Technetium (99mTc) tetrofosmin, 99mTc-Tetrofosmin
テトロホスミンテクネチウム (99mTc)
| Formula | C36H80O10P4Tc |
|---|---|
| Molar mass | 895.813 g/mol |
| CAS Number |
|
|---|
UNII42FOP1YX93
2-[bis(2-ethoxyethyl)phosphanyl]ethyl-bis(2-ethoxyethyl)phosphane;technetium-98;dihydrate
Technetium Tc 99m tetrofosmin; Technetium Tc-99m tetrofosmin; TECHNETIUM TC-99M TETROFOSMIN KIT; Tc-99m tetrofosmin; Technetium-99 tetrofosmin; Technetium (99mTc) tetrofosmin
Technetium Tc-99m Tetrofosmin is a radiopharmaceutical consisting of tetrofosmin, composed of two bidentate diphosphine ligands chelating the metastable radioisotope technetium Tc-99 (99mTc), with potential imaging activity upon SPECT (single photon emission computed tomography). Upon administration, technetium Tc 99m tetrofosmin is preferentially taken up by, and accumulates in, myocardial cells. Upon imaging, myocardial cells can be visualized and changes in ischemia and/or perfusion can be detected.
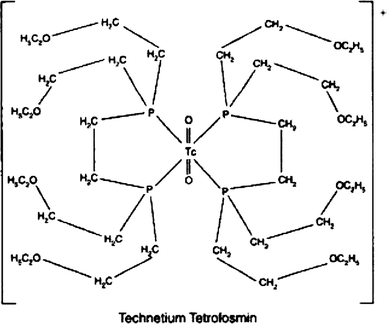
Technetium Tc-99m tetrofosmin is a drug used in nuclear myocardial perfusion imaging. The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin. It is a lipophilic technetium phosphine dioxo cation that was formulated into a freeze-dried kit which yields an injection.[A31592] Technetium Tc-99m tetrofosmin was developed by GE Healthcare and FDA approved on February 9, 1996.
Technetium Tc-99m tetrofosmin is a drug used in nuclear myocardial perfusion imaging. The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin. It is a lipophilic technetium phosphine dioxo cation that was formulated into a freeze-dried kit which yields an injection.[1] Technetium Tc-99m tetrofosmin was developed by GE Healthcare and FDA approved on February 9, 1996.
Technetium (99mTc) tetrofosmin is a drug used in nuclear medicine cardiac imaging. It is sold under the brand name Myoview (GE Healthcare). The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin.[1][2]
Tc-99m tetrofosmin is rapidly taken up by myocardial tissue and reaches its maximum level in approximately 5 minutes. About 66% of the total injected dose is excreted within 48 hours after injection (40% urine, 26% feces). Tc-99m tetrofosmin is indicated for use in scintigraphic imaging of the myocardium under stress and rest conditions. It is used to determine areas of reversible ischemia and infarcted tissue in the heart. It is also indicated to detect changes in perfusion induced by pharmacologic stress (adenosine, lexiscan, dobutamine or persantine) in patients with coronary artery disease. Its third indication is to assess left ventricular function (ejection fraction) in patients thought to have heart disease. No contraindications are known for use of Tc-99m tetrofosmin, but care should be taken to constantly monitor the cardiac function in patients with known or suspected coronary artery disease. Patients should be encouraged to void their bladders as soon as the images are gathered, and as often as possible after the tests to decrease their radiation doses, since the majority of elimination is renal. The recommended dose of Tc-99m tetrofosmin is between 5 and 33 millicuries (185-1221 megabecquerels). For a two-dose stress/rest dosing, the typical dose is normally a 10 mCi dose, followed one to four hours later by a dose of 30 mCi. Imaging normally begins 15 minutes following injection.[3]

Amersham (formerly Nycomed Amersham , now GE Healthcare ) has developed and launched 99mTc-tetrofosmin (Myoview) as an injectable nuclear imaging agent for ischemic heart disease in several major territories and for use in detecting breast tumors
Technetium (99mTc) tetrofosmin is a drug used in nuclear medicine cardiac imaging. It is sold under the brand name Myoview (GE Healthcare). The radioisotope, technetium-99m, is chelated by two 1, 2-bis-[bis-(2-ethoxyethyl)phosphino] ethane ligands, which belong to the group of diphosphines and which are referred to as tetrofosmin and has the structural Formula 1 :
Formula 1
99mTc -based radiopharmaceuticals are commonly used in diagnostic nuclear medicine, especially for in vivo imaging (e.g. via immunoscintigraphy or radiolabeling). Usually cold kits are manufactured in advance in accordance with strict requirements of Good Manufacturing Practice (GMP) Guidelines, containing the chemical ingredients (e.g. 99mTc -coordinating ligands, preservatives) in lyophilized form. The radioactive isotope 99mTc (ti/2 = 6h) is added to those kits shortly before application to the patient via intravenous or subcutaneous injection.
