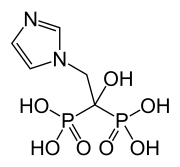
Zoledronic acid
- CGP-42446, ZOL-446
- ATC:M05BA08
- MW:272.09 g/mol
- CAS-RN:118072-93-8
Derivatives
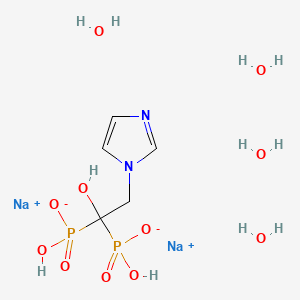
Disodium salt tetrahydrate
- Formula:C5H8N2Na2O7P2 • 4H2O
- MW:388.11 g/mol
- CAS-RN:165800-07-7
Trisodium salt hydrate
- Formula:C5H7N2Na3O7P2 • 2/5H2O
- MW:1726.21 g/mol
- CAS-RN:165800-08-8
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Zoledronate disodium | 7D7GS1SA24 | 165800-07-7 | IEJZOPBVBXAOBH-UHFFFAOYSA-L |
| Zoledronate trisodium | ARL915IH66 | 165800-08-8 | Not applicable |
| Zoledronic acid hemipentahydrate | 1K9U67HDID | Not Available | AZZILOGHCMYHQY-UHFFFAOYSA-N |
| Zoledronic acid monohydrate | 6XC1PAD3KF | 165800-06-6 | FUXFIVRTGHOMSO-UHFFFAOYSA-N |
Zoledronate (zoledronic acid, marketed by Novartis under the trade names Zometa and Reclast) is a bisphosphonate. Zometa is used to prevent skeletal fractures in patients with cancers such as multiple myeloma and prostate cancer. It can also be used to treat hypercalcemia of malignancy and can be helpful for treating pain from bone metastases.
An annual dose of Zoledronate may also prevent recurring fractures in patients with a previous hip fracture.
Zoledronate is a single 5 mg infusion for the treatment of Paget’s disease of bone. In 2007, the FDA also approved Reclast for the treatment of postmenopausal osteoporosis.
Zoledronic acid, also known as zoledronate, is a medication used to treat a number of bone diseases.[1] This include osteoporosis, high blood calcium due to cancer, bone breakdown due to cancer, and Paget’s disease of bone.[1] It is given by injection into a vein.[1]
Common side effects include fever, joint pain, high blood pressure, diarrhea, and feeling tired.[1] Serious side effects may include kidney problems, low blood calcium, and osteonecrosis of the jaw.[1] Use during pregnancy may result in harm to the baby.[1] It is in the bisphosphonate family of medications.[1] It works by blocking the activity of osteoclast cells and thus decreases the breakdown of bone.[1]
Zoledronic acid was approved for medical use in the United States in 2001.[1] It is on the World Health Organization’s List of Essential Medicines, the most effective and safe medicines needed in a health system.[3] The wholesale cost in the developing world is between 5.73 USD and 26.80 USD per vial.[4] In the United Kingdom, as of 2015, a dose costs the NHS about 220 pounds.[5]
Medical uses
Bone complications of cancer
Zoledronic acid is used to prevent skeletalfractures in patients with cancers such as multiple myeloma and prostate cancer, as well as for treating osteoporosis.[6] It can also be used to treat hypercalcemia of malignancy and can be helpful for treating pain from bone metastases.[7]
It can be given at home rather than in hospital. Such use has shown safety and quality-of-life benefits in people with breast cancer and bone metastases.[8]
Osteoporosis
Zoledronic acid may be given as a 5 mg infusion once per year for treatment of osteoporosis in men and post-menopausal women at increased risk of fracture.[9]
In 2007, the U.S. Food and Drug Administration (FDA) also approved it for the treatment of postmenopausal osteoporosis.[10][11]
Paget’s disease
A single 5 mg dose of zoledronic acid is used for the treatment of Paget’s disease.[medical citation needed][12]
Contraindications
- Poor renal function (e.g. CrCl<30 mL/min)[13]
- Hypocalcaemia
- Pregnancy
- Paralysis
Side effects
Side effects can include fatigue, anemia, muscle aches, fever, and/or swelling in the feet or legs. Flu-like symptoms are common after the first infusion, although not subsequent infusions, and are thought to occur because of its potential to activate human γδ T cells(gamma/delta T cells).
