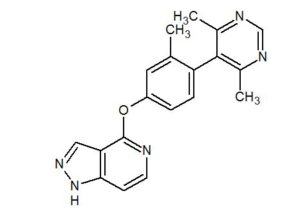
PF 2562
CAS 1609258-91-4
MF C19 H17 N5 O


Jennifer Elizabeth Davoren
Principal Scientist at Pfizer
SYNTHESIS

-
Dopamine acts upon neurons through two families of dopamine receptors, D1-like receptors (D1Rs) and D2-like receptors (D2Rs). The D1-like receptor family consists of D1 and D5 receptors which are expressed in many regions of the brain. D1 mRNA has been found, for example, in the striatum and nucleus accumbens. See e.g., Missale C, Nash S R, Robinson S W, Jaber M, Caron M G “Dopamine receptors: from structure to function”, Physiological Reviews 78:189-225 (1998). Pharmacological studies have reported that D1 and D5 receptors (D1/D5), namely D1-like receptors, are linked to stimulation of adenylyl cyclase, whereas D2, D3, and D4 receptors, namely D2-like receptors, are linked to inhibition of cAMP production.
-
Dopamine D1 receptors are implicated in numerous neuropharmacological and neurobiological functions. For example, D1 receptors are involved in different types of memory function and synaptic plasticity. See e.g., Goldman-Rakic P S et al., “Targeting the dopamine D1 receptor in schizophrenia: insights for cognitive dysfunction”, Psychopharmacology 174(1):3-16 (2004). Moreover, D1 receptors have been implicated in a variety of psychiatric, neurological, neurodevelopmental, neurodegenerative, mood, motivational, metabolic, cardiovascular, renal, ophthalmic, endocrine, and/or other disorders described herein including schizophrenia (e.g., cognitive and negative symptoms in schizophrenia), cognitive impairment associated with D2 antagonist therapy, ADHD, impulsivity, autism spectrum disorder, mild cognitive impairment (MCI), age-related cognitive decline, Alzheimer’s dementia, Parkinson’s disease (PD), Huntington’s chorea, depression, anxiety, treatment-resistant depression (TRD), bipolar disorder, chronic apathy, anhedonia, chronic fatigue, post-traumatic stress disorder, seasonal affective disorder, social anxiety disorder, post-partum depression, serotonin syndrome, substance abuse and drug dependence, Tourette’s syndrome, tardive dyskinesia, drowsiness, sexual dysfunction, migraine, systemic lupus erythematosus (SLE), hyperglycemia, dislipidemia, obesity, diabetes, sepsis, post-ischemic tubular necrosis, renal failure, resistant edema, narcolepsy, hypertension, congestive heart failure, postoperative ocular hypotonia, sleep disorders, pain, and other disorders in a mammal. See e.g., Goulet M, Madras B K “D(1) dopamine receptor agonists are more effective in alleviating advanced than mild parkinsonism in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated monkeys”, Journal of Pharmacology and Experimental Therapy 292(2):714-24 (2000); Surmeier D J et al., “The role of dopamine in modulating the structure and function of striatal circuits”, Prog. Brain Res. 183:149-67 (2010).New or improved agents that modulate (such as agonize or partially agonize) D1 are needed for developing new and more effective pharmaceuticals to treat diseases or conditions associated with dysregulated activation of D1, such as those described herein.
