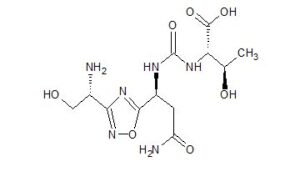
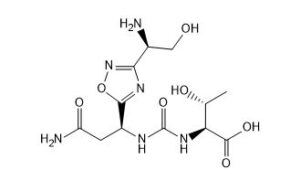
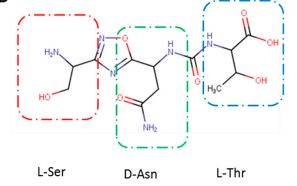
(2S,3R)-2-(3-((S)-3-amino-1-(3-((R)-1-amino-2-hydroxyethyl)-1,2,4-oxadiazol-5-yl)-3-oxopropyl)ureido)-3-hydroxybutanoic acid
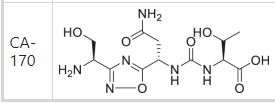
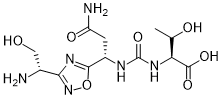

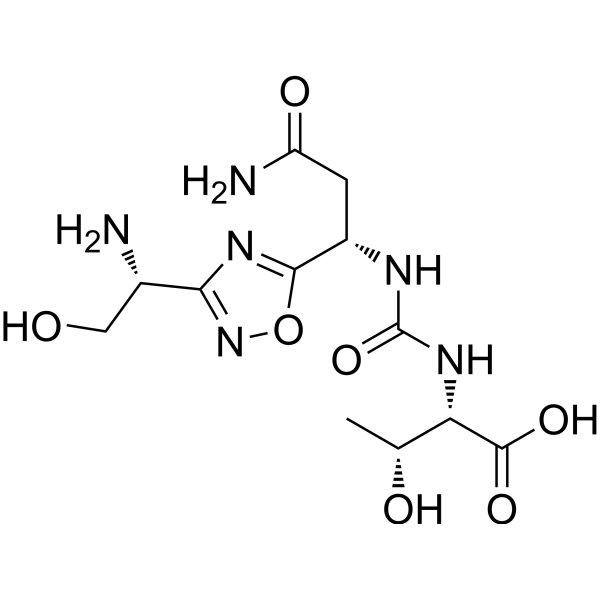
| Molecular Weight (MW) | 360.33 |
|---|---|
| Formula | C12H20N6O7 |
| CAS No. | 1673534-76-3 |
N-[[[(1S)-3-Amino-1-[3-[(1R)-1-amino-2-hydroxyethyl]-1,2,4-oxadiazol-5-yl]-3-oxopropyl]amino]carbonyl]-L-threonine
L-Threonine, N-[[[(1S)-3-amino-1-[3-[(1R)-1-amino-2-hydroxyethyl]-1,2,4-oxadiazol-5-yl]-3-oxopropyl]amino]carbonyl]-
AUPM 170, CA 170, AUPM-170, CA-170, PD-1-IN-1
Novel inhibitor of programmed cell dealth-1 (PD-1)
CA-170 (also known as AUPM170 or PD-1-IN-1) is a first-in-class, potent and orally available small molecule inhibitor of the immune checkpoint regulatory proteins PD-L1 (programmed cell death ligand-1), PD-L2 and VISTA (V-domain immunoglobulin (Ig) suppressor of T-cell activation (programmed death 1 homolog; PD-1H). CA-170 was discovered by Curis Inc. and has potential antineoplastic activities. CA-170 selectively targets PD-L1 and VISTA, both of which function as negative checkpoint regulators of immune activation. Curis is currently investigating CA-170 for the treatment of advanced solid tumours and lymphomas in patients in a Phase 1 trial (ClinicalTrials.gov Identifier: NCT02812875).
References: www.clinicaltrials.gov (NCT02812875); WO 2015033299 A1 20150312.