Kidneys
There is a risk of severe renal impairment. Appropriate hydration is important prior to administration, as is adequate calcium and vitamin D intake prior to Aclasta therapy in patients with preexisting hypocalcaemia, and for ten days following Aclasta in patients with Paget’s disease of the bone. Monitoring for other mineral metabolism disorders and the avoidance of invasive dental procedures for those who develop osteonecrosis of the jaw is recommended.[14]
Zoledronate is rapidly processed via the kidneys; consequently its administration is not recommended for patients with reduced renal function or kidney disease.[15] Some cases of acute renal failure either requiring dialysis or having a fatal outcome following Reclast use have been reported to the U.S. Food and Drug Administration (FDA).[16] This assessment was confirmed by the European Medicines Agency (EMA), whose Committee for Medicinal Products for Human Use (CHMP) specified new contraindications for the medication on 15 December 2011, which include hypocalcaemia and severe renal impairment with a creatinine clearance of less than 35 ml/min.[17]
Bone
A rare complication that has been recently observed in cancer patients being treated with bisphosphonates is osteonecrosis of the jaw. This has mainly been seen in patients with multiple myeloma treated with zoledronate who have had dental extractions.[18]
Atypical fractures : After approving the drug on 8 July 2009, the European Medicines Agency conducted a class review of all bisphosphonates, including Zoledronate, after several cases of atypical fractures were reported.[19] In 2008, the EMA’s Pharmacovigilance Working Party (PhVWP) noted that alendronic acid was associated with an increased risk of atypical fracture of the femur that developed with low or no trauma. In April 2010, the PhVWP noted that further data from both the published literature and post-marketing reports were now available which suggested that atypical stress fractures of the femur may be a class effect. The European Medicines Agency then reviewed all case reports of stress fractures in patients treated with bisphosphonates, relevant data from the published literature, and data provided by the companies which market bisphosphonates. The Agency recommended that doctors who prescribe bisphosphonate-containing medicines should be aware that atypical fractures may occur rarely in the femur, especially after long-term use, and that doctors who are prescribing these medicines for the prevention or treatment of osteoporosis should regularly review the need for continued treatment, especially after five or more years of use.[19]
Mechanism of action
Zoledronic acid slows down bone resorption, allowing the bone-forming cells time to rebuild normal bone and allowing bone remodeling.[20]
Research
Zoledronic acid has been found to have a direct antitumor effect and to synergistically augment the effects of other antitumor agents in osteosarcoma cells.[21]
Zoledronate has shown significant benefits versus placebo over three years, with a reduced number of vertebral fractures and improved markers of bone density.[22][11] An annual dose of zoledronic acid may also prevent recurring fractures in patients with a previous hip fracture.[9]
Zoledronate also attenuates accumulation of DNA damage in mesenchymal stem cells and protects their function.[23] Given this characteristic, its potential to affect conditions arising from stem-cell dysfunction makes it a promising medicine for a range of age-related diseases[24]
With hormone therapy for breast cancer
An increase in disease-free survival (DFS) was found in the ABCSG-12 trial, in which 1,803 premenopausal women with endocrine-responsive early breast cancer received anastrozole with zoledronic acid.[25] A retrospective analysis of the AZURE trial data revealed a DFS survival advantage, particularly where estrogen had been reduced.[26]
In a meta-analysis of trials where upfront zoledronic acid was given to prevent aromatase inhibitor-associated bone loss, active cancer recurrence appeared to be reduced.[27]
As of 2010 “The results of clinical studies of adjuvant treatment on early-stage hormone-receptor-positive breast-cancer patients under hormonal treatment – especially with the bisphosphonate zoledronic acid – caused excitement because they demonstrated an additive effect on decreasing disease relapses at bone or other sites. A number of clinical and in vitro and in vivo preclinical studies, which are either ongoing or have just ended, are investigating the mechanism of action and antitumoral activity of bisphosphonates.”[28]
A 2010 review concluded that “adding zoledronic acid 4 mg intravenously every 6 months to endocrine therapy in premenopausal women with hormone receptor-positive early breast cancer … is cost-effective from a US health care system perspective”.[29]
Synthesis
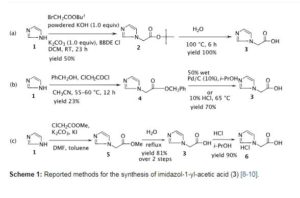
PAPER
J Med Chem 2002,45(17),3721
https://pubs.acs.org/doi/10.1021/jm020819i
Highly Potent Geminal Bisphosphonates. From Pamidronate Disodium (Aredia) to Zoledronic Acid (Zometa)

Bisphosphonates (BPs) are pyrophosphate analogues in which the oxygen in P−O−P has been replaced by a carbon, resulting in a metabolically stable P−C−P structure. Pamidronate (1b, Novartis), a second-generation BP, was the starting point for extensive SAR studies. Small changes of the structure of pamidronate lead to marked improvements of the inhibition of osteoclastic resorption potency. Alendronate (1c, MSD), with an extra methylene group in the N-alkyl chain, and olpadronate (1h, Gador), the N,N-dimethyl analogue, are about 10 times more potent than pamidronate. Extending one of the N-methyl groups of olpadronate to a pentyl substituent leads to ibandronate (1k, Roche, Boehringer-Mannheim), which is the most potent close analogue of pamidronate. Even slightly better antiresorptive potency is achieved with derivatives having a phenyl group linked via a short aliphatic tether of three to four atoms to nitrogen, the second substituent being preferentially a methyl group (e.g., 4g, 4j, 5d, or 5r). The most potent BPs are found in the series containing a heteroaromatic moiety (with at least one nitrogen atom), which is linked via a single methylene group to the geminal bisphosphonate unit. Zoledronic acid (6i), the most potent derivative, has an ED50 of 0.07 mg/kg in the TPTX in vivo assay after sc administration. It not only shows by far the highest therapeutic ratio when comparing resorption inhibition with undesired inhibition of bone mineralization but also exhibits superior renal tolerability. Zoledronic acid (6i) has thus been selected for clinical development under the registered trade name Zometa. The results of the clinical trials indicate that low doses are both efficacious and safe for the treatment of tumor-induced hypercalcemia, Paget’s disease of bone, osteolytic metastases, and postmenopausal osteoporosis.