Tc-99m tetrofosmin is rapidly taken up by myocardial tissue and reaches its maximum level in approximately 5 minutes. About 66% of the total injected dose is excreted within 48 hours after injection (40% urine, 26% feces). Tc-99m tetrofosmin is indicated for use in scintigraphic imaging of the myocardium under stress and rest conditions. It is used to determine areas of reversible ischemia and infarcted tissue in the heart. It is also indicated to detect changes in perfusion induced by pharmacologic stress (adenosine, lexiscan, dobutamine or persantine) in patients with coronary artery disease. Its third indication is to assess left ventricular function (ejection fraction) in patients thought to have heart disease. No contraindications are known for use of Tc-99m tetrofosmin, but care should be taken to constantly monitor the cardiac function in patients with known or suspected coronary artery disease. Patients should be encouraged to void their bladders as soon as the images are gathered, and as often as possible after the tests to decrease their radiation doses, since the majority of elimination is renal. The recommended dose of Tc-99m tetrofosmin is between 5 and 33 millicuries (185-1221 megabecquerels). For a two-dose stress/rest dosing, the typical dose is normally a 10 mCi dose, followed one to four hours later by a dose of 30 mCi. Imaging normally begins 15 minutes following injection.
99mTc -Tetrofosmin is also described to be useful for tumor diagnostics, in particular of breast cancer and parathyroid gland cancer, and for multidrug resistance (MDR) research.
US5045302 discloses 99mTc-coordinating diphosphine ligands (L), wherein one preferred example thereof is the ether functionalized diphosphine ligand l,2-bis[bis(2-ethoxy- ethyl)phosphino]ethane according to Formula 1, called tetrofosmin (“P53”), that forms a dimeric cationic technetium (V) dioxo phosphine complex, [TCO2L2] with 99mTc, useful as myocardial imaging agent. Example 1 of said patent described the process for preparing tetrofosmin by reacting ethyl vinyl ether, bis(diphosphino)ethane in the presence of a-azo-isobutyronitrile (AIBN) in a fischer pressure-bottle equipped with a teflon stirring bar followed by removal of volatile materials and non-distillable material obtained, as per below mentioned Scheme 1.
Scheme 1
Formula 2 Formula 3 Formula 1
CN 1184225 C discloses tetrofosmin salts containing chloride or bromide or aryl sulfonates as negatively charged counter ions, which can be used for the preparation of a 99mTc- Tetrofosmin radiopharmaceutical composition. According to this patent tetrofosmin hydrochloride is a viscous liquid. Own experiments of the inventors of the present invention revealed that the halide salts of tetrofosmin are hygroscopic oils, which are complicated to handle, e.g. when weighed. The oily and hygrospcopic
properties of tetrofosmin hydrochloride hampers its use in pharmaceutical preparations. Attempts to synthesize the subsalicylate salt of tetrofosmin failed because the starting material sulfosalicylic acid was not soluble in ether in the concentration specified in the patent (3.4 g in 15 ml).
WO2006/064175A1 discloses tetrofosmin was converted to tetrofosmin subsalicylate by reaction with 2.3 to 2.5 molar equivalents of 5-sulfosalicyclic acid at room temperature in ethanol, followed by recrystallisation from ethanol/ether.
WO2015/114002A1 relates to tetrafluoroborate salt of tetrafosmin and its process for the preparation thereof. Further this application also discloses one-vial and two vial kit formulation with tetrafluoroborate salt of tetrafosmin.
The article Proceedings of the International Symposium, 7th, Dresden, Germany, June 18-22, 2000 by Amersham Pharmacia Biotech UK Limited titled “The synthesis of [14C]tetrofosmin, a compound vital to the development of Myoview, Synthesis and Applications of Isotopically Labelled Compounds” disclosed a process for the preparation of tetrofosmin as per below mentioned Scheme 2:
Scheme 2
Formula 1A Formula 7
The starting material was bis(2- ethoxyethyl)benzylphosphine of Formula 4 . This was prepared from benzyl phosphonate, PhCH2P(0)(OEt)2 by reduction with lithium aluminium hydride to give the intermediate benzylphosphine, PhCH2PH2, followed by a photolysis reaction in the presence of ethyl vinyl ether to give compound of Formula 4. The compound of Formula 4 in acetonitrile was treated with dibromo[U-14C]ethane to give compound of Formula 6, further it was treated with excess of 30% aqueous sodium hydroxide in ethanol. The mixture was stirred at room temperature for 24 hours. The solvent was removed and the residue was treated with excess concentrated hydrochloric acid at 0°C. Aqueous work up gave compound of Formula 7. Then compound of Formula 7 in dry benzene was treated with hexachlorodisilane and hydrolysed with excess 30% aqueous sodium hydroxide at 0°C. Aqueous work up followed by flash column chromatography on silica gave [bisphosphinoethane- 1,2-14C]tetrofosmin of formula 1A.