Aurigene Discovery Technologies Limited INNOVATOR

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
CURIS AND AURIGENE ANNOUNCE AMENDMENT OF COLLABORATION FOR THE DEVELOPMENT AND COMMERCIALIZATION OF CA-170
PRESS RELEASE
Curis and Aurigene Announce Amendment of Collaboration for the Development and Commercialization of CA-170
– Aurigene to fund and conduct a Phase 2b/3 randomized study of CA-170 in patients with non-squamous non-small cell lung cancer (nsNSCLC) –
– Aurigene to receive Asia rights for CA-170; Curis entitled to royalty payments in Asia –
LEXINGTON, Mass., February 5, 2020 /PRNewswire/ — Curis, Inc. (NASDAQ: CRIS), a biotechnology company focused on the development of innovative therapeutics for the treatment of cancer, today announced that it has entered into an amendment of its collaboration, license and option agreement with Aurigene Discovery Technologies, Ltd. (Aurigene). Under the terms of the amended agreement, Aurigene will fund and conduct a Phase 2b/3 randomized study evaluating CA-170, an orally available, dual
inhibitor of VISTA and PDL1, in combination with chemoradiation, in approximately 240 patients with nonsquamous
non-small cell lung cancer (nsNSCLC). In turn, Aurigene receives rights to develop and commercialize CA-170 in Asia, in addition to its existing rights in India and Russia, based on the terms of the original agreement. Curis retains U.S., E.U., and rest of world rights to CA-170, and is entitled to receive royalty payments on potential future sales of CA-170 in Asia.
In 2019, Aurigene presented clinical data from a Phase 2a basket study of CA-170 in patients with multiple tumor types, including those with nsNSCLC. In the study, CA-170 demonstrated promising signs of safety and efficacy in nsNSCLC patients compared to various anti-PD-1/PD-L1 antibodies.
“We are pleased to announce this amendment which leverages our partner Aurigene’s expertise and resources to support the clinical advancement of CA-170, as well as maintain our rights to CA-170 outside of Asia,” said James Dentzer, President and Chief Executive Officer of Curis. “Phase 2a data presented at the European Society for Medical Oncology (ESMO) conference last fall supported the potential for CA-170 to serve as a therapeutic option for patients with nsNSCLC. We look forward to working with our partner Aurigene to further explore this opportunity.”
“Despite recent advancements, patients with localized unresectable NSCLC struggle with high rates of recurrence and need for expensive intravenous biologics. The CA-170 data presented at ESMO 2019 from Aurigene’s Phase 2 ASIAD trial showed encouraging results in Clinical Benefit Rate and Prolonged PFS and support its potential to provide clinically meaningful benefit to Stage III and IVa nsNSCLC patients, in combination with chemoradiation and as oral maintenance” said Kumar Prabhash, MD, Professor of Medical Oncology at Tata Memorial Hospital, Mumbai, India.
Murali Ramachandra, PhD, Chief Executive Officer of Aurigene, commented, “Development of CA-170, with its unique dual inhibition of PD-L1 and VISTA, is the result of years of hard-work and commitment by many people, including the patients who participated in the trials, caregivers and physicians, along with the talented teams at Aurigene and Curis. We look forward to further developing CA-170 in nsNSCLC.”
About Curis, Inc.
Curis is a biotechnology company focused on the development of innovative therapeutics for the treatment of cancer, including fimepinostat, which is being investigated in combination with venetoclax in a Phase 1 clinical study in patients with DLBCL. In 2015, Curis entered into a collaboration with Aurigene in the areas of immuno-oncology and precision oncology. As part of this collaboration, Curis has exclusive licenses to oral small molecule antagonists of immune checkpoints including, the VISTA/PDL1 antagonist CA-170, and the TIM3/PDL1 antagonist CA-327, as well as the IRAK4 kinase inhibitor, CA- 4948. CA-4948 is currently undergoing testing in a Phase 1 trial in patients with non-Hodgkin lymphoma.
In addition, Curis is engaged in a collaboration with ImmuNext for development of CI-8993, a monoclonal anti-VISTA antibody. Curis is also party to a collaboration with Genentech, a member of the Roche Group, under which Genentech and Roche are commercializing Erivedge® for the treatment of advanced basal cell carcinoma. For more information, visit Curis’ website at www.curis.com.
About Aurigene
Aurigene is a development stage biotech company engaged in discovery and clinical development of novel and best-in-class therapies to treat cancer and inflammatory diseases and a wholly owned subsidiary of Dr. Reddy’s Laboratories Ltd. (BSE: 500124, NSE: DRREDDY, NYSE: RDY). Aurigene is focused on precision- oncology, oral immune checkpoint inhibitors, and the Th-17 pathway. Aurigene currently has several programs from its pipeline in clinical development. Aurigene’s ROR-gamma inverse agonist AUR-101 is currently in phase 2 clinical development under a US FDA IND. Additionally, Aurigene has multiple compounds at different stages of pre-clinical development. Aurigene has partnered with many large and mid-pharma companies in the United States and Europe and has 15 programs currently in clinical development. For more information, please visit Aurigene’s website at https://www.aurigene.com/
Curis with the option to exclusively license Aurigene’s orally-available small molecule antagonist of programmed death ligand-1 (PD-L1) in the immuno-oncology field
Addressing immune checkpoint pathways is a well validated strategy to treat human cancers and the ability to target PD-1/PD-L1 and other immune checkpoints with orally available small molecule drugs has the potential to be a distinct and major advancement for patients.