SYN 1
AU 8781453; EP 0275821; JP 1988150291; US 4939130
Zoledronate sodium can be prepared by reaction of 2-(1-imidazolyl)acetic acid hydrochloride (I) with PCl3, with optional presence of phosphoric acid, in refluxing chlorobenzene, followed by hydrolysis with refluxing 9N hydrochloric acid and final formation of the sodium salt by treatment with aqueous NaOH.
SYN
PAPER
https://www.sciencedirect.com/science/article/pii/S0969804311006385

Clip
https://link.springer.com/article/10.1007/s11094-015-1205-0
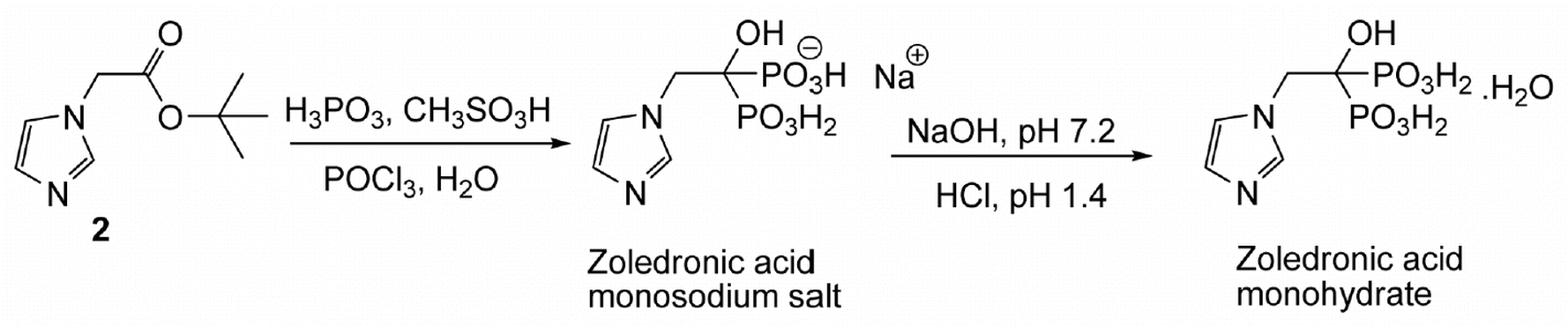
A One-Pot and Efficient Synthesis of Zoledronic Acid Starting from Tert-butyl Imidazol-1-yl Acetate
A one-pot synthesis of zoledronic acid in high yield is described. The procedure involves a non-aqueous ester cleavage of the tert-butyl imidazol-1-yl acetate under dry conditions in the presence of methanesulfonic acid as solubilizer and chlorobenzene as solvent to afford in situthe corresponding imidazolium methanesulfonate salt which yields zoledronic acid upon reaction with phosphoric acid and phosphorus oxychloride. A possible chemical mechanism for the synthesis of this acid is described.

