The solvent was removed and the crude oil [1 H NMR (400 MHz, acetone-d6): δ 9.33 (s, 1H, imine)] obtained was diluted in THF (600 mL) and EtOH (100 mL). To this solution was added at 0 o C NaBH4 (7.2 g) portionwise. After 1 h of stirring, aqueous ammonium acetate was added. The mixture was extracted with ethyl acetate. The combined organic fractions were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified on silica gel (toluene/EtOAc; 1:1) to give the title compound (39.4 g, 54%).
FREE FORM
1 H NMR (400 MHz, acetone-d6): δ 7.65 (m, 2H), 7.50 (m, 3H), 7.38 (m, 3H), 6.35 (s, 1H), 6.06 (s, 1H), 4.70 (m, 2H), 2.21 (m, 1H), 2.11 (m, 1H), 0.98 (t, 3H);
HRMS calcd for C22H17F4N3O4 [MH+]: 464.1233; found: 464.1228.
PATENT
CLIP
J. Org. Chem. 2010, 75, 4154−4160
Synthesis of a 5-Lipoxygenase Inhibitor
Practical, chromatography-free syntheses of 5-lipoxygenase inhibitor MK-0633 p-toluenesulfonate (1) are described. The first route used an asymmetric zincate addition to ethyl 2,2,2-trifluoropyruvate followed by 1,3,4-oxadiazole formation and reductive amination as key steps. An improved second route features an inexpensive diastereomeric salt resolution of vinyl hydroxy-acid 22 followed by a robust end-game featuring a through-process hydrazide acylation/1,3,4-oxadiazole ring closure/salt formation sequence to afford MK-0633 p-toluenesulfonate (1).

Leukotriene metabolism plays a central role in inflammatory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and atherosclerosis. In particular, the activation of the enzyme 5-lipoxygenase (5-LO) and its associated protein, 5-LO activating protein (FLAP), initiates a cascade that transforms arachidonic acid into inflammatory leukotrienes. Consequently, compounds that can inhibit 5-LO have potential as new treatments for the conditions listed above. Gosselin and co-workers at Merck describe two routes towards one such compound (MK-0633) brought forward as a development candidate at Merck ( J. Org. Chem. 2010, 75, 4154−4160). The first route used an asymmetric zincate addition to ethyl 2,2,2-trifluoropyruvate followed by 1,3,4-oxadiazole formation and reductive amination as key steps. An improved second route (shown here) featured an inexpensive diastereomeric salt resolution of a vinyl hydroxy-acid followed by a through-process hydrazide acylation/1,3,4-oxadiazole ring-closure/salt-formation sequence to afford MK-0633 as the p-toluenesulfonate salt.
Francis Gosselin†, Robert A. Britton†, Ian W. Davies‡, Sarah J. Dolman†, Danny Gauvreau†, R. Scott Hoerrner‡, Gregory Hughes†, Jacob Janey‡, Stephen Lau†, Carmela Molinaro†, Christian Nadeau†, Paul D. O’Shea†, Michael Palucki‡ and Rick Sidler‡
† Department of Process Research, Merck Frosst Centre for Therapeutic Research, 16711 Route Transcanadienne, Kirkland, Québec, Canada H9H 3L1
‡ Department of Process Research, Merck Research Laboratories, P.O. Box 2000, Rahway, New Jersey 07065
J. Org. Chem., 2010, 75 (12), pp 4154–4160
DOI: 10.1021/jo100561u
MK-0633 tosylate salt (1) was obtained as a white solid (6.64 kg, 91.4% yield): mp 164−165 °C;
[α]20D − 0.86 (c 10.0, EtOH);
1H NMR (500 MHz, DMSO-d6) δ 8.58 (1 H, t, J = 6.2 Hz), 7.62 (2 H, dd, J = 8.3, 5.4 Hz), 7.49 (2 H, d, J = 7.8 Hz), 7.47−7.38 (4 H, m), 7.33 (1 H, d, J = 8.3 Hz), 7.13 (2 H, d, J = 7.7 Hz), 6.44 (1 H, s), 4.53 (2 H, d, J = 5.6 Hz), 2.30 (3 H, s), 2.17−2.05 (1 H, m), 2.03−1.93 (1 H, m), 0.90 (3 H, t, J = 7.37 Hz);
13C NMR (125 MHz, DMSO-d6) δ 164.1, 162.9 (d, J = 246.8 Hz), 159.6, 156.1, 153.7, 153.6, 145.5, 143.7, 137.7, 131.1 (d, J = 3.5 Hz), 130.9 (d, J = 8.7 Hz), 128.1, 126.8, 125.4, 124.5 (q, J = 286.6 Hz), 123.5, 117.4, 115.9 (d, J = 22.0 Hz), 115.4, 114.7, 73.7 (q, J = 28.6 Hz), 45.4, 26.1, 20.8, 7.0;
19F NMR (375 MHz, DMSO-d6) δ −79.7, −113.1;
HRMS calcd for C22H18F4N3O4 [M + H] 464.1228, found 464.1246.
IR (cm−1, NaCl thin film) 3324, 3010, 2977, 1735, 1716, 1618, 1510, 1428, 1215, 1178.
HPLC analysis: eclipse XDB-phenyl column 4.6 mm × 15 cm (0.1% aq H3PO4/CH3CN 65:35 to 10:90 over 50 min, 1.0 mL/min, 210 nm, 25 °C); MK-0633 (1) tR = 16.86 min. Chiral HPLC analysis: Chiralpak AD-H column 4.6 mm × 25 cm (EtOH/hexane 60:40, hold 15 min, 0.5 mL/min, 300 nm, 30 °C); (S)-enantiomer tR = 9.5 min; (R)-enantiomer tR = 11.5 min.


| Patent ID |
Patent Title |
Submitted Date |
Granted Date |
| US2016193168 |
Treatment of Pulmonary Arterial Hypertension with Leukotriene Inhibitors |
2015-11-30 |
2016-07-07 |
| US2013251787 |
Treatment of Pulmonary Hypertension with Leukotriene Inhibitors |
2013-03-15 |
2013-09-26 |
| US7915298 |
Compounds and methods for leukotriene biosynthesis inhibition |
2009-04-02 |
2011-03-29 |
| US2009227638 |
Novel Pharmaceutical Compounds |
2009-09-10 |
|
| US7553973 |
Pharmaceutical compounds |
2007-06-28 |
2009-06-30 |
| US2009030048 |
Novel pharmaceutical compounds |
2009-01-29 |
/////////////MK 0633, PHASE 2
CCC(C1=NN=C(O1)NCC2=CC3=C(C=C2)C(=CC(=O)O3)C4=CC=C(C=C4)F)(C(F)(F)F)O