Through its collaboration with Aurigene, Curis is now engaged in the discovery and development of the first ever orally bioavailable, small molecule antagonists that target immune checkpoint receptor-ligand interactions, including PD-1/PD-L1 interactions. In the first half of 2016, Curis expects to file an IND application with the U.S. FDA to initiate clinical testing of CA-170, the first small molecule immune checkpoint antagonist targeting PD-L1 and VISTA. The multi-year collaboration with Aurigene is focused on generation of small molecule antagonists targeting additional checkpoint receptor-ligand interactions and Curis expects to advance additional drug candidates for clinical testing in the coming years. The next immuno-oncology program in the collaboration is currently targeting the immune checkpoints PD-L1 and TIM3.
In November 2015, preclinical data were reported. Data demonstrated tha the drug rescued and sustained activation of T cells functions in culture. CA-170 resulted in anti-tumor activity in multiple syngeneic tumor models including melanoma and colon cancer. Similar data were presented at the 2015 AACR-NCI-EORTC Molecular Targets and Cancer Therapeutics Conference in Boston, MA
By August 2015, preclinical data had been reported. Preliminary data demonstrated that in in vitro studies, small molecule PD-L1 antagonists induced effective T cell proliferation and IFN-gamma production by T cells that were specifically suppressed by PD-L1 in culture. The compounds were found to have effects similar to anti-PD1 antibodies in in vivo tumor models
(Oral Small Molecule PD-L1/VISTAAntagonist)
Certain human cancers express a ligand on their cell surface referred to as Programmed-death Ligand 1, or PD-L1, which binds to its cognate receptor, Programmed-death 1, or PD-1, present on the surface of the immune system’s T cells. Cell surface interactions between tumor cells and T cells through PD-L1/PD-1 molecules result in T cell inactivation and hence the inability of the body to mount an effective immune response against the tumor. It has been previously shown that modulation of the PD-1 mediated inhibition of T cells by either anti-PD1 antibodies or anti-PD-L1 antibodies can lead to activation of T cells that result in the observed anti-tumor effects in the tumor tissues. Therapeutic monoclonal antibodies targeting the PD-1/PD-L1 interactions have now been approved by the U.S. FDA for the treatment of certain cancers, and multiple therapeutic monoclonal antibodies targeting PD-1 or PD-L1 are currently in development.
In addition to PD-1/PD-L1 immune regulators, there are several other checkpoint molecules that are involved in the modulation of immune responses to tumor cells1. One such regulator is V-domain Ig suppressor of T-cell activation or VISTA that shares structural homology with PD-L1 and is also a potent suppressor of T cell functions. However, the expression of VISTA is different from that of PD-L1, and appears to be limited to the hematopoietic compartment in tissues such as spleen, lymph nodes and blood as well as in myeloid hematopoietic cells within the tumor microenvironment. Recent animal studies have demonstrated that combined targeting/ blockade of PD-1/PD-L1 interactions and VISTA result in improved anti-tumor responses in certain tumor models, highlighting their distinct and non-redundant functions in regulating the immune response to tumors2.
As part of the collaboration with Aurigene, in October 2015 Curis licensed a first-in-class oral, small molecule antagonist designated as CA-170 that selectively targets PD-L1 and VISTA, both of which function as negative checkpoint regulators of immune activation. CA-170 was selected from the broad PD-1 pathway antagonist program that the companies have been engaged in since the collaboration was established in January 2015. Preclinical data demonstrate that CA-170 can induce effective proliferation and IFN-γ (Interferon-gamma) production (a cytokine that is produced by activated T cells and is a marker of T cell activation) by T cells that are specifically suppressed by PD-L1 or VISTA in culture. In addition, CA-170 also appears to have anti-tumor effects similar to anti-PD-1 or anti-VISTA antibodies in multiple in vivo tumor models and appears to have a good in vivo safety profile. Curis expects to file an IND and initiate clinical testing of CA-170 in patients with advanced tumors during the first half of 2016.