Paper
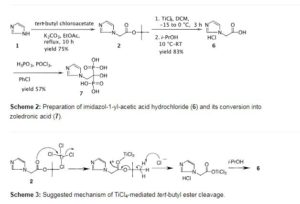
https://www.beilstein-journals.org/bjoc/articles/4/42
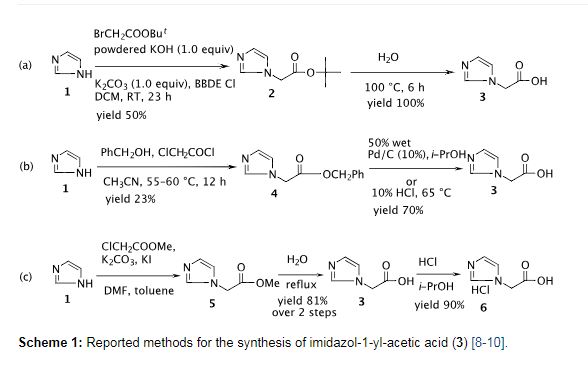
Preparation of imidazol-1-yl-acetic acid tert-butyl ester (2)
To a solution of imidazole (10.0 g, 0.15 mol) in ethyl acetate (160 mL) was added powdered K2CO3 (29.0 g, 0.21 mol) followed by tert-butyl chloroacetate (25.7 mL, 0.18 mol) at room temperature and the mixture was refluxed for 10.0 h. After completion of the reaction as indicated by TLC (10% MeOH/CHCl3, I2 active), the reaction mass was quenched with cold water (80 mL) and the ethyl acetate layer was separated. The aqueous layer was extracted with ethyl acetate (2 × 80 mL) and the combined ethyl acetate layers were washed with brine, dried with anhydrous sodium sulfate and then concentrated under vacuum. The resulting solid was stirred with hexane (50 mL) at RT, filtered and washed with hexane (2 × 20 mL) to afford the title compound as an off-white solid (20.0 g, 75%). mp: 111.3–113.2 °C (Lit [10]: 111–113 °C). IR (cm−1): 3458, 3132, 3115, 2999, 2981, 2884, 1740, 1508, 1380, 1288, 1236, 1154, 1079, 908, 855, 819, 745, 662, 583; 1H NMR (300 MHz, CDCl3) δ 1.47 (s, 9H), 4.58 (s, 2H), 6.94 (s, 1H), 7.09 (s, 1H), 7.49 (s, 1H); 13C NMR (75 MHz, CDCl3) δ 27.7, 48.6, 82.9, 119.8, 129.2, 137.7, 166.3; MS (m/z) 183.0 [M+1, 100%], 127.0.
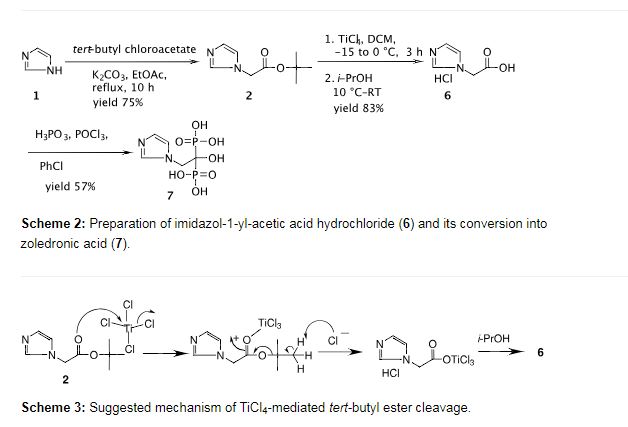
Preparation of imidazol-1-yl-acetic acid hydrochloride (6)
To a solution of imidazol-1-yl-acetic acid tert-butyl ester (2) (10.0 g, 0.05 mol) in dichloromethane (100 mL) was added titanium tetrachloride (8.0 mL, 0.07 mol) dropwise slowly at −15 to −10 °C over 1 h and the mixture was stirred at −5 to 0 °C for 2 h. Isopropyl alcohol (25 mL) was added at 0 to −10 °C over 0.5 h and the reaction mass was stirred at room temperature for 0.5 h. Additional isopropyl alcohol (125 mL) was added dropwise at room temperature over 0.5 h and the mixture was stirred for 1 h. Dichloromethane was distilled out under a low vacuum and the resulting crystalline solid precipitated was filtered to afford the title compound as an off-white crystalline solid (7.4 g, 83%). mp 200.3–202.3 °C; IR (cm−1): 3175, 3125, 3064, 2945, 2869, 2524, 2510, 1732, 1581, 1547, 1403, 1223, 1193, 1081, 780, 650; 1H NMR (300 MHz, D2O + 3-(trimethylsilyl)propionic acid sodium salt) δ 5.1 (s, 3H, -CH2– + HCl), 7.5 (br s, 2H), 8.7 (s, 1H); 13C NMR (75 MHz, D2O + 3-(trimethylsilyl)propionic acid sodium salt) 52.7, 122.4, 125.9, 138.8, 172.8; MS (m/z) 127.0 [M+1, 100%]; HCl-content: found 21.8% (along with 3.25% moisture), calcd 22.43% for C5H6N2O2·HCl.
