
Olutasidenib
- FT-2102
- FT2102
C18H15ClN4O2
354.79
CAS1887014-12-1
Rezlidhia (Forma Therapeutics)
SYN Caravella JA, et al. Structure-Based Design and Identification of FT-2102 (Olutasidenib), a Potent Mutant-Selective IDH1 Inhibitor. J Med Chem. 2020 Feb 27;63(4):1612-1623. doi: 10.1021/acs.jmedchem.9b01423. Epub 2020 Feb 12.
FDA 12/1/2022, To treat adults with relapsed or refractory acute myeloid leukemia with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation, Rezlidhia
Olutasidenib, sold under the brand name Rezlidhia, is an anticancer medication used to treat relapsed or refractory acute myeloid leukemia.[1][2] Olutasidenib is an isocitrate dehydrogenase-1 (IDH1) inhibitor.[1] It is taken by mouth.[1]
Olutasidenib was approved for medical use in the United States in December 2022.[1][2][3][4]
Medical uses
Olutasidenib is indicated for the treatment of adults with relapsed or refractory acute myeloid leukemia with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test.[1][2]
Society and culture
Names
Olutasidenib is the international nonproprietary name.[5]
Olutasidenib is an isocitrate dehydrogenase-1 (IDH1) inhibitor indicated for the treatment of patients with relapsed or refractory acute myeloid leukemia with a susceptible IDH1 mutation as detected by an FDA-approved test.
Olutasidenib (FT-2102) is a selective and potent isocitrate dehydrogenase-1 (IDH1) inhibitor approved by the FDA in December 2022.5,6 It is indicated for the treatment of relapsed or refractory acute myeloid leukemia (AML) in patients with a susceptible IDH1 mutation as determined by an FDA-approved test.5 IDH1 mutations are common in different types of cancer, such as gliomas, AML, intrahepatic cholangiocarcinoma, chondrosarcoma, and myelodysplastic syndromes (MDS), and they lead to an increase in 2-hydroxyglutarate (2-HG), a metabolite that participates in tumerogenesis.1,2 Olutasidenib inhibits the mutated IDH1 specifically, and provides a therapeutic benefit in IDH1-mutated cancers.1,5
Other IDH1 inhibitors, such as ivosidenib, have also been approved for the treatment of relapsed or refractory AML.3,4 Olutasidenib is orally bioavailable and capable of penetrating the blood-brain barrier, and is also being evaluated for the treatment of myelodysplastic syndrome (MDS), as well as solid tumors and gliomas (NCT03684811).4
SYN
https://pubs.acs.org/doi/10.1021/acs.jmedchem.9b01423
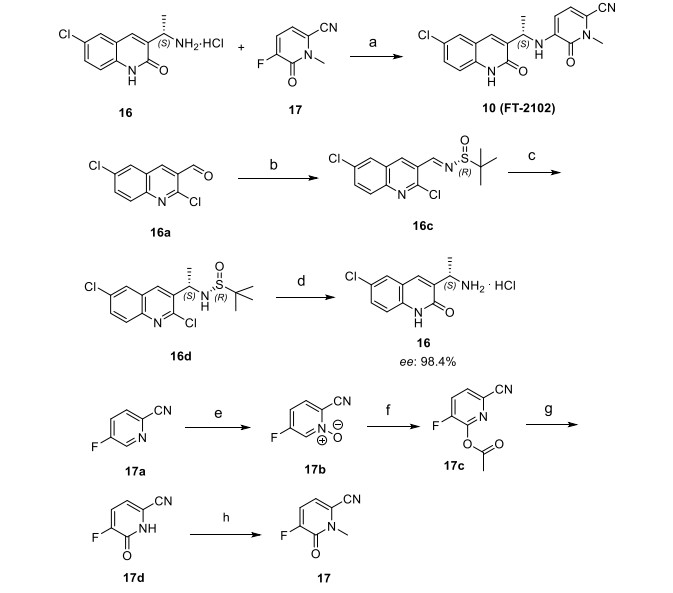
a Reagents and conditions: (a) DIEA, DMSO, 80−110 °C, 16 h, 67%; (b) (R)-2-methylpropane-2-sulfinamide, CuSO4, 55 °C, DCE, 16 h, 81%; (c) MeMgBr, DCM, −50 to −60 °C, 3 h, 63%; (d) 1 N HCl, dioxane, reflux, 16 h, >98%, 98.4% ee; (e) m-CPBA, CHCl3, reflux, 4 days, 52%; (f) Ac2O, reflux, 3 days, 60%; (g) K2CO3, MeOH, 4 h, 92%; (h) MeI, K2CO3, DMF, 45 min, 67%.