 Jan 21, 2015
Jan 21, 2015
Curis and Aurigene Announce Collaboration, License and Option Agreement to Discover, Develop and Commercialize Small Molecule Antagonists for Immuno-Oncology and Precision Oncology Targets
— Agreement Provides Curis with Option to Exclusively License Aurigene’s Antagonists for Immuno-Oncology, Including an Antagonist of PD-L1 and Selected Precision Oncology Targets, Including an IRAK4 Kinase Inhibitor —
— Investigational New Drug (IND) Application Filings for Both Initial Collaboration Programs Expected this Year —
— Curis to issue 17.1M shares of its Common Stock as Up-front Consideration —
— Management to Host Conference Call Today at 8:00 a.m. EST —
LEXINGTON, Mass. and BANGALORE, India, Jan. 21, 2015 (GLOBE NEWSWIRE) — Curis, Inc. (Nasdaq:CRIS), a biotechnology company focused on the development and commercialization of innovative drug candidates for the treatment of human cancers, and Aurigene Discovery Technologies Limited, a specialized, discovery stage biotechnology company developing novel therapies to treat cancer and inflammatory diseases, today announced that they have entered into an exclusive collaboration agreement focused on immuno-oncology and selected precision oncology targets. The collaboration provides for inclusion of multiple programs, with Curis having the option to exclusively license compounds once a development candidate is nominated within each respective program. The partnership draws from each company’s respective areas of expertise, with Aurigene having the responsibility for conducting all discovery and preclinical activities, including IND-enabling studies and providing Phase 1 clinical trial supply, and Curis having responsibility for all clinical development, regulatory and commercialization efforts worldwide, excluding India and Russia, for each program for which it exercises an option to obtain a license.
The first two programs under the collaboration are an orally-available small molecule antagonist of programmed death ligand-1 (PD-L1) in the immuno-oncology field and an orally-available small molecule inhibitor of Interleukin-1 receptor-associated kinase 4 (IRAK4) in the precision oncology field. Curis expects to exercise its option to obtain exclusive licenses to both programs and file IND applications for a development candidate from each in 2015.
“We are thrilled to partner with Aurigene in seeking to discover, develop and commercialize small molecule drug candidates generated from Aurigene’s novel technology and we believe that this collaboration represents a true transformation for Curis that positions the company for continued growth in the development and eventual commercialization of cancer drugs,” said Ali Fattaey, Ph.D., President and Chief Executive Officer of Curis. “The multi-year nature of our collaboration means that the parties have the potential to generate a steady pipeline of novel drug candidates in the coming years. Addressing immune checkpoint pathways is now a well validated strategy to treat human cancers and the ability to target PD-1/PD-L1 and other immune checkpoints with orally available small molecule drugs has the potential to be a distinct and major advancement for patients. Recent studies have also shown that alterations of the MYD88 gene lead to dysregulation of its downstream target IRAK4 in a number of hematologic malignancies, including Waldenström’s Macroglobulinemia and a subset of diffuse large B-cell lymphomas, making IRAK4 an attractive target for the treatment of these cancers. We look forward to advancing these programs into clinical development later this year.”
Dr. Fattaey continued, “Aurigene has a long and well-established track record of generating targeted small molecule drug candidates with bio-pharmaceutical collaborators and we have significantly expanded our drug development capabilities as we advance our proprietary drug candidates in currently ongoing clinical studies. We believe that we are well-positioned to advance compounds from this collaboration into clinical development.”
CSN Murthy, Chief Executive Officer of Aurigene, said, “We are excited to enter into this exclusive collaboration with Curis under which we intend to discover and develop a number of drug candidates from our chemistry innovations in the most exciting fields of cancer therapy. This unique collaboration is an opportunity for Aurigene to participate in advancing our discoveries into clinical development and beyond, and mutually align interests as provided for in our agreement. Our scientists at Aurigene have established a novel strategy to address immune checkpoint targets using small molecule chemical approaches, and have discovered a number of candidates that modulate these checkpoint pathways, including PD-1/PD-L1. We have established a large panel of preclinical tumor models in immunocompetent mice and can show significant in vivo anti-tumor activity using our small molecule PD-L1 antagonists. We are also in the late stages of selecting a candidate that is a potent and selective inhibitor of the IRAK4 kinase, demonstrating excellent in vivo activity in preclinical tumor models.”