Preparation of zoledronic acid (7)
To a suspension of imidazol-1-yl-acetic acid hydrochloride (6) (7.0 g, 0.043 mol) and phosphorous acid (9.5 g, 0.116 mol) in chlorobenzene (50 mL) was added phosphorous oxychloride (9.6 ml, 0.103 mol) at 80–85 °C over a period of 2 h then heated to 90–95 °C for 2.5 h. The reaction mass was cooled to 60–65 °C and water (100 mL) was added at the same temperature. The aqueous layer was separated, collected and refluxed for 18 h. It was then cooled to room temperature and diluted with methanol (140 mL). The mixture was cooled to 0–5 °C and stirred for 3 h. The precipitated solid was filtered, washed with cold water followed by methanol and then dried under vacuum at 60 °C for 12 h to afford the title compound (6.6 g, 57% yield) as a white solid; mp 237–239 °C (lit [1] 239 °C with decomposition).
PATENT
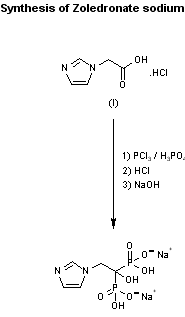
https://patents.google.com/patent/CN104610357A/en
Sodium Zoledronic (Zoledronate sodium, I), chemical name [1-yl light -2- (lH- imidazol-1-yl) ethylidene] bisphosphonic acid monosodium salt monohydrate, is by the Novartis (Novartis) developed imidazole heterocyclic bisphosphonates, belongs to the third generation of bisphosphonates bisphosphonate drugs, in October 2000, first marketed in Canada. Subsequently approved in the European Union, the United States more than 80 countries or regions, trade name Zometa, for the treatment of hypercalcemia of malignancy (HCM) and multiple myeloma and bone metastases of solid tumors. The drug is effective in treating cancer caused by HCM, advanced bone metastases and Paget’s disease, reduce the incidence of skeletal related events, relieve symptoms and improve quality of life, is also expected to be used to treat osteoporosis. Compared with other similar drugs, high efficacy, dosage, ease of administration, better security, etc., is currently the only FDA-approved for metastatic bone tumor effective bisphosphonate drugs.Currently bisphosphonate drugs in our country is still in the initial stages of clinical applications, but in recent years has made rapid progress, broad market prospect.
[0003] The prior art synthesis reaction conditions zoledronate sodium harsh, toxicity and use methanol, chloroform and chlorobenzene, easily exceeding the amount of residual organic solvents, low yield, low product purity, contamination environment, does not meet the medical criteria, is not conducive to industrial production. Environmental pollution has attracted increasing attention around the world today, the development of new green efficient synthesis of a pharmaceutical drug synthesis is an important issue facing the Institute. In recent years, room temperature ionic liquids as a reaction medium is environmentally friendly, has been widely used in a variety of organic synthesis reactions. Compared with traditional organic solvents, ionic liquids have very low vapor pressure, non-flammable, good thermal stability, both as a reaction medium underway catalysis, can be recycled and many other advantages.

Example 1 of zoledronic alendronate
(1) Synthesis of imidazol-1-yl acetate were added successively imidazole (13.62g, 0.2mol) and [bmim] BF4 (IOOmL) a three-necked flask, heated with stirring warmed to 60 ° C, incubated under reflux was slowly added dropwise chlorination ethyl acetate (24. 51g, 0. 2mol), dropwise addition time is about 2h, dropwise, with stirring maintained at reflux for 16 h, the reaction monitored by TLC showed no starting material end point, completion of the reaction, cooled to room temperature, to give imidazole -1 – ethyl crude, about 24g, crude without purification, was used directly in the next reaction.
[0014] (2) Synthesis of imidazol-1-yl acetate hydrochloride A solution of 24g 1-yl imidazole prepared above was added crude ethyl necked flask, concentrated hydrochloric acid (34 mL), exotherm to 85 ° C, warmed to reflux heating was continued, the reaction was stirred at reflux for 10H, the reaction was completed, the solvent was evaporated under reduced pressure, 20ml of absolute ethanol was added to the residue, vigorously stirred for 2h, filtered off with suction, the filter cake finally at 80 ° C blast pressure and dried to give a white solid imidazol-1-yl acetate hydrochloride about 25. 65g, 79.4% overall yield.
[0015] (3) Synthesis of zoledronic acid monohydrate were added imidazol-1-yl acetate hydrochloride (17. 26g, 0. 137mol) a three-necked flask, in an ionic liquid with stirring – n-butyl-3- methylimidazolium tetrafluoroborate [bmim] BF4 (40mL) and concentration of 85% phosphoric acid solution (16mL), heating to 60 ° C was added dropwise phosphorus trichloride (30mL) , about 4h dropwise, reaction was continued under reflux for 4h at 65 ° C, the reaction was complete, cooled to 40 ° C, filtered off with suction, the filter cake was added to a molar concentration in 80mL 9mol / L hydrochloric acid, heated with stirring state the reaction was refluxed for 6h, the reaction was completed, filtered hot, the filter cake was added to a molar concentration in 80mL 9mol / L hydrochloric acid, the above-described operation is repeated to continue the combined filtrate was evaporated to dryness under reduced pressure to give a yellow oily residue was slowly added to the residue volume ratio of 1: 1 acetone – ethanol mixture 240 mL, was stirred, and the precipitated solid was 15min, filtered off with suction, the filter cake was recrystallized in 30mL of deionized water, suction filtered to give a white solid that is zoledronic acid monohydrate , about 35. 8g, yield 90.1%, determined by HPLC, purity> 98.5%.