The article Polyhedron (1995), 14(8), 1057-65, titled “Synthesis and characterization of Group 10 metal complexes with a new trifunctional ether phosphine. The X-ray crystal structures of bis[bis(2-ethoxyethyl)benzylphosphine]dichloronickel(II) and bis[bis(2-ethoxyethyl)benzylphosphine]chlorophenylnickel(II)” disclosed the process for the preparation of bis(2-ethoxyethyl)benzylphosphine as per below mentioned Scheme 3:
Scheme 3
Formula 8 Formula 9 Formula 4
The compound bis(2-ethoxyethyl)benzylphosphine of Formula 4 was prepared by first reduction of diethylbenzylphosphonate of Formula 8 using lithium aluminium hydride to obtain benzyl phosphine of Formula 9 followed by radical catalysed coupling reaction with ethyl vinyl ether carried out by using UV photolysis.
Tetrofosmin is extremely sensitive to atmospheric oxygen, which makes synthesis of the substance, as well as manufacturing and handling of the kit complicated as the substance has constantly to be handled in an oxygen free atmosphere.
High purity and stability under dry and controlled conditions are pivotal requirements for chemical compounds used as active ingredients in pharmaceuticals.
The processes disclosed in prior art for the preparation of compound of Formula 4 involves that coupling reaction of benzyl phosphine of Formula 9 with ethyl vinyl ether carried out by using photolytic conditions. Such technology is expensive as it requires separate instruments including isolated facility (to avoid the UV radiation exposure etc.), also it is not suitable for commercial scale production.
PATENT
WO-2018162964
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018162964&tab=PCTDESCRIPTION&maxRec=1000
Example 1
Preparation of benzyl phosphine:
A mixture of lithium aluminium hydride (25 g) in methyl tertiary butyl ether (MTBE) (800 ml) was cooled to 0 to 5°C and added a solution of diethylbenzylphosphonate in methyl tertiary butyl ether (100 g in 200ml). The temperature of reaction mixture was raised to 25 to 30 °C and stirred for 14 to 16 hour. After completion of the reaction, the reaction mixture was cooled to 0 to 5°C and 6N hydrochloric acid was added slowly. Further raised the temperature of reaction mixture to 25 to 30 °C and stirred for 30-45 minutes. The layers were separated, the aqueous layer was extracted with MTBE (250ml) and the combined organic layer was washed with deoxygenated water. The organic layer was dried over sodium sulfate and concentrated to obtain the title compound as non-distillable liquid.
Example 2
Preparation of benzylbis(2-ethoxyethyl)phosphane:
To a mixture of benzyl phosphine (obtained from example 1) and vinyl ethyl ether (250 ml) in pressure RB flask was added a-azo-isobutyronitrile (AIBN) (1.5g). The resulting reaction mixture was maintained at 80 to 90°C for 14 to 16 hours. The mixture was cooled to 20 to 30°C and AIBN (0.5g) added, then continued to heat the reaction mixture at 80 to 90°C for 6 to 7 hours. After completion of the reaction, the reaction mixture was allowed to cool to room temperature and distilled under vacuum to obtain title compound as an oil (107 g).
Example 3
Preparation of Ethane- 1,2-diylbis (benzylbis(2-ethoxyethyl) phosphonium) bromide:
To a mixture of benzylbis(2-ethoxyethyl)phosphane 107.g) in acetonitrile (100ml) in pressure bottle was added 1, 2-dibromoethane (30.5 g). The reaction mixture was maintained at 80 to 90°C for 20 to 25 hours. After completion of the reaction, the reaction mass was cooled to room temperature and stirred for 45 to 60 minutes to obtain the solid. To the solid obtained was added methyl tertiary butyl ether (MTBE) (500ml) and stirred at room temperature for 2 to 3 hour. The reaction mass was filtered, washed with MTBE and suck dried. Further the filtered solid was heated in acetone (400ml) at 50 to 55°C for 2 to 3 hour. Then cooled the reaction mixture to room temperature, stirred, filtered and washed with acetone to obtain the title compound as white solid. (85g)
Example 4
Preparation of Ethane- 1, 2-diylbis (bis (2-ethoxy ethyl) phosphine oxide):
To a mixture of Ethane- 1,2-diylbis (benzylbis(2-ethoxyethyl) phosphonium) bromide (80g) in ethanol (480 ml) was added an aq. solution of sodium hydroxide ( 48g in 160 ml water) at room temperature. The reaction mass was maintained at 25 to 35°C for 10 to 12 hour. After completion of the reaction, the reaction mass was cone, under vacuum to obtained the residue. The residue was dissolved in deoxygenated water (400 ml) and washed with MTBE (400 ml x 2). The layers were separated, the aqueous layer was cooled to 10 to 20°C and 6N hydrochloric acid (200 ml) was added slowly. Then extracted the aqueous layer with dichloromethane (2000 ml), washed the organic layer with deoxygenated water (160 ml), dried the organic layer using sodium sulfate, filtered, and distilled under vacuum to obtain the residue. Further MTBE (160 ml x 2) was added to the residue and continued distillation under vacuum, degassed to obtain the solid. To the obtained solid, MTBE (400 ml) was added and heated at 45 to 50°C for 1-2 hour, further slowly cooled the reaction mass to 25 to 30°C, filtered the solid product. Again MTBE (400 ml) was added to the solid product and heated at 45 to 50°C for 1-2 hour, further slowly cooled the reaction mass to 25 to 30°C, filtered, washed with MTBE and dried under vacuum to obtain the title compound as white solid (32g).