PATENT
Example 6
4-[4-(4,6-Dimethylpyrimidin-5-yl)-3-methylphenoxy]-1H-pyrazolo[4,3-c]pyridine (6)
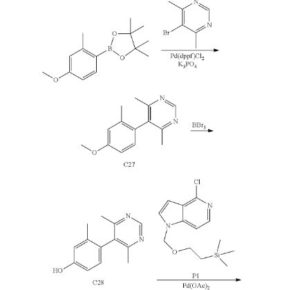
Step 1. Synthesis of 4-[4-(4,6-dimethylpyrimidin-5-yl)-3-methylphenoxy]-1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazolo[4,3-c]pyridine (C31)
Cesium carbonate (1.03 g, 3.16 mmol) and palladium(II) acetate (24 mg, 0.11 mmol) were added to a solution of C28 (225 mg, 1.05 mmol) and P3 (250 mg, 1.05 mmol) in 1,4-dioxane (10 mL) in a sealable reaction vessel, and the solution was purged with nitrogen for 10 minutes. Di-tert-butyl[3,4,5,6-tetramethyl-2′,4′,6-tri(propan-2-yl)biphenyl-2-yl]phosphane (97%, 104 mg, 0.210 mmol) was added, and the reaction mixture was briefly purged with nitrogen. The vessel was sealed and the reaction mixture was stirred at 100° C. for 3 hours. After cooling to room temperature, the mixture was filtered through Celite and the filter pad was washed with ethyl acetate; the combined filtrates were concentrated in vacuo and purified via silica gel chromatography (Eluents: 20%, then 50%, then 100% ethyl acetate in heptane). The product was obtained as an off-white solid. Yield: 272 mg, 0.655 mmol, 62%. LCMS m/z 416.5 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 8.99 (s, 1H), 8.11 (d, J=0.6 Hz, 1H), 7.99 (d, J=6.0 Hz, 1H), 7.25-7.27 (m, 2H, assumed; partially obscured by solvent peak), 7.20-7.24 (m, 1H), 7.10 (d, J=8.4 Hz, 1H), 5.73 (dd, J=9.4, 2.5 Hz, 1H), 4.04-4.10 (m, 1H), 3.74-3.82 (m, 1H), 2.49-2.59 (m, 1H), 2.28 (s, 6H), 2.08-2.21 (m, 2H), 2.04 (s, 3H), 1.66-1.84 (s, 3H).
Step 2. Synthesis of 4-[4-(4,6-dimethylpyrimidin-5-yl)-3-methylphenoxy]-1H-pyrazolo[4,3-c]pyridine (6)
C31 (172 mg, 0.414 mmol) was dissolved in 1,4-dioxane (5 mL) and dichloromethane (5 mL), and cooled to 0° C. A solution of hydrogen chloride in 1,4-dioxane (4 M, 1.04 mL, 4.16 mmol) was added, and the reaction mixture was allowed to stir at room temperature for 45 hours. After removal of solvent in vacuo, the residue was partitioned between saturated aqueous sodium bicarbonate solution and dichloromethane. The aqueous layer was extracted twice with dichloromethane, and the combined organic layers were dried over sodium sulfate, filtered, and concentrated under reduced pressure, affording the product as an off-white solid. Yield: 130 mg, 0.392 mmol, 95%. LCMS m/z 332.3 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 9.00 (s, 1H), 8.20 (br s, 1H), 7.99 (d, J=6.0 Hz, 1H), 7.28-7.30 (m, 1H), 7.23-7.27 (m, 1H), 7.16 (dd, J=6.0, 1.0 Hz, 1H), 7.11 (d, J=8.2 Hz, 1H), 2.28 (s, 6H), 2.05 (s, 3H).
Preparation P8
6-(4-Hydroxy-2-methylphenyl)-1,5-dimethylpyrazin-2(1H)-one (P8)
Step 1. Synthesis of 1-(4-methoxy-2-methylphenyl)propan-2-one (C8)
Four batches of this experiment were carried out (4×250 g substrate). Tributyl(methoxy)stannane (400 g, 1.24 mol), 1-bromo-4-methoxy-2-methylbenzene (250 g, 1.24 mol), prop-1-en-2-yl acetate (187 g, 1.87 mol), palladium(II) acetate (7.5 g, 33 mmol) and tris(2-methylphenyl)phosphane (10 g, 33 mmol) were stirred together in toluene (2 L) at 100° C. for 18 hours. After cooling to room temperature, the reaction mixture was treated with aqueous potassium fluoride solution (4 M, 400 mL) and stirred for 2 hours at 40° C. The resulting mixture was diluted with toluene (500 mL) and filtered through Celite; the filter pad was thoroughly washed with ethyl acetate (2×1.5 L). The organic phase from the combined filtrates was dried over sodium sulfate, filtered, and concentrated in vacuo. Purification via silica gel chromatography (Gradient: 0% to 5% ethyl acetate in petroleum ether) provided the product as a yellow oil. Combined yield: 602 g, 3.38 mol, 68%. LCMS m/z 179.0 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 7.05 (d, J=8.3 Hz, 1H), 6.70-6.77 (m, 2H), 3.79 (s, 3H), 3.65 (s, 2H), 2.22 (s, 3H), 2.14 (s, 3H).