After [0016] (4) Synthesis of zoledronic sodium phosphinate obtained above azole zoledronic acid monohydrate (46.4g, 0. 16mol) washed with water (450 mL of) was dissolved, was added sodium hydroxide (5. 6g, 0. IOmol), were refluxed for 30min, cooling and crystallization, filtration, to obtain a crude product zoledronate sodium, crude mother liquor was concentrated and then half with distilled water (410 mL), isopropanol (60 mL), heated to dissolve the combined, activated carbon bleaching, charcoal filtered off, cooling and crystallization, filtration, washed with water, dried at 40-60 ° C to about crystallization water containing one to give zoledronate sodium (42. 4g, 85%), total yield of more than 60% by HPLC assay, purity 99. 8%, mp239 ° C. IR: 711011 ^ 671 (^ 1 is the stretching vibration peak of the PC, 1643〇 ^ 1 = 0 (: stretching vibration peak, 3011〇 ^ 1 = (: – stretching vibration peak 11 ^ 1 is CN 1406〇 the stretching vibration, 1643CHT1 is C = N stretching vibration peak of 3447 (3485 ^^ (^ 1 is the stretching vibration peak of OH, 1459CHT1 symmetrical bending vibration of CH, 2830CHT1 stretching vibration of CH, 1324CHT1 is P = O the stretching vibration, 1094CHT1 stretching vibration peak of .1HNMR PO (400MHz, D20), S: 8.68 (lH, s), 7.48 (lH, s), 7.34 (lH, s), 4.67 (2H, t).
Effects [0017] Example 2 was added dropwise phosphorus trichloride fixed time on the yield other conditions remain unchanged, only the changes of phosphorus trichloride dropwise addition, dropping zoledronic Table 1 Effect of Sodium yield Experimental results show that excessive phosphorus trichloride was added dropwise, and instantly generate a large amount of gas, the reaction is very intense, the liquid splashing, a rapid rise in temperature, resulting in the low yield, if slowly added dropwise, the reaction rate is too slow, consumption too long, and therefore is the best 4h dropping time.
Zoledronic fixed effect of sodium yield other conditions remain unchanged, imidazol-1-yl acetic acid hydrochloride with phosphorus trichloride and phosphoric acid condensation reaction temperature is zoledronic acid monohydrate embodiment the reaction temperature Example 3 Effect yield (Table 2). The results show that, with increasing temperature, increasing the yield, but at higher temperatures to reflux, shows a decreasing trend in yield, due to decomposition sake phosphorus trichloride, resulting in reduction reaction. Further, when the temperature is too high, solvent evaporation and solvent leakage losses will increase, the reflux temperature is low, the reaction rate is slow, the reaction is insufficient, therefore the yield is low, and therefore the optimum reaction temperature is about 65 ° C.
Example 4
Effect of the ionic liquid frequency reuse sodium zoledronic yield of the reaction medium can be recovered and reused important concern is “green chemistry” used in the present embodiment examines the sodium ionic liquid used in the synthesis of zoledronic repeated use, the experiment results shown in Table 3. Seen from Table 3, the ionic liquid after 5 subsequent to use, product yield began to decrease, the ionic liquid may be recovered and reused effectively, and repeated five times using good performance, and therefore is an ionic liquid in this reaction green solvents may be recycled.
Example 5 different ionic liquids zoledronic same impact conditions were examined yield sodium 1-butyl-3-methylimidazolium tetrafluoroborate ionic liquids ([bmim] BF4), N- ethyl pyridinium tetrafluoroborate ([EPy] BF4), l- butyl-3-methylimidazolium hexafluorophosphate ([bmim] PF6), 1- hydroxyethyl-2,3-dimethyl imidazolium chloride (LOH), 1- propyl-3-carbonitrile methylimidazolium chloride (the LCN) and 1-carboxyethyl-3-methyl imidazolium chloride (LOOH) Effects of sodium zoledronic yield the results are shown in Table 4, the test results show little effect on the synthesis of ionic liquids yield.
Clip
Mar 5, 2013 –
Dr. Reddy’s Laboratories announced today that it has launched Zoledronic Acid Injection (4 mg/5 mL), a bioequivalent generic version of Zometa® (zoledronic acid) 4 mg/5 mL Injection in the US market on March 4, 2013, following the approval by the United States Food & Drug Administration (USFDA) of Dr. Reddy’s ANDA for Zoledronic Acid Injection (4 mg/5 mL).