Example 5
Preparation of tetrofosmin free base:
To a mixture of ethane- 1, 2-diylbis (bis (2-ethoxyethyl) phosphine oxide (18g) in toluene (180ml) in pressure RB flask argon/nitrogen gas was purged for 5 minute and hexachlorodisilane (30g) was added. The reaction mixture was heated to 80 to 90°C, stirred for 10 to 12 hour, further slowly cooled to -5 to 0°C and slowly added 30% aqueous sodium hydroxide solution (45g sodium hydroxide in 150 ml deoxygenated water) the temperature of reaction mixture was raised to 25 to 30°C and stirred for 1 to 2 hour. The layers were separated and the aq. layer was extracted with Toluene (180 ml). The combined organic layer was washed with deoxygenated water (180 ml). Further dried the organic layer using sodium sulfate, distilled under vacuum to obtain the residue of tetrofosmin free base (15.5g).
Example 6
Preparation of tetrofosmin disulfosalicylate salt:
To the residue of tetrofosmin free base (15.5g) was added an aq. solution of 5-sulfosalicylic acid dihydrate (21.6g in 75ml deoxygenated water) and stirred at 25 to 30°C for 25 to 30 minutes. Further heated the reaction mass to 55 to 60°C, stirred for 15 to 30 minute, slowly cooled the reaction mass to 10 to 15°C and stirred for 1-2 hour. Filtered, washed with chilled deoxygenated water, and dried under vacuum to obtain the title compound as white solid. (30g).
Example 7
Preparation of Form J of tetrofosmin disulfosalicylate salt:
An aq. solution of 5-sulfosalicylic acid dihydrate (21.6g in 75ml deoxygenated water) was added slowly into tetrofosmin free base (15.5g) and stirred at room temperature for 30 to 40 minutes. The temperature of reaction mixture was further raised to 50 to 60°C, stirred for 20 to 30 minute, cooled the reaction mass to 10 to 15°C and stirred for 1-2 hour. Filtered, washed with chilled deoxygenated water, and dried under vacuum to obtain the title compound.
PATENT
EP337654 ,
PATENT
US9549999
FDA Orange Book Patents
| FDA Orange Book Patents: 1 of 1 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 9549999 |
| Expiration | Mar 10, 2030 |
| Applicant | GE HEALTHCARE |
| Drug Application |
|
References
- Jump up^ Kelly JD, Alan M. Forster AM, Higley B, et al. (February 1993). “Technetium-99m-Tetrofosmin as a new radiopharmaceutical for myocardial perfusion imaging”. Journal of Nuclear Medicine. 34 (2): 222–227. PMID 8429340.
- Jump up^ Elhendy A, Schinkel AF, et al. (December 2005). “Risk stratification of patients with angina pectoris by stress 99mTc-tetrofosmin myocardial perfusion imaging”. Journal of Nuclear Medicine. 46 (12): 2003–2008. PMID 16330563.
- Jump up^ Myoview package insert. Arlington Heights, IL: GE Healthcare, 2006, Aug.
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Intravenous |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Identifiers | |
| CAS Number |
|
| Chemical and physical data | |
| Formula | C36H80O10P4Tc |
| Molar mass | 895.813 g/mol |
External links
Myoview Prescribing Information Page
//////////99mTc-Tetrofosmin, Technetium (99mTc) tetrofosmin, テトロホスミンテクネチウム (99mTc)
CCOCCP(CCOCC)CCP(CCOCC)CCOCC.CCOCCP(CCOCC)CCP(CCOCC)CCOCC.O.O.[Tc]