In connection with the transaction, Curis has issued to Aurigene approximately 17.1 million shares of its common stock, or 19.9% of its outstanding common stock immediately prior to the transaction, in partial consideration for the rights granted to Curis under the collaboration agreement. The shares issued to Aurigene are subject to a lock-up agreement until January 18, 2017, with a portion of the shares being released from the lock-up in four equal bi-annual installments between now and that date.
The agreement provides that the parties will collaborate exclusively in immuno-oncology for an initial period of approximately two years, with the option for Curis to extend the broad immuno-oncology exclusivity.
In addition Curis has agreed to make payments to Aurigene as follows:
- for the first two programs: up to $52.5 million per program, including $42.5 million per program for approval and commercial milestones, plus specified approval milestone payments for additional indications, if any;
- for the third and fourth programs: up to $50 million per program, including $42.5 million per program for approval and commercial milestones, plus specified approval milestone payments for additional indications, if any; and
- for any program thereafter: up to $140.5 million per program, including $87.5 million per program in approval and commercial milestones, plus specified approval milestone payments for additional indications, if any.
Curis has agreed to pay Aurigene royalties on any net sales ranging from high single digits to 10% in territories where it successfully commercializes products and will also share in amounts that it receives from sublicensees depending upon the stage of development of the respective molecule.
About Immune Checkpoint Modulation and Programmed Death 1 Pathway
Modulation of immune checkpoint pathways has emerged as a highly promising therapeutic approach in a wide range of human cancers. Immune checkpoints are critical for the maintenance of self-tolerance as well as for the protection of tissues from excessive immune response generated during infections. However, cancer cells have the ability to modulate certain immune checkpoint pathways as a mechanism to evade the immune system. Certain immune checkpoint receptors or ligands are expressed by various cancer cells, targeting of which may be an effective strategy for generating anti-tumor activity. Some immune-checkpoint modulators, such as programmed death 1 (PD-1) protein, specifically regulate immune cell effector functions within tissues. One of the mechanisms by which tumor cells block anti-tumor immune responses in the tumor microenvironment is by upregulating ligands for PD-1, such as PD-L1. Hence, targeting of PD-1 and/or PD-L1 has been shown to lead to the generation of effective anti-tumor responses.
About Curis, Inc.
Curis is a biotechnology company focused on the development and commercialization of novel drug candidates for the treatment of human cancers. Curis’ pipeline of drug candidates includes CUDC-907, a dual HDAC and PI3K inhibitor, CUDC-427, a small molecule antagonist of IAP proteins, and Debio 0932, an oral HSP90 inhibitor. Curis is also engaged in a collaboration with Genentech, a member of the Roche Group, under which Genentech and Roche are developing and commercializing Erivedge®, the first and only FDA-approved medicine for the treatment of advanced basal cell carcinoma. For more information, visit Curis’ website at www.curis.com.
About Aurigene
Aurigene is a specialized, discovery stage biotechnology company, developing novel and best-in-class therapies to treat cancer and inflammatory diseases. Aurigene’s Programmed Death pathway program is the first of several immune checkpoint programs that are at different stages of discovery and preclinical development. Aurigene has partnered with several large- and mid-pharma companies in the United States and Europe and has delivered multiple clinical compounds through these partnerships. With over 500 scientists, Aurigene has collaborated with 6 of the top 10 pharma companies. Aurigene is an independent, wholly owned subsidiary of Dr. Reddy’s Laboratories Ltd. (NYSE:RDY). For more information, please visit Aurigene’s website at http://aurigene.com/.
POSTER
WO2011161699, WO2012/168944, WO2013144704 and WO2013132317 report peptides or peptidomimetic compounds which are capable of suppressing and/or inhibiting the programmed cell death 1 (PD1) signaling pathway.