Dr. Reddy’s Zoledronic Acid Injection 4 mg/5mL is available in a single use vial of concentrate.
Zoledronic acid (INN) or zoledronate (marketed by Novartis under the trade names Zometa, Zomera, Aclasta and Reclast) is a bisphosphonate. Zometa is used to prevent skeletal fractures in patients with cancers such as multiple myeloma and prostate cancer, as well as for treating osteoporosis.It can also be used to treat hypercalcemia of malignancy and can be helpful for treating pain from bone metastases.
An annual dose of zoledronic acid may also prevent recurring fractures in patients with a previous hip fracture.
Reclast is a single 5 mg infusion for the treatment of Paget’s disease of bone. In 2007, the U.S. Food and Drug Administration (FDA) also approved Reclast for the treatment of postmenopausal osteoporosis.

About Dr. Reddy’s Laboratories Ltd.
Dr. Reddy’s Laboratories Ltd. (NYSE: RDY) is an integrated global pharmaceutical company, committed to providing affordable and innovative medicines for healthier lives. Through its three businesses – Pharmaceutical Services and Active Ingredients, Global Generics and Proprietary Products – Dr. Reddy’s offers a portfolio of products and services including APIs, custom pharmaceutical services, generics, biosimilars, differentiated formulations and NCEs. Therapeutic focus is on gastro-intestinal, cardiovascular, diabetology, oncology, pain management, anti-infective and pediatrics. Major markets include India, USA, Russia and CIS, Germany, UK, Venezuela, S. Africa, Romania, and New Zealand. For more information, log on to: http://www.drreddys.com
Zometa® is a registered trademark of Novartis AG
References
-
- US 4 939 130 (Ciba-Geigy; 3.7.1990; CH-prior. 21.11.1986).
-
transdermal formulation:
- EP 407 344 (Ciba-Geigy; appl. 28.6.1990; CH-prior. 7.7.1989).
-
treatment of angiogenesis:
- WO 2 000 071 104 (Novartis AG; appl. 19.5.2000; GB-prior. 21.5.1999).
PATENT
References
- ^ Jump up to:a b c d e f g h i j k “Zoledronic Acid”. The American Society of Health-System Pharmacists. Retrieved 8 December 2017.
- Jump up^ Drugs.com International trade names for zoledronic acid Page accessed Jan 14, 2015
- Jump up^ “WHO Model List of Essential Medicines (20th List)” (PDF). World Health Organization. March 2017. Retrieved 29 June 2017.
- Jump up^ “Single Drug Information”. International Medical Products Price Guide. Retrieved 9 December 2017.
- Jump up^ British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. p. 528. ISBN 9780857111562.
- Jump up^ National Prescribing Service (2009). “Zoledronic Acid for Osteoporosis”. Medicines Update, Available at “Archived copy”. Archived from the original on April 23, 2010. Retrieved January 20, 2010.
- Jump up^ http://www.health.gov.il/units/pharmacy/trufot/alonim/533.pdf Zomera prescribing information
- Jump up^ Wardley, A; Davidson, N; Barrett-Lee, P; et al. (May 2005). “Zoledronic acid significantly improves pain scores and quality of life in breast cancer patients with bone metastases: a randomised, crossover study of community vs hospital bisphosphonate administration”. Br. J. Cancer. 92 (10): 1869–76. doi:10.1038/sj.bjc.6602551. PMC 2361764
 . PMID 15870721.
. PMID 15870721. - ^ Jump up to:a b Lyles K, et al. (2007). “Zoledronic Acid and Clinical Fractures and Mortality after Hip Fracture”. N. Engl. J. Med. 357 (18): 1799–809. doi:10.1056/NEJMoa074941. PMC 2324066
 . PMID 17878149.
. PMID 17878149. - Jump up^ “Biotech PRESS RELEASE: Novartis’s Reclast Receives FDA Approval FOR Women With Postmenopausal Osteoporosis”, FierceBiotech, A Division of Questex A FierceMarkets Publication Aug 20, 2007. Retrieved 2018-03-27
- ^ Jump up to:a b Black; et al. (2007). “Once-Yearly Zoledronic Acid for Treatment of Postmenopausal Osteoporosis”. NEJM. 356 (18): 1809–1822. doi:10.1056/nejmoa067312. PMID 17476007.
- Jump up^ “Paget’s Disease of Bone”. www.rheumatology.org. Retrieved 2015-07-09.
- Jump up^ Vondracek, S. F. (2010). “Managing osteoporosis in postmenopausal women”. American Journal of Health-System Pharmacy. 67 (7 Suppl 3): S9–19. doi:10.2146/ajhp100076. PMID 20332498.