1H NMR (300 MHz,
DMSO-d6) δ 12.07 (s, 1 H), 7.71−7.76 (m, 2 H), 7.51 (dd, J = 8.79,
2.35 Hz, 1 H), 7.31 (d, J = 8.79 Hz, 1 H), 6.97 (d, J = 7.92 Hz, 1 H),
6.93 (d, J = 7.92 Hz, 1 H), 5.95 (d, J = 7.92 Hz, 1 H), 4.62−4.75 (m,
1 H), 3.58 (s, 3 H), 1.50 (d, J = 6.74 Hz, 3 H); 13C NMR (75 MHz,
DMSO-d6) δ 161.0, 155.9, 141.4, 136.6, 135.0, 133.4, 129.8, 126.7,
125.8, 120.1, 119.4, 116.7, 115.1, 104.5, 103.7, 47.4, 34.0, 20.3; LCMS
(method 2) >95% purity; tR 10.18 min; m/z 355, 357 [M + H]+
;
HRMS (ESI) calcd for C18H16ClN4O2 [M + H]+ 355.0962 found
356.0956.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
 |
|
| Clinical data | |
|---|---|
| Trade names | Rezlidhia |
| Other names | FT-2102 |
| License data |
|
| Routes of administration |
By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| Chemical and physical data | |
| Formula | C18H15ClN4O2 |
| Molar mass | 354.79 g·mol−1 |
| 3D model (JSmol) | |
References
- ^ Jump up to:a b c d e f https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215814s000lbl.pdf
- ^ Jump up to:a b c d https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/215814Orig1s000ltr.pdf
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Rigel Announces U.S. FDA Approval of Rezlidhia (olutasidenib) for the Treatment of Adult Patients with Relapsed or Refractory Acute Myeloid Leukemia with a Susceptible IDH1 Mutation”. Rigel Pharmaceuticals, Inc. (Press release). 1 December 2022. Retrieved 2 December 2022.
- ^ “Rigel Announces U.S. FDA Approval of Rezlidhia (olutasidenib) for the Treatment of Adult Patients with Relapsed or Refractory Acute Myeloid Leukemia with a Susceptible IDH1 Mutation” (Press release). Rigel Pharmaceuticals. 1 December 2022. Retrieved 2 December 2022 – via PR Newswire.
- ^ World Health Organization (2019). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 82”. WHO Drug Information. 33 (3). hdl:10665/330879.
Further reading
- Liu X, Gong Y (2019). “Isocitrate dehydrogenase inhibitors in acute myeloid leukemia”. Biomarker Research. 7: 22. doi:10.1186/s40364-019-0173-z. PMC 6806510. PMID 31660152.
- Watts JM, Baer MR, Yang J, Prebet T, Lee S, Schiller GJ, et al. (November 2022). “Olutasidenib alone or with azacitidine in IDH1-mutated acute myeloid leukaemia and myelodysplastic syndrome: phase 1 results of a phase 1/2 trial”. The Lancet Haematology. doi:10.1016/S2352-3026(22)00292-7. PMID 36370742. S2CID 253471380.
External links
- “Olutasidenib”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT02719574 for “Open-label Study of FT-2102 With or Without Azacitidine or Cytarabine in Patients With AML or MDS With an IDH1 Mutation” at ClinicalTrials.gov
/////////////Olutasidenib, FDA 2022, APPROVALS 2022, Rezlidhia, FT-2102, FT 2102