Step 2. Synthesis of 1-(4-methoxy-2-methylphenyl)propane-1,2-dione (C9)
C8 (6.00 g, 33.7 mmol) and selenium dioxide (7.47 g, 67.3 mmol) were suspended in 1,4-dioxane (50 mL) and heated at 100° C. for 18 hours. The reaction mixture was cooled to room temperature and filtered through Celite; the filtrate was concentrated in vacuo. Silica gel chromatography (Eluent: 10% ethyl acetate in heptane) afforded the product as a bright yellow oil. Yield: 2.55 g, 13.3 mmol, 39%. LCMS m/z 193.1 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 7.66 (d, J=8.6 Hz, 1H), 6.81 (br d, half of AB quartet, J=2.5 Hz, 1H), 6.78 (br dd, half of ABX pattern, J=8.7, 2.6 Hz, 1H), 3.87 (s, 3H), 2.60 (br s, 3H), 2.51 (s, 3H).
Step 3. Synthesis of 6-(4-methoxy-2-methylphenyl)-5-methylpyrazin-2(1H)-one (C10)
C9 (4.0 g, 21 mmol) and glycinamide acetate (2.79 g, 20.8 mmol) were dissolved in methanol (40 mL) and cooled to −10° C. Aqueous sodium hydroxide solution (12 N, 3.5 mL, 42 mmol) was added, and the resulting mixture was slowly warmed to room temperature. After stirring for 3 days, the reaction mixture was concentrated in vacuo. The residue was diluted with water, and 1 N aqueous hydrochloric acid was added until the pH was approximately 7. The aqueous phase was extracted with ethyl acetate, and the combined organic extracts were washed with saturated aqueous sodium chloride solution, dried over magnesium sulfate, filtered, and concentrated under reduced pressure. The resulting residue was slurried with 3:1 ethyl acetate/heptane, stirred for 5 minutes, filtered, and concentrated in vacuo. Silica gel chromatography (Eluent: ethyl acetate) provided the product as a tan solid that contained 15% of an undesired regioisomer; this material was used without further purification. Yield: 2.0 g. LCMS m/z 231.1 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 8.09 (s, 1H), 7.14 (d, J=8.2 Hz, 1H), 6.82-6.87 (m, 2H), 3.86 (s, 3H), 2.20 (s, 3H), 2.11 (s, 3H).
Step 4. Synthesis of 6-(4-methoxy-2-methylphenyl)-1,5-dimethylpyrazin-2(1H)-one (C11)
C10 (from the previous step, 1.9 g) was dissolved in N,N-dimethylformamide (40 mL). Lithium bromide (0.86 g, 9.9 mmol) and sodium bis(trimethylsilyl)amide (95%, 1.91 g, 9.89 mmol) were added, and the resulting solution was stirred for 30 minutes. Methyl iodide (0.635 mL, 10.2 mmol) was added and stirring was continued at room temperature for 18 hours. The reaction mixture was then diluted with water and brought to a pH of approximately 7 by slow portion-wise addition of 1 N aqueous hydrochloric acid. The aqueous layer was extracted with ethyl acetate and the combined organic layers were washed several times with water, dried over magnesium sulfate, filtered, and concentrated. Silica gel chromatography (Gradient: 75% to 100% ethyl acetate in heptane) afforded the product as a viscous orange oil. Yield: 1.67 g, 6.84 mmol, 33% over two steps. LCMS m/z 245.1 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 8.17 (s, 1H), 7.03 (br d, J=8 Hz, 1H), 6.85-6.90 (m, 2H), 3.86 (s, 3H), 3.18 (s, 3H), 2.08 (br s, 3H), 2.00 (s, 3H).