- Jump up^ http://www.nps.org.au/__data/assets/pdf_file/0006/60945/nvcaclin.pdf
- Jump up^ “Zometa 4mg/5ml Concentrate for Solution for Infusion”. medicines.org.uk.
- Jump up^ “FDA Alert: Reclast (zoledronic acid): Drug Safety Communication – New Contraindication and Updated Warning on Kidney Impairment”. drugs.com.
- Jump up^ “European Medicines Agency – Human medicines”. europa.eu.
- Jump up^ Durie BG, Katz M, Crowley J (2005). “Osteonecrosis of the jaw and bisphosphonates”. N. Engl. J. Med. 353 (1): 99–102; discussion 99–102. doi:10.1056/NEJM200507073530120. PMID 16000365.
- ^ Jump up to:a b “European Medicines Agency – Human medicines”. europa.eu.
- Jump up^ Aclasta label- Australia
- Jump up^ Koto K, Murata H, Kimura S, et al. (July 2010). “Zoledronic acid inhibits proliferation of human fibrosarcoma cells with induction of apoptosis, and shows combined effects with other anticancer agents”. Oncol. Rep. 24 (1): 233–9. doi:10.3892/or_00000851. PMID 20514467.
- Jump up^ Reid IR, Brown JP, Burckhardt P, Horowitz Z, Richardson P, Trechsel U, Widmer A, Devogelaer JP, Kaufman JM, Jaeger P, Body JJ, Brandi ML, Broell J, Di Micco R, Genazzani AR, Felsenberg D, Happ J, Hooper MJ, Ittner J, Leb G, Mallmin H, Murray T, Ortolani S, Rubinacci A, Saaf M, Samsioe G, Verbruggen L, Meunier PJ (2002). “Intravenous zoledronic acid in postmenopausal women with low bone mineral density”. N. Engl. J. Med. 346 (9): 653–61. doi:10.1056/NEJMoa011807. PMID 11870242.
- Jump up^ Juhi Misra, Sindhu T. Mohanty, Sanjeev Madan, James A. Fernandes, F. Hal Ebetino, R. Graham, G. Russell, Ilaria Bellantuono. (December 2015). Zoledronate attenuates accumulation of DNA damage in mesenchymal stem cells and protects their function. Stem Cells, doi:10.1002/stem.2255
- Jump up^ “Bone drug protects stem cells from aging.” ScienceDaily. 17 December 2015
- Jump up^ PMID 19213681 Gnant, Mlineritsch. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med 2009; 360:679-691 February 12, 2009 Full Free Text [1]
- Jump up^ Coleman RE, Winter MC, Cameron D, et al. (March 2010). “The effects of adding zoledronic acid to neoadjuvant chemotherapy on tumour response: exploratory evidence for direct anti-tumour activity in breast cancer”. Br. J. Cancer. 102 (7): 1099–105. doi:10.1038/sj.bjc.6605604. PMC 2853093
 . PMID 20234364.
. PMID 20234364. - Jump up^ Brufsky A, Bundred N, Coleman R, et al. (May 2008). “Integrated analysis of zoledronic acid for prevention of aromatase inhibitor-associated bone loss in postmenopausal women with early breast cancer receiving adjuvant letrozole”. Oncologist. 13 (5): 503–14. doi:10.1634/theoncologist.2007-0206. PMID 18515735.
- Jump up^ Tonyali O, Arslan C, Altundag K (November 2010). “The role of zoledronic acid in the adjuvant treatment of breast cancer: current perspectives”. Expert Opin Pharmacother. 11(16): 2715–25. doi:10.1517/14656566.2010.523699. PMID 20977404.
- Jump up^ Delea TE, Taneja C, Sofrygin O, Kaura S, Gnant M (August 2010). “Cost-effectiveness of zoledronic acid plus endocrine therapy in premenopausal women with hormone-responsive early breast cancer”. Clin. Breast Cancer. 10 (4): 267–74. doi:10.3816/CBC.2010.n.034. PMID 20705558.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Reclast, Zometa, others[2] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605023 |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
Intravenous |
| Drug class | Bisphosphonate[1] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 22% |
| Metabolism | Nil |
| Elimination half-life | 146 hours |
| Excretion | Kidney (partial) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| Chemical and physical data | |
| Formula | C5H10N2O7P2 |
| Molar mass | 272.09 g/mol |
| 3D model (JSmol) | |
Judith Aronhime, Revital Lifshitz-Liron, “Zoledronic acid crystal forms, zoledronate sodium salt crystal forms, amorphous zoledronate sodium salt, and processes for their preparation.” U.S. Patent US20050054616, issued March 10, 2005., US20050054616
/////////////////disodium zoledronate tetrahydrate, zoledronic acid, ZOMETA, CGP-42446, CGP-42446A
OC(CN1C=CN=C1)(P(O)(O)=O)P(O)(O)=O