PATENT
Inventors
- SASIKUMAR, Pottayil Govindan Nair
- RAMACHANDRA, Muralidhara
- NAREMADDEPALLI, Seetharamaiah Setty Sudarshan
Priority Data
| 4011/CHE/2013 | 06.09.2013 | IN |
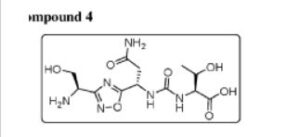
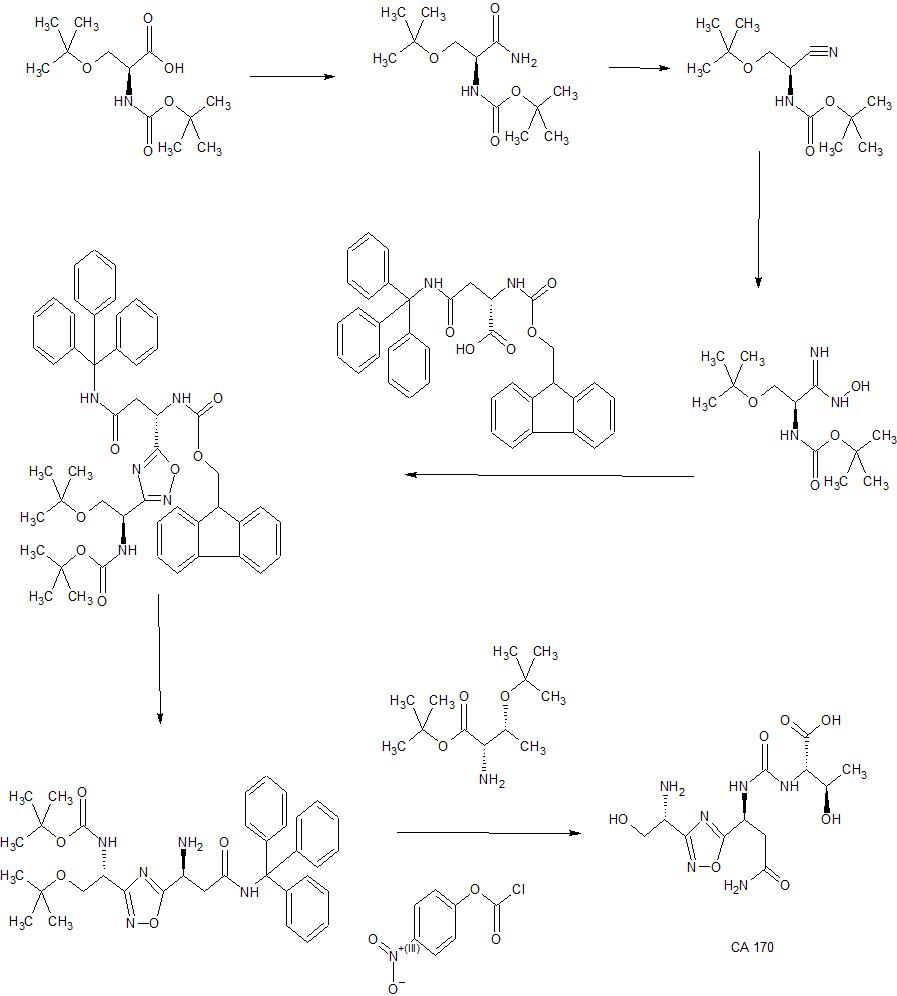
Example 4: Synthesis of Co
The compound was synthesised using similar procedure as depicted in Example 2 for synthesising compound 2 using
instead of H-Ser(‘Bu)-0’Bu (in synthesis of compound 2b) to yield 0.35 g crude material of the title compound. The crude solid material was purified using preparative HPLC described under experimental conditions. LCMS: 361.2 (M+H)+, HPLC: tR = 12.19 min.


REFERENCES
US20150073024
| WO2011161699A2 | 27 Jun 2011 | 29 Dec 2011 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| WO2012168944A1 | 21 Dec 2011 | 13 Dec 2012 | Aurigene Discovery Technologies Limited | Therapeutic compounds for immunomodulation |
| WO2013132317A1 | 4 Mar 2013 | 12 Sep 2013 | Aurigene Discovery Technologies Limited | Peptidomimetic compounds as immunomodulators |
| WO2013144704A1 | 28 Mar 2013 | 3 Oct 2013 | Aurigene Discovery Technologies Limited | Immunomodulating cyclic compounds from the bc loop of human pd1 |
http://www.curis.com/images/stories/pdfs/posters/Aurigene_PD-L1_VISTA_AACR-NCI-EORTC_2015.pdf
References:
1) https://bmcimmunol.biomedcentral.com/articles/10.1186/s12865-021-00446-4
2) https://www.nature.com/articles/s42003-021-02191-1
3) https://www.esmoopen.com/article/S2059-7029(20)30108-3/fulltext
4) https://www.mdpi.com/1420-3049/24/15/2804
////////Curis, Aurigene, AUPM 170, CA 170, AUPM-170, CA-170, PD-L1, VISTA antagonist, PD-1-IN-1, phase 2, CANCER
N[C@@H](CO)c1nc(on1)[C@@H](NC(=O)N[C@H](C(=O)O)[C@@H](C)O)CC(N)=O