Step 5. Synthesis of P8
To a −78° C. solution of C11 (1.8 g, 7.37 mmol) in dichloromethane (40 mL) was added a solution of boron tribromide in dichloromethane (1 M, 22 mL, 22 mmol). The cooling bath was removed after 30 minutes, and the reaction mixture was allowed to warm to room temperature and stir for 18 hours. The reaction was cooled to −78° C., and methanol (10 mL) was slowly added; the resulting mixture was slowly warmed to room temperature. The reaction mixture was concentrated in vacuo, methanol (20 mL) was added, and the mixture was again concentrated under reduced pressure. The residue was diluted with ethyl acetate (300 mL) and water (200 mL) and the aqueous layer was brought to pH 7 via portion-wise addition of saturated aqueous sodium carbonate solution. The mixture was extracted with ethyl acetate (3×200 mL). The combined organic extracts were washed with water and with saturated aqueous sodium chloride solution, dried over magnesium sulfate, filtered, and concentrated in vacuo to afford the product as a light tan solid. Yield: 1.4 g, 6.0 mmol, 81%. LCMS m/z 231.1 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 8.21 (s, 1H), 6.98 (d, J=8.2 Hz, 1H), 6.87-6.89 (m, 1H), 6.85 (br dd, J=8.2, 2.5 Hz, 1H), 3.22 (s, 3H), 2.06 (br s, 3H), 2.03 (s, 3H).
Step 1. Synthesis of 5-(4-methoxy-2-methylphenyl)-4,6-dimethylpyrimidine (C27)
1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)-dichloromethane complex (5 g, 6 mmol) was added to a degassed mixture of 2-(4-methoxy-2-methylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (30 g, 120 mmol), 5-bromo-4,6-dimethylpyrimidine (22.5 g, 120 mmol), and potassium phosphate (76.3 g, 359 mmol) in 1,4-dioxane (300 mL) and water (150 mL). The reaction mixture was heated at reflux for 4 hours, whereupon it was filtered and concentrated in vacuo. Purification via silica gel chromatography (Gradient: ethyl acetate in petroleum ether) provided the product as a brown solid. Yield: 25 g, 110 mmol, 92%. LCMS m/z 229.3 [M+H+]. 1H NMR (300 MHz, CDCl3) δ 8.95 (s, 1H), 6.94 (d, J=8.2 Hz, 1H), 6.87-6.89 (m, 1H), 6.84 (dd, J=8.3, 2.5 Hz, 1H), 3.86 (s, 3H), 2.21 (s, 6H), 1.99 (s, 3H).
Step 2. Synthesis of 4-(4,6-dimethylpyrimidin-5-yl)-3-methylphenol (C28)
Boron tribromide (3.8 mL, 40 mmol) was added drop-wise to a solution of C27 (3.0 g, 13 mmol) in dichloromethane (150 mL) at −70° C. The reaction mixture was stirred at room temperature for 16 hours, then adjusted to pH 8 with saturated aqueous sodium bicarbonate solution. The aqueous layer was extracted with dichloromethane (3×200 mL), and the combined organic layers were dried over sodium sulfate, filtered, and concentrated in vacuo. Silica gel chromatography (Gradient: 60% to 90% ethyl acetate in petroleum ether) afforded the product as a yellow solid. Yield: 1.2 g, 5.6 mmol, 43%. LCMS m/z 215.0 [M+H+]. 1H NMR (400 MHz, CDCl3) δ 8.98 (s, 1H), 6.89 (d, J=8.0 Hz, 1H), 6.86 (d, J=2.3 Hz, 1H), 6.80 (dd, J=8.3, 2.5 Hz, 1H), 2.24 (s, 6H), 1.96 (s, 3H).
//////////////PF 2562, non-catechol dopamine 1 receptor agonist, PFIZER, Jennifer Elizabeth Davoren, Amy Beth Dounay, Ivan Viktorovich Efremov, David Lawrence Firman Gray, Scot Richard Mente, Steven Victor O’Neil, Bruce Nelsen Rogers, Chakrapani Subramanyam, Lei Zhang, 1609258-91-4
Now at #MEDI 1st time disclosures David Gray of @pfizer on a non-catechol dopamine 1 receptor agonist #ACSSanFran
Cc1ncnc(C)c1c2ccc(cc2C)Oc4nccc3